Neonatology
U.S. News & World Report voting
 Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.
Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.
How to determine your voting eligibility
Voting for the U.S. News & World Report Best Children’s Hospitals rankings can be done only through Doximity.
To participate, physicians must:
- Be board-certified and meet the eligibility criteria for the voting categories.
- For child and adolescent psychologists, your account must be up to date with your specialty and subspecialty correctly marked.
- Be a credential-verified member of Doximity (you must have an active and claimed Doximity profile).
- Have all certifications and board documents currently up-to-date in your Doximity profile.
View the full eligibility criteria
How to claim your Doximity profile to vote
- You have to claim your profile on Doximity.com to participate in the online survey. If you have not yet claimed your Doximity profile, go to Doximity.com, and click “Find My Profile.”
- Once your profile has been claimed, you must confirm your email address and board certifications.
- Verified Doximity members will receive an email inviting them to participate in the U.S. News survey.
- For more information on how to claim your profile, visit Doximity.com
How to update and verify existing Doximity account information
Your Doximity profile must have up-to-date licenses, certifications and board documents.
- Once you are logged in, your profile will automatically be in “Edit Mode.” You are able to add new items or edit existing information.
- Update your Doximity profile and ensure your information is current.
Once registered, users wishing to participate in the online survey should:
- Watch for an email from Doximity about the annual member survey.
- Even if you don’t see the email, if you are a registered Doximity user, you can still vote by logging in to Doximity.com with your username and password during the voting period.
- Once logged in, look for a U.S. News graphic or button on the homepage and click on it.
- The survey asks users to name the hospitals that provide the best care in your respective specialty, without consideration to location or cost. Pediatric specialists will list 10 hospitals. The order in which you list the hospitals does not matter.
Please note: Children’s National Hospital is listed as “Children’s National Hospital Washington, DC” on the survey.
Visit Doximity’s FAQs if you have issues or questions about registration or claiming your profile.
How to cast your vote
In February 2026 when voting opens, all survey-eligible physicians will receive a notification on the Doximity app for Android or iOS. If you do not use the Doximity app, you will receive an email when voting opens.
- Log in to your Doximity account at doximity.com or via the mobile app.
- Click the Notifications icon or tap the “Submit your Nominations” button on the homepage. You can also search for “U.S. News Best Hospitals”
- Select 10 hospitals in your respective specialty that you believe provide the best care in the United States.
- Submit your vote
Having technical issues?
If you have difficulty registering with Doximity or completing the survey, please visit Doximity support for assistance.
Vote
The 2026 U.S. News & World Report Best Children’s Hospitals reputation voting will open in mid-February. Look for your Doximity notification to vote.
The best of 2025 from Innovation District

In 2025, Innovation District readers gravitated toward stories that explored how research and clinical innovation are reshaping pediatric care in real time. This year’s most popular articles highlighted advances in complex surgical care, evidence-based treatments for chronic and neuropsychiatric conditions and emerging technologies — from wearable data to artificial intelligence — that are changing how clinicians diagnose, treat and support children and families. Read on for our list of the most popular articles we published on Innovation District in 2025.
1. Life-changing care: How Children’s National tackles pediatric cervical spine injuries
The Cervical Spine program at Children’s National Hospital is responsible for treating a range of conditions, including trauma, congenital abnormalities and tumors. These conditions can lead to instability or misalignment of the cervical spine. “There are unique challenges in pediatric cases due to anatomical differences. The cervical spines of children are more at risk for injury because of their developmental stage and structural characteristics,” says Matthew Oetgen, MD, MBA, chief of Orthopaedic Surgery and Sports Medicine at Children’s National.
(2 min. read)
2. Pioneering evidence-based treatments for substance addictions
Increasing evidence-based treatment is a key component of the Addictions Program at Children’s National Hospital, created in 2022 and led by Sivabalaji Kaliamurthy, MD. “We really want to focus on intervening in an evidence-based manner in the primary care setting because that is where most of our patients are going to first access care outside of the emergency room,” explains Dr. Kaliamurthy.
(3 min. read)
3. Breaking barriers in growth disorder treatment for families
For many children with short stature and other rare genetic growth disorders, there have been no next steps after usual treatment options prove ineffective. Researchers at Children’s National Hospital are digging deeper to find the root genetic causes of short stature disorders and creating novel, nuanced treatment options that have the opportunity to change how the field approaches these cases.
(4 min. read)
4. The link between metabolic acidosis and cardiovascular disease in children with CKD
Denver D. Brown, MD, nephrologist at Children’s National, is looking at whether untreated metabolic acidosis could potentially contribute to cardiovascular outcomes in children with chronic kidney disease (CKD). Here, she explains her motivation, findings and future directions for this critical research.
(3 min. read)
5. Therapy approach shows promise for PANS/PANDAS
A multidisciplinary therapy model developed at Children’s National shows promise for children with PANS and PANDAS, significantly reducing symptoms through structured cognitive-behavioral therapy and family-centered care. The approach could offer a new standard for treating these rare, complex neuropsychiatric disorders.
(2 min. read)
6. Wearable tech data shows promise in ADHD detection
A study from Children’s National reveals that common wearable devices like Fitbits may hold the key to improving how we identify Attention-Deficit/Hyperactivity Disorder (ADHD) in adolescents. By analyzing patterns in heart rate, activity levels and energy expenditure, researchers were able to predict ADHD diagnoses with striking accuracy, offering a glimpse into a future where objective, real-time data supports earlier and more personalized mental healthcare.
(2 min. read)
7. Novel pediatric pacemaker shows safety, effectiveness for fragile infants in multi-center study
A novel implantable pacemaker designed specifically for infants has demonstrated safety and effectiveness in stabilizing heart rhythms for at least two years. The multi-center study of 29 infants showed stable pacing, normal electrical parameters and expected battery life, offering a viable alternative to standard-size devices for the smallest children.
(2 min. read)
8. Socioeconomic disadvantage associated with higher long-term mortality for children after heart surgery
Children who had heart surgery and come from less advantaged neighborhoods in the Washington, D.C., region are much more likely to die in the long term than those from neighborhoods with more wealth and opportunity. The finding was part of a presentation titled, Socioeconomic Disadvantage Is Associated with Higher Long-Term Mortality After Cardiac Surgery, by Jennifer Klein, MD, MPH, cardiologist at Children’s National Hospital, during the Society of Thoracic Surgeons Annual Meeting in Los Angeles.
(2 min. read)
9. Children’s National brings AI into the RHD early diagnosis equation
Experts from Children’s National traveled to Uganda to continue work on a pilot program applying artificial intelligence (AI) to the diagnosis of rheumatic heart disease (RHD). The team created a tool that uses AI to predict RHD by identifying leaky heart valves on handheld ultrasound devices, then prompts a referral for a full echocardiogram.
(2 min. read)
10. Fighting food insecurity with fresh produce and education
Food insecurity is rising in Washington, D.C. and it’s hitting families with children the hardest. That’s why Children’s National Hospital created the Family Lifestyle Program (FLiP) – a multi-layered intervention, which offers Patient Navigation (FLiP-PN) and a Produce Prescription Intervention (FLiPRx). FLiP is a Food Is Medicine, clinical-community initiative that helps families get access to fresh food, build healthy habits and lower their risk of diet-related diseases like diabetes and obesity.
(3 min. read)
Children’s National in the News: 2025
 In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.
In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.
1. Gene therapy treatment gives new hope to sickle cell patients
Children’s National patient Wedam, 19, begins the first steps for intensive gene therapy for sickle cell disease, discussing his skepticism while his mother expresses her joy and gratitude for the treatment. (NBC News)
2. Little pacemakers ‘reliable’ in stabilizing newborns needing pacing
Charles Berul, MD, pediatric electrophysiologist and emeritus chief of Cardiology, discusses his study highlighting the safety and efficacy of an innovative smaller pacemaker designed for newborns with critical congenital heart disease. (Healio)
3. Why this clinical trial is offering some young cancer patients hope
Catherine Bollard, MBChB, MD, senior vice president and chief research officer, and the NexTGen team are poised to recruit patients for a new clinical trial that will take on an old, implacable foe: children’s solid tumors. (The Washington Post)
4. 11-year-old receives living donor heart valve to replace artificial one in breakthrough surgery
Yves d’Udekem, MD, PhD, chief of Cardiac Surgery, talked to Good Morning America about how an 11-year-old’s groundbreaking partial heart transplant will change his life and the lives of other children in need of valve replacements. (Good Morning America)
5. 9-year-old fights brain cancer with magic in her heart
The Lilabean Foundation along with Brian Rood, MD, medical director of the Brain Tumor Institute, talked about how patients like Kasey Zachman are the motivation behind finding a cure for brain cancer. (ABC News)
6. How Little League helps father-son duo recover from kidney transplant
USA Today Sports spoke with Gavin Brown and his parents, as well as Yi Shi, MD, a pediatric nephrologist at Children’s National Hospital, about their kidney transplant journey. (USA Today)
7. Jayden Daniels surprises Children’s National patient
After Jayden Daniels visited Commanders fan Sarah Addison at Children’s National Hospital while she was being treated for myeloid leukemia, they quickly became friends. (ESPN)
8. At D.C. children’s hospital, opera singers offer light, hope and healing
International opera stars performed for children, their families and employees at Children’s National Hospital. (The Washington Post)
9. ‘Gift of life:’ Tiny heart transplant saves life of Maryland baby
A baby boy in Maryland is back home after being given a second chance at life, just before his first birthday. The boy’s mother and his surgeon, Manan Desai, MD, share the remarkable story of a moment that changed all of their lives. (NBC4)
10. AI psychosis: Kids left delusional and paranoid over conversations with chatbots
Ashley Maxie-Moreman, PhD, clinical psychologist, spoke to ABC7 about what AI psychosis is and what parents need to know. (ABC7)
A roadmap to uncover the causes of congenital anomalies

A new white paper in Developmental Biology outlines a roadmap to uncover the causes of congenital anomalies through better genomic analysis, functional research and patient partnerships.
Congenital anomalies affect more than 6% of live births worldwide. In the United States, about one in every 33 babies is born with one of these conditions. They are a leading cause of infant mortality, childhood hospitalizations and lifelong disability. Yet despite their impact, congenital anomalies remain underrecognized and underfunded, and for many families, unexplained.
That gap between burden and understanding is the focus of a new white paper published in Developmental Biology as part of a special issue on congenital anomalies. The paper brings together five lead authors, more than 60 contributing scientists and clinicians, and endorsements from major scientific societies. Irene Zohn, PhD, principal investigator in the Center for Precision Medicine and Genomics Research at Children’s National Hospital, is among the authors helping shape this field-wide call to action.
The message is simple. We have the tools to identify the causes of many more congenital anomalies, but progress depends on investing, collaborating more effectively and treating patients and families as research partners.
Why so many families still lack answers
Advances in genome sequencing have transformed the search for genetic causes of disease. For some families, sequencing has provided long-awaited answers. For many others, the search continues.
More than half of individuals with congenital anomalies still do not receive a definitive genetic explanation. Even when sequencing is performed, results often include variants of uncertain significance, genetic changes that cannot yet be clearly linked to disease. Families may spend years navigating tests and referrals without answers, often described as the “diagnostic odyssey”. The white paper argues that this reflects system-level gaps rather than a lack of scientific potential.
Two priorities to move the field forward
The authors outline a framework centered on two key investments. First, they call for expanded and improved genomic analysis. This includes broader access to genome sequencing, better analytic tools and stronger data sharing across institutions. It also requires consistent terminology to accurately measure the true scope of congenital anomalies and recognize the magnitude of the problem.
Second, the paper emphasizes the need for mechanistic research. Identifying a genetic variant is only the first step. Researchers must also understand how that variant disrupts development and whether the consequences can be prevented or treated. This requires sustained support for functional studies using cell models, patient-derived tissues and animal systems. Together, these approaches can shorten the diagnostic journey, improve confidence in genetic findings, and accelerate the development of new therapies.
Putting patients and families at the center
A defining feature of the roadmap is its focus on patient partnership. Families should be treated as active participants in research, including through the return of results and clear communication about uncertainties. Genomic data should also be reanalyzed over time, since findings that are unclear today may become meaningful as science advances. Building systems that support this process is essential to improving care.
A pivotal moment for the field
Congenital anomalies are not rare when considered together. They represent a significant public health challenge with lifelong consequences for children and families. Science is advancing rapidly, but progress depends on aligning investment, infrastructure and collaboration with the scale of the problem. This white paper argues that the field is at a turning point. With focused support for genomic diagnosis, functional research and patient-centered partnerships, researchers and clinicians can deliver answers and improve outcomes for children who have waited too long.
You can read the full article, “Challenges and opportunities for understanding the genetic causes of congenital anomalies,” in Developmental Biology here.
Children’s National Hospital once again ranked among the nation’s best by U.S. News & World Report
 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
For the fifteenth straight year, Children’s National ranked in 10 specialty services and is the highest U.S. News ranked children’s hospital in Washington, D.C., Maryland and Virginia. Last year, U.S. News introduced pediatric & adolescent behavioral health as a service line in its rankings. While there are no ordinal rankings for behavioral health, the Children’s National program was named one of the top 50 programs in the country for the second year in a row.
“To be named among the nation’s top children’s hospitals for nine years in a row is a reflection of the extraordinary expertise, innovation and heart that our teams bring to every child and family we serve,” said Michelle Riley-Brown, MHA, FACHE, president and chief executive officer of Children’s National. “Our leadership in specialties like neurology, cancer, and diabetes and endocrinology underscores the national impact of our work, and we remain focused on setting new standards in pediatric care.”
The annual rankings are the most comprehensive source of quality-related information on U.S. pediatric hospitals and recognizes the nation’s top 50 pediatric hospitals based on a scoring system developed by U.S. News.
“Being a top-ranked pediatric hospital means more than just excelling in a single specialty — it means being a pillar of outstanding care for your entire region,” said Ben Harder, chief of health analysis and managing editor at U.S. News. “Our rankings acknowledge these hospitals for their comprehensive excellence, helping families find the very best care conveniently located within their state and community.”
The bulk of the score for each specialty service is based on quality and outcomes data. The process includes a survey of relevant specialists across the country, who are asked to list hospitals they believe provide the best care for patients with the most complex conditions.
The Children’s National specialty services that U.S. News ranked in the top 10 nationally are:
- Neurology and Neurosurgery (No. 4), led by Division Chiefs William D. Gaillard, MD, and Robert F. Keating, MD
- Diabetes and Endocrinology (No. 6), led by Division Chief Andrew Dauber, MD, MMSc
- Cancer (No. 8), led by Division Chief Eugene Hwang, MD
- Nephrology (No. 10), led by Division Chief Marva Moxey-Mims, MD
- Orthopedics (No. 10), led by Division Chief Matthew Oetgen, MD, MBA
The other six specialties ranked among the top 50 are Behavioral Health, Cardiology and Heart Surgery, Gastroenterology and GI Surgery, Neonatology, Pulmonology and Lung Surgery, and Urology.
Transarterial embolization offers hope for infants with hemimegalencephaly
A new study from Children’s National Hospital reveals how a minimally invasive procedure is transforming care for infants with hemimegalencephaly and medically refractory seizures, offering seizure relief with fewer surgical risks.
Until recently, families of infants with hemimegalencephaly and medically refractory seizures had few treatment options, almost all involved major brain surgery such as hemispherectomy, to remove or disconnect the affected portion of the brain. These procedures, while sometimes effective, carry high risks in very young infants due to the fragility of their developing brains and invasive nature of the surgery.
Now, specialists from the Hemimegalencephaly Program at Children’s National Hospital have pioneered a minimally invasive alternative. Leveraging the combined expertise of a multidisciplinary team — spanning neurology, neurointerventional radiology, prenatal pediatrics, neonatology, neurogenetics and neurodevelopmental pediatrics, neuropsychology and neurosurgery — they have developed a technique that induces controlled, targeted strokes in the affected hemisphere using transarterial embolization (TAE). This approach mimics the effect of surgery by reducing seizure activity, without the need for open brain procedures.
This novel strategy is the focus of a new study led by Monica Pearl, MD, director of the Neurointerventional Radiology Program. The research represents the largest retrospective review of its kind, examining 41 TAE procedures performed over an 11-year span in 13 infants under three months of age.
The results are promising: 73% of patients achieved Engel Class I seizure outcomes at follow-up, suggesting that with careful technique and patient selection, TAE may provide meaningful seizure control with fewer perioperative risks than traditional surgery. The study also highlights the technical complexity of the procedure, early complications and the critical role of evolving protocols in ensuring patient safety.
Authors from Children’s National include Monica Pearl, MD, Tayyba Anwar, MD, Shani Israel, BS, Lindsay Ruffini, CPNP, CPNP-AC, CPNP-PC, Panagiotis Kratimenos, MD, PhD, Kyle Spagnolo, DO, Wei-Liang Chen, MD, Madison Berl, PhD, William D. Gaillard, MD, Tammy N. Tsuchida, MD, PhD and Chima Oluigbo, MD
To learn more about the clinical outcomes and evolving protocols behind this approach, you can read the full study, Transarterial embolization for infants under 3 months of age with refractory seizures due to hemimegalencephaly: complication analysis and evolution of treatment strategy, in the Journal of Neurointerventional Surgery.
REI Week 2025 empowers the future in pediatric research and innovation
Children’s National Hospital hosted its fifteenth annual Research, Education and Innovation Week from March 31–April 4, 2025, bringing together clinicians, scientists, educators and innovators from across the institution to celebrate discovery and collaboration. This year’s theme, “Empowering the Future in Pediatric Research and Innovation with Equity, Technology and a Global Reach,” served as a call to action for advancing science that improves child health both locally and around the world.
Each day of the week-long event featured thought-provoking lectures — now available to watch — dynamic panel discussions, interactive workshops and vibrant poster sessions, all highlighting the diverse and interdisciplinary work taking place across Children’s National.
Centering the patient and the planet
REI Week began on Monday with a powerful keynote lecture from Lynn R. Goldman, MD, MS, MPH, Michael and Lori Milken dean of the Milken Institute School of Public Health at the George Washington University. In her talk, “Children: Uniquely vulnerable to climate-related threats,” Dr. Goldman underscored the urgent need to protect children from the environmental hazards of a changing climate and to integrate climate science into pediatric care and advocacy.
At mid-morning, Mary-Anne “Annie” Hartley, MD, PhD, MPH, director of the LiGHT Laboratory at École Polytechnique Fédérale de Lausanne, introduced the “MOOVE” platform — Massive Open Online Validation and Evaluation of clinical LLMs. Her talk demonstrated how artificial intelligence, when rigorously validated, has the potential to transform clinical decision-making and global health equity.
Monday’s final keynote, “Zinc and childhood diarrhea,” was presented by Christopher Duggan, MD, MPH, director of the Division of Nutrition at Harvard Medical School. Dr. Duggan highlighted the global health impact of zinc supplementation in reducing childhood mortality — a reminder that simple, evidence-based interventions can save millions of lives.
In that first day, the first poster session of the week showcased projects in adolescent medicine, global health, infectious diseases, oncology and more. The session reflected the full breadth of research taking place across Children’s National.
Ambroise Wonkam, MD, PhD, professor of genetic medicine at Johns Hopkins University, then delivered Tuesday’s Global Health Keynote Lecture, “Harnessing our common African genomes to improve health and equity globally.” His work affirmed that inclusive genomics is key to building a healthier world.
Later, the Global Health Initiative event and GCAF Faculty Seminar encouraged attendees to pursue collaborative opportunities at home and abroad, reflecting the growing global footprint of Children’s National research programs.
Transforming education and care delivery
On Wednesday, Larrie Greenberg, MD, professor emeritus of pediatrics, kicked off the day with a Grand Rounds keynote on educational transformation: “Shouldn’t teachers be more collaborative with their learners?” He followed with a CAPE workshop exploring the effectiveness of case-based learning.
The Nursing Sponsored Keynote Lecture by Vincent Guilamo-Ramos, PhD, MPH, LCSW, ANP-BC, PMHNP-BC, FAAN, explored “Redesigning the U.S. broken health system.” He offered an urgent and inspiring call to reimagine pediatric care by addressing social determinants of health.
In the Jill Joseph Grand Rounds Lecture, Deena J. Chisolm, PhD, director of the Center for Child Health Equity at Nationwide Children’s Hospital, challenged attendees to move beyond dialogue into action in her talk, “Health equity: A scream to a whisper?,” reminding researchers and clinicians that advocacy and equity must be foundational to care.
The day continued with a poster session spotlighting medical education, neonatology, urology and neuroscience, among other fields.
Posters and pathways to progress
Throughout the week, poster sessions highlighted cutting-edge work across dozens of pediatric disciplines. These sessions gave attendees the opportunity to engage directly with investigators and reflect on the shared mission of discovery across multiple disciplines, including:
- Neuroscience and Neuroprotection – Novel approaches to brain injury treatment and neurodevelopmental disorders.
- Genetics and Precision Medicine – Advances in understanding genetic predisposition and innovative therapies.
- Health Disparities and Global Health – Strategies to bridge gaps in pediatric healthcare access worldwide.
- Innovative Technology in Pediatrics – Breakthroughs in AI, bioinformatics and medical device development.
Honoring excellence across Children’s National
The REI Week 2025 Awards Ceremony celebrated outstanding contributions in research, mentorship, education and innovation. The winners in each category were:
POSTER SESSION AWARDS
Basic & Translational Research
Faculty: Benjamin Liu, PhD
“Genetic Conservation and Diversity of SARS-CoV-2 Envelope Gene Across Variants of Concern”
Faculty: Steve Hui, PhD
“Brain Metabolites in Neonates of Mothers with COVID-19 Infection During Pregnancy”
Faculty: Raj Shekhar, PhD
“StrepApp: Deep Learning-Based Identification of Group A Streptococcal (GAS) Pharyngitis”
Post docs/Fellows/Residents: Dae-young Kim, PhD
“mhGPT: A Lightweight Domain-Specific Language Model for Mental Health Analysis”
Post docs/Fellows/Residents: Leandros Boukas, MD, PhD
“De Novo Variant Identification From Duo Long-Read Sequencing: Improving Equitable Variant Interpretation for Diverse Family Structures”
Staff: Naseem Maghzian
“Adoptive T Lymphocyte Administration for Chronic Norovirus Treatment in Immunocompromised Hosts (ATLANTIC)”
Graduate Students: Abigail Haffey
“Synergistic Integration of TCR and CAR T Cell Platforms for Enhanced Adoptive Immunotherapy in Brain Tumors”
High School/Undergraduate Students: Medha Pappula
“An ADHD Diagnostic Interface Based on EEG Spectrograms and Deep Learning Techniques”
Clinical Research
Faculty: Folasade Ogunlesi, MD
“Poor Air Quality in Sub-Saharan Africa is Associated with Increase Health Care Utilization for Pain in Sickle Cell Disease Patients”
Faculty: Ayman Saleh, MD
“Growth Parameters and Treatment Approaches in Pediatric ADHD: Examining Differences Across Race”
Post docs/Fellows/Residents: Nicholas Dimenstein, MD, MPH
“Pre-Exposure Prophylaxis (PrEP) Eligibility in the Pediatric Emergency Department”
Staff: Tayla Smith, MPH
“The Public Health Impact of State-Level Abortion and Firearm Laws on Health Outcomes”
Graduate Students: Natalie Ewing
“Patterns of Bacteriuria and Antimicrobial Resistance in Patients Presenting for Primary Cloacal Repair: Is Assisted Bladder Emptying Associated with Bacteriuria?”
