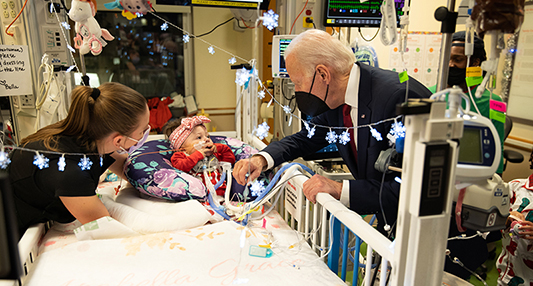Living tissue heart valve replaces mechanical mitral valve through partial heart transplant
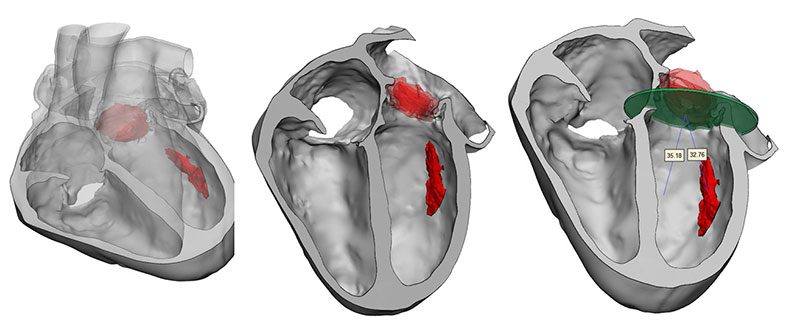
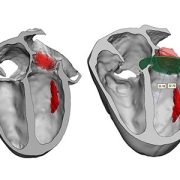
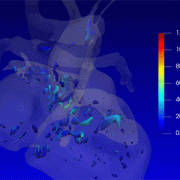
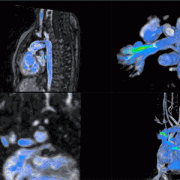
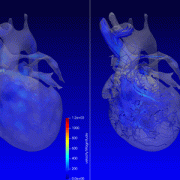
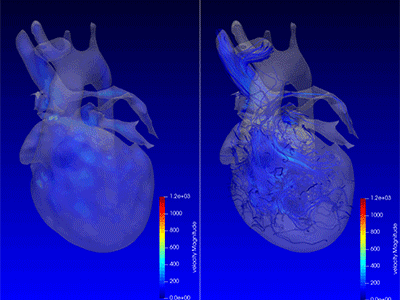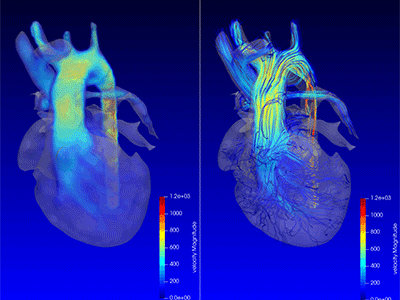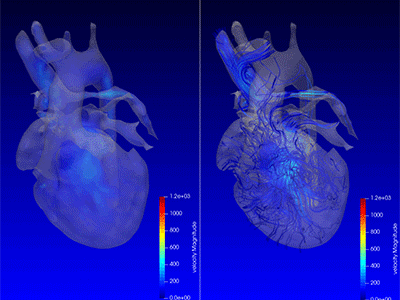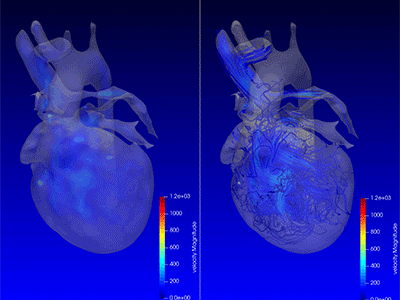
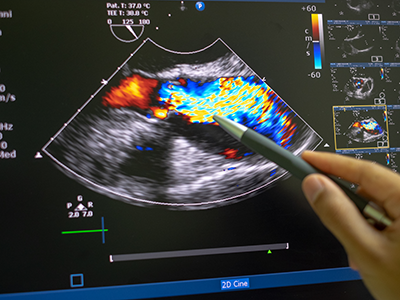
3D model for heart valve replacement planning.
