NIH awards $1.8 million to trial pacemaker delivery system for children
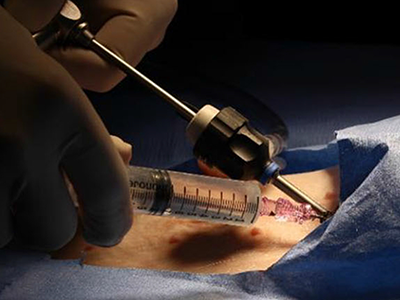
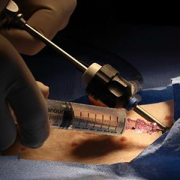
The PeriPath access port makes it possible for pacing and defibrillating leads to be placed in the smallest children through holes the size of a straw.
A $1.8 million Small Business Innovation Research (SBIR) grant from the National Institutes of Health (NIH) is funding the first clinical trial of a novel device called PeriPath. The device makes it possible for pacing and defibrillating leads (or wires) to be placed in the smallest children through holes the size of a straw, eliminating thoracotomy or sternotomy procedures for children who are too small for transvenous implantation.
Even the tiniest pacemakers and defibrillators on the market today aren’t small enough for infants and young children with heart rhythm abnormalities. Innovating smaller devices, including adapting current technology like the Medtronic Micra for pediatric use, is a good start but won’t be enough to eliminate some of the challenges for these patients. When a newborn or young child needs any pacemaker or defibrillator, they face open chest surgery. Their arteries and veins are just too small for even the smallest size transvenous pacemaker catheter.
The research goal
Charles Berul, M.D., division chief of Cardiology and co-director of the Children’s National Heart Institute, partnered with engineers in the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Hospital to develop and test a first-of-its-kind minimally invasive pericardial access tool. The tool allows doctors to place pacing and defibrillation leads to the epicardial surface of the heart under direct visualization from an endoscope.
The team hypothesizes that this tool will allow for pacing and defibrillation therapy to be delivered through a single small port inserted through the skin that is about the size of a drinking straw.
Why it matters: Less pain, shorter and fewer surgeries
If successful, the device will eliminate the need for open chest surgery in patients who aren’t candidates for transvenous placement. The ability to place these leads percutaneously should:
- Reduce pain and infection risk.
- Decrease procedure times.
- Minimize surgery complications that arise from open surgery.
- Improve better visualization for pericardial punctures.
- Allow other novel therapies such as epicardial ablation or, in the future, even drug/gene delivery into the pericardial space.
Any implanted pacemaker or defibrillator must be replaced every 5-10 years. A young child in critical need of such devices could face surgeries 10 or more times to replace the device and/or leads.
Pre-clinical testing shows early data that this percutaneous approach is as safe and effective as an open surgical technique, although it remains in early-stage evaluation.
What’s next
The NIH SBIR funding will allow the research team to assess long-term safety and efficacy and commercialize the PeriPath tool. Next steps are to:
- Refine the design of PeriPath for production manufacturing, integrate testing protocols into a Quality Management System and conduct a pilot verification build. Success is defined as manufacturing production devices that pass 510(k) verification and validation testing.
- Demonstrate substantial equivalence to predicate trocars through performance and handling validation testing using PeriPath to implant an epicardial lead in a pediatric simulator. If successful, the team will demonstrate equivalence and obtain investigational device exception (IDE).
- In the latter part of the plan, to perform a first in human feasibility clinical study using PeriPath to implant a commercial pacemaker lead with institutional review board (IRB) approval in infants at Children’s National.
Bottom line
Dr. Berul says, “This research could have a transformative impact on current clinical practice by converting an open surgical approach to a minimally invasive percutaneous procedure.”
He also notes that while the study design focuses on the unique needs of infants and children with congenital heart disease – who are the primary focus of the device – the results of the trial may benefit thousands of adult patients who need pacemakers or defibrillators but who are not candidates for the transvenous placement.