Graduate Students: Manuela Iglesias, MS
“Exploring the Relationship Between Child Opportunity Index and Bayley-III Scores in Young Children”
High School/Undergraduate Students: Nicholas Lohman
“Preliminary Findings: The Efficacy, Feasibility and Acceptability of Group Videoconference Cognitive Behavioral Therapy with Exposure and Response Prevention for Treating Obsessive-Compulsive Disorder Among Children and Young People”
Community-Based Research
Faculty: Sharon Shih, PhD
“Assessing Pediatric Behavioral Health Access in DC using Secret Shopper Methodology”
Post docs/Fellows/Residents: Georgios Sanidas, MD
“Arrested Neuronal Maturation and Development in the Cerebellum of Preterm Infants”
Staff: Sanam Parwani
“Intersectionality of Gender and Sexuality Diversity in Autistic and Non-Autistic Individuals”
Graduate Student: Margaret Dearey
“Assessing the Burden of Period Poverty for Youth and Adolescents in Washington, DC: A Pilot Study”
Quality and Performance Improvement
Faculty: Nichole L. McCollum, MD
“A Quality Improvement Study to Increase Nurse Initiated Care from Triage and Improve Timeliness to Care”
Post docs/Fellows/Residents: Hannah Rodriguez, MD
“Reducing Unnecessary Antibiotic Use in a Level IV NICU”
Staff: Amber K. Shojaie, OTD, OTR/L
“Implementing Dynamic Axilla Splints in a Large Burn Patient”
MENTORSHIP AWARDS
Basic Science Research
Conrad Russell Y. Cruz, MD, PhD
Clinical Research
Bench to Bedside Research
Ioannis Koutroulis, MD, PhD, MBA
ELDA ARCE TEACHING SCHOLAR AWARD
SUZANNE FEETHAM NURSING RESEARCH SUPPORT AWARD
Eileen P. Engh, PhD RN
“Rare Disease Organization Lifecycle” Role in Helping Parents with Everyday Life Information Seeking and Connection (RDO-HELIX)
EXPLORATIONS IN GLOBAL HEALTH PILOT AWARDS
Launchpad Awards
Mi Ran Shin, MD, MPH
“Establishing Interdisciplinary Rehabilitation for Birth and Burn Injuries in Ethiopia”
Susan Harvey, MSN, CPNP-AC
“Implementation of Sickle Cell Pilot Program in Ndhiwa Sub County, Kenya”
Meleah Boyle, PhD, MPH
“Understanding and Addressing Environmental Sustainability to Protect the Health of the Children’s National and Global Communities”
Eiman Abdulrahman, MD
“Research Capacity Building to Improve Pediatric Emergency and Critical Care in Ethiopia”
Pilot Awards
Alexander Andrews, MD
“EEG as a Diagnostic and Prognostic Marker in Severe Pediatric Malaria, Blantyre Malawi”
Daniel Donoho, MD & Timothy Singer, MD
“Feasibility Study of a Novel Artificial Intelligence-Based Educational Platform to Improve Neurosurgical Operative Skills in Tanzania”
Hasan Syed, MD
“Bridging the Gap an Educational Needs Assessment for Pediatric Neurosurgery Training in Pakistan”
Sofia Perazzo, MD & Lamia Soghier, MD, MEd, MBA
“QI Mentorship to Improve Pediatric Screening and Follow-up in Rural Argentina”
Benjamin Liu, PhD
“AI-Empowered Real-Time Sequencing Assay for Rapid Detection of Schistosomiasis in Senegal”
Rae Mittal, MD
“Assessment and Enhancement of Proficiency in Emergency Child Neurology Topics for Post-Graduate Emergency Medicine Trainees in India”
Innovation Day ignites bold thinking
Thursday, REI Week shifted to the Children’s National Research & Innovation Campus for Innovation Day, a celebration of how bold ideas and collaborative culture can accelerate progress in pediatric medicine.
Brandy Salmon, PhD, associate vice president of Innovation and Partnerships at Virginia Tech, opened the day with “The Alchemy of Innovation,” focusing on how institutions can build a culture that fuels transformative partnerships.
A multidisciplinary panel discussion moderated by Nathan Kuppermann, MD, MPH, and Catherine Bollard, MBChB, MD, featured Nehal Mehta, MD, Julia Finkel, MD, Kevin Cleary, PhD, Ioannis Koutroulis, MD, PhD, MBA, Francesca Joseph, MD and Patrick Hanley, PhD, who shared how innovation can be advanced and promoted, especially as a core institutional priority.
A shared vision for the future
REI Week 2025 reaffirmed the values that define Children’s National: a commitment to excellence, collaboration and equity in pediatric research and care. As discoveries continue to emerge from our hospital and our research campuses, the connections built and ideas sparked during this week will help shape the future of pediatric health — locally and globally.
By elevating voices from the bedside to the bench, with the support of the executive sponsors Nathan Kuppermann, MD, MBChB, Catherine Bollard, MBChB, MD, Kerstin Hildebrandt, MSHS, Linda Talley, MS, RN, NE-BC and David Wessel, MD, REI Week demonstrated that we must embrace the community in all aspects of our work. Because we know that there are answers we can only get from the patients that we serve—and we need to be their voice.
Research, Education & Innovation Week will be back next year on April 13-17, 2026.
Posters at the REI Week 2025 Monday, March 31 poster session.

Panelists discuss innovation during REI Week 2025.

Global Health Initiative community engagement event during REI Week 2025.

Chris Rees presents his REI Week 2025 lecture.

Nathan Kuppermann listens to a presenter during the REI Week 2025 Tuesday, April 1, poster session.

Michelle Riley-Brown, Nathan Kuppermann, Catherine Bollard and Naomi Luban on stage during the REI Week 2025 awards ceremony.

Brandy Salmon presents on innovation programs at Virginia Tech during the REI Week 2025 Innovation Day.

Catherine Bollard listens to a presenter during the REI Week 2025 Monday, March 21 poster session.
Ambroise Wonkman poses for a picture with Children’s National staff.

Tanzeem Choudhury presenting during REI Week 2025.

Predictive analytics elevates NICU patient care

Connecting the Ventilator to Etiometry through the data acquisition module. ECS shown in red circle.
Managing the delicate health of premature infants in the NICU requires more than just vigilant care — it demands real-time insights and precision. With high-tech tools like the Etiometry platform, clinicians at Children’s National can now track changes in oxygen levels and other vital data, making timely adjustments to optimize each patient’s care.
Why it matters
This predictive analytics system allows doctors and nurses to visualize trends over time and collaborate seamlessly, even from a distance.
“By analyzing data and developing new algorithms, we can quickly identify if a patient is deteriorating, allowing us to intervene before further decline occurs. We can also fine-tune care, weaning patients who are ready faster and delaying it for those who aren’t, ultimately ensuring more targeted and effective treatment,” Lamia Soghier, MD, MED, MBA, neonatologist and the NICU Quality and Safety Officer.
How it works
“The technology relies on software and a piece of equipment called EC10, which connects ventilators to monitors. However, there aren’t enough of these devices to have one in each NICU room, leading to risks of misplacement or damage when patients move between rooms,” said Sofia Perazzo, MD, program lead for Neonatology at Children’s National.
“The $35,000 grant Children’s National received from the Founders Auxiliary Board at Children’s National provides funding to equip each room with an EC10, ensuring it’s always available. Additionally, the grant will support the purchase of modules to connect ventilators to the cloud, improving data capture and ensuring that important patient information is not missed.”
Not only does the technology improve patient care in real-time, but it also opens new opportunities for research and data-driven improvements.
What excites you about this?
Dr. Soghier says the ability to quickly analyze data has transformed how clinicians track patient outcomes, cutting down research time from months to weeks. This has streamlined research and made it easier to automatically track quality improvement (QI) metrics, changing the clinical environment.
The Etiometry software platform also allows us to review past data and combine it with additional patient information, enabling us to conduct studies, including a recent retrospective analysis of 879 premature babies.
“By examining oxygen treatment compliance, we found that lower compliance rates were linked to a higher likelihood of developing conditions like retinopathy of prematurity (ROP) and bronchopulmonary dysplasia (BDP), said Dr. Perazzo. “We look forward to developing new algorithms by increasing time in compliance to improve patient outcomes.”
This important research will be presented at the Pediatric Academy Society meeting later this month.
New study finds prenatal opioid exposure linked to smaller newborn brain volumes
 A new study out of the Center for Prenatal, Neonatal & Maternal Health Research, directed by Catherine Limperopoulos, PhD, led by Yao Wu, PhD and Stephanie Merhar, MD, MS, out of Cincinnati Children’s Hospital Medical Center, sought answers to the question: “Do brain volumes differ in opioid-exposed vs. unexposed newborns?” In one of the largest studies of this kind, researchers found that prenatal exposure to opioids is associated with smaller brain volumes in newborns. These findings from the landmark Outcomes of Babies with Opioid Exposure (OBOE) study build on, reinforce smaller studies, and achieve a better understanding of the impacts of prenatal opioid exposure (POE).
A new study out of the Center for Prenatal, Neonatal & Maternal Health Research, directed by Catherine Limperopoulos, PhD, led by Yao Wu, PhD and Stephanie Merhar, MD, MS, out of Cincinnati Children’s Hospital Medical Center, sought answers to the question: “Do brain volumes differ in opioid-exposed vs. unexposed newborns?” In one of the largest studies of this kind, researchers found that prenatal exposure to opioids is associated with smaller brain volumes in newborns. These findings from the landmark Outcomes of Babies with Opioid Exposure (OBOE) study build on, reinforce smaller studies, and achieve a better understanding of the impacts of prenatal opioid exposure (POE).
Dive Deeper
The Advancing Clinical Trials in Neonatal Opioid Withdrawal (ACT-NOW)’s OBOE study is a multi-site observational study of newborns with prenatal opioid exposure and a control group of unexposed newborns from four different sites in the United States – Case Western Reserve University, Cincinnati Children’s Hospital Medical Center, University of Alabama at Birmingham, and Children’s Hospital of Philadelphia.
In a study involving 173 newborns who were exposed to opioids during pregnancy and 96 newborns not exposed to opioids prenatally showed smaller brain sizes in several key areas. Specifically, these exposed babies had smaller total brain volumes, as shown through MRI, as well as reduced volumes in important parts of the brain, including the cortex (outer layer of the brain), deep gray matter (areas that control movement and emotions), white matter (which helps transmit signals in the brain), cerebellum (responsible for coordination and movement), brainstem (controls basic functions like breathing), and the amygdala (involved in emotions and memory).
Further details showed that newborns exposed to medication for opioid use disorder (MOUD) during pregnancy with methadone, had smaller white matter volumes, while those exposed to MOUD with buprenorphine had smaller volumes specifically in the right amygdala. Additionally, newborns who were exposed to opioids plus additional substances such as THC and gabapentin had smaller volumes in even more brain areas compared to those who were only exposed to opioids.
What’s Next
The OBOE study sets the groundwork for further research into the long-term impact of opioid exposure during pregnancy. Additional work is necessary to expand on these findings and how they relate to functions in childhood – including exploring the way these reduced brain volumes may impact cognitive, behavioral, and motor impairments. The study raises important questions about how current guidelines for MOUD during pregnancy – specifically with methadone and buprenorphine – might evolve considering these findings. This study highlights the need for further research to assess the long-term effects of MOUD regimens on both maternal and infant outcomes.
This significant study underscores the importance of multi-disciplinary collaboration in opioid exposure research, effective regulation, and policy interventions – involving healthcare providers, researchers, policymakers, and affected families – to best mitigate the consequences and improve the health outcomes of children affected by prenatal opioid exposure.
You can read the full study, Antenatal Opioid Exposure and Global and Regional Brain Volumes in Newborns, and its companion editorial, Following the Developing Brain Affected by Opioid Exposure, in the Journal of the American Medical Association.
Additional authors from Children’s National include Kushal Kapse, BS, MS, and Josepheen De Asis-Cruz, MD, PhD. Other authors include Carla M. Bann, PhD, Jamie E. Newman, PhD4, Nicole Mack, MS, Sara B. De Mauro, MD, MSCE, Namasivayam Ambalavanan, MD, Jonathan M. Davis, MD, Scott A. Lorch, MD, MSCE5, Deanne Wilson-Costello, MD, Brenda B. Poindexter, MD and Myriam Peralta-Carcelen, MD.
Children’s National delivers on the promise in 2024

The Children’s National 2023-2024 Academic Annual Report show on a tablet.
Children’s National Hospital has released its 2023-2024 Academic Annual Report, showcasing a year of transformative progress in pediatric medicine. The report highlights achievements across its research centers, institutes and Innovation Ventures, underscoring the hospital’s role as a leader in advancing child health through innovation and collaboration.
“This year’s report reflects the remarkable progress we have made in advancing the frontiers of pediatric medicine,” said Nathan Kuppermann, MD, MPH, Chief Academic Officer and Chair of Pediatrics. “It highlights groundbreaking work across our research centers, institutes, and Innovation Ventures, showcasing the collaborative spirit that drives our mission forward. These achievements underscore our shared commitment to delivering transformative research and the best possible outcomes for children and families.”
Delivering across centers
The report captures the contributions of each of Children’s National’s research centers, each pushing the boundaries of pediatric healthcare:
- Center for Cancer & Immunology Research (CCIR): Delivering on the promise of cell and gene therapies, offering innovative treatments for pediatric cancers and immune disorders.
- Center for Genetic Medicine Research (CGMR): Advancing pediatric genetic medicine through interdisciplinary efforts, addressing complex genetic conditions with cutting-edge science.
- Center for Neuroscience Research (CNR): A year of growth in scientific excellence, advancing the understanding of brain development and neurological conditions.
- Center for Prenatal, Neonatal & Maternal Health Research (CPHNMR): Revolutionizing neonatal care with its pioneering infant brain health neuromonitoring program.
- Center for Translational Research (CTR): Facilitating groundbreaking work by new K awardees and driving translational research to bridge the gap between discovery and clinical care.
- Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI): Leading the way in advanced research projects in pediatric surgery, pushing technological boundaries to improve outcomes for children worldwide.
Taking the lead in innovation through collaboration
Innovation Ventures at Children’s National is advancing pediatric health security, addressing unique challenges with transformative solutions. Meanwhile, the Children’s National Research & Innovation Campus (CNRIC) continues to thrive as a hub for discovery and collaboration, hosting conferences on topics like artificial intelligence in healthcare, cell and gene therapy, and pediatric epilepsy research.
A vision for the future
The report also highlights Children’s National’s focus on integrating cutting-edge technologies like artificial intelligence into its research and clinical practices, as well as addressing global health challenges such as the effects of climate change on children’s health. These efforts reflect the hospital’s commitment to improving outcomes for children everywhere through innovation, teamwork, and forward-thinking leadership.
The 2023-2024 Academic Annual Report serves as a testament to the dedication and expertise of the Children’s National community, showcasing how collaboration and innovation are shaping the future of pediatric healthcare.
Children’s National in the News: 2024
 In 2024, Children’s National Hospital continued to make remarkable strides across diverse areas of pediatric medicine, from groundbreaking technological innovations to critical health advocacy. The following compilation showcases ten significant stories that demonstrate the breadth and depth of the hospital’s impact, as featured in major national news outlets including NBC Nightly News, CNN, The Washington Post, The New York Times, NPR, The Today Show, Healio, and POLITICO. Delve into our 2024 news highlights for more.
In 2024, Children’s National Hospital continued to make remarkable strides across diverse areas of pediatric medicine, from groundbreaking technological innovations to critical health advocacy. The following compilation showcases ten significant stories that demonstrate the breadth and depth of the hospital’s impact, as featured in major national news outlets including NBC Nightly News, CNN, The Washington Post, The New York Times, NPR, The Today Show, Healio, and POLITICO. Delve into our 2024 news highlights for more.
1. World’s smallest pacemaker gives new hope to babies with heart defects
Charles Berul, M.D., and a patient family talk about the pill-sized pacemaker that saved the life of Abby, an infant born with deadly heart defects. (NBC Nightly News)
2. ‘A $10 death trip’: Fentanyl is killing teens. Meet one fighting for his life
Sivabalaji