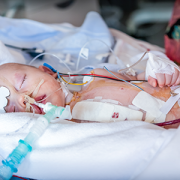
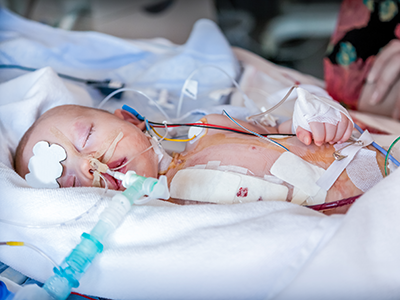
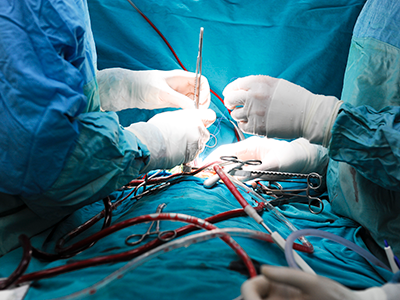
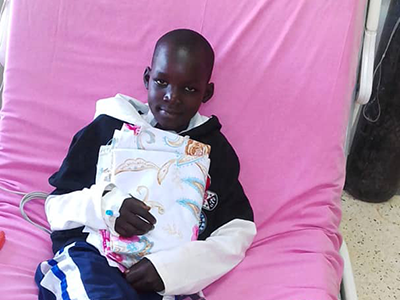
An 11-year-old boy is the first in the world to have an artificial heart valve replaced with a live tissue valve from a donated heart through a partial heart transplant. The procedure took place at Children’s National Hospital in Washington, D.C. The successful surgery, performed by the cardiac surgery team, is also the region’s first partial heart transplant.
“I am honored this family trusted our hospital and our team’s expertise to perform this life-changing first-of-its-kind procedure for Preston,” says Cardiac Surgery Chief Yves d’Udekem, MD, PhD. “I look forward to hearing about all the new activities and adventures he and his family can do once he is completely recovered from surgery.”
“Everyone is ecstatic with his progress so far,” says Lauren Porter, who is the patient’s mother. “We hope having this surgery will give him a lot more freedom to do the things he loves in his life, and we hope that by sharing our story we are helping to make procedures like this more available to kids who need them in the future.”
Artificial heart valves are the standard of care for a failing valve in a child born with congenital heart disease. But Dr. d’Udekem says they are exceptionally difficult in children. First, a traditional artificial tissue valve lasts only about a decade, so children like Preston who have their first valve inserted before age 2 will inevitably face at least two to three additional open-heart surgeries before age 40. Additionally, just like adults, an artificial mechanical heart valve requires the patient to take blood thinners and major precautions against injury for their entire lives. Research has also shown that the placement of an artificial heart valve causes the heart to change shape over time, impacting heart function later in life and leading to a shortened life span.
Replacing this valve with a living transplanted valve will give Preston freedom from a lifetime of blood-thinning medication. Research also shows these live tissue implants should grow along with him, greatly decreasing the likelihood of future open-heart surgeries.
“Everyone is ecstatic with his progress so far,” says Lauren Porter, who is the patient’s mother. “We hope having this surgery will give him a lot more freedom to do the things he loves in his life, and we hope that by sharing our story we are helping to make procedures like this more available to kids who need them in the future.”
Children’s National is the first hospital to remove a child’s previously implanted artificial valve and replace it with a live working valve from a donated heart through a relatively new procedure called a partial heart transplant. The Children’s National partial heart transplant replaced the heart’s mitral valve, which is the valve between the left upper chamber (left atrium) and the left lower chamber (left ventricle) of the heart. In general, partial heart transplants are rare. Prior to this surgery, four U.S. hospitals have used partial heart transplants to replace a failing, living heart valve with a valve from a donor heart, but no organization to date has ever replaced a prosthetic valve with a real one.
“Making this procedure an option for certain children who need a heart valve replacement is critical to having patients live their best lives and to providing hope to their family as they grow into adolescence and adulthood,” says Wayne J. Franklin, MD, senior vice president of the Children’s National Heart Center and a congenital cardiology specialist. “I am proud of our team that conducts such important research to innovate better clinical solutions for all of our patients with congenital heart disease.”
Live tissue partial heart transplants also offer an additional benefit. Donated hearts that do not qualify for use in a total heart transplant may have healthy components, like valves, that can be used for patients who don’t require total replacement. Candidates and potential donors are listed in a registry and matched according to biological factors including blood type, similar to the process for determining full heart transplant candidates.