Faster diagnosis and treatment of Kawasaki disease are key to preventing heart-related complications

“Kawasaki disease is highly treatable, yet too many children around the world face delayed diagnosis or limited access to care,” says Dr. Ashraf Harahsheh, an international Kawasaki disease expert.
Delayed diagnosis and barriers to treatment can increase the risk that a child with Kawasaki disease may develop a serious cardiac condition, according to a science advisory published in the Journal of the American Heart Association.
The advisory calls for global collaboration in diagnosis, care and research to reduce persistent care gaps and ensure children have timely access to diagnosis and treatment for Kawasaki disease no matter where they live in the world.
The advisory’s writing group was chaired by Ashraf S. Harahsheh, MD, director of the Kawasaki Disease Program and the Quality Outcomes in Cardiology Program, at Children’s National Hospital. In an AHA press release, Dr. Harahsheh said, “This science advisory underscores the power of international collaboration to advance research and improve care for patients everywhere. By sharing data, expertise and best practices, we can reduce disparities and improve heart health outcomes for children with Kawasaki disease, wherever they live.”
Why it matters
The media alert points out that more than 4,200 children are diagnosed with Kawasaki disease in the U.S. each year. Kawasaki disease is a rare but serious illness that can cause inflamed blood vessels throughout the body. If untreated, children with Kawasaki disease develop coronary artery aneurysms around 25% of the time. However, prompt diagnosis and treatment with intravenous immunoglobulin (IVIG) can lower the risk of coronary artery aneurysm to less than 5%.
Unfortunately, the authors note that Kawasaki disease management has improved mostly in large, experienced and economically advanced countries, while less advanced areas continue to face challenges in identifying Kawasaki and treating it early enough with IVIG to prevent potentially deadly coronary artery aneurysms.
What’s next
The AHA news release includes several key takeaways including:
- Strengthening diagnostic and treatment capacity in regions where access to care is most limited is essential.
- Most current Kawasaki disease collaborative networks lack formal funding, but strong research collaborations, shared expertise and coordinated treatment plans have been responsible for diagnosis and treatment advances.
- International collaborations in Kawasaki disease must consider the cultural needs of local communities, prioritize uncovering and reducing barriers to care, monitor results to improve outcomes, and promote evidence-based care across regions, especially in lower- and middle-income countries.
- Patients, families and advocacy groups must be included in any collaborative effort in Kawasaki disease to support patient-centered care and improve access to care.
The science advisory was prepared by a volunteer writing group on behalf of the American Heart Association’s Rheumatic Fever, Endocarditis, Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young (the Young Hearts Council).
“Kawasaki disease is highly treatable, yet too many children around the world face delayed diagnosis or limited access to care,” Dr. Harahsheh says.
Read the news release from the American Heart Association and the full science advisory, Importance of Fostering International Collaboration for Optimal Outcomes of Kawasaki Disease Worldwide: A Science Advisory From the American Heart Association


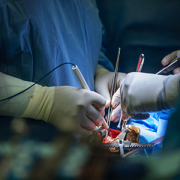
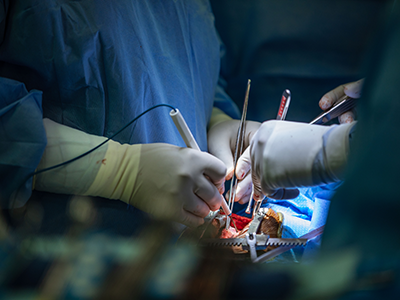





 The division of pediatric cardiac surgery at
The division of pediatric cardiac surgery at 





 On February 6, 2026,
On February 6, 2026, 
 Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.
Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.





 In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.
In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.









 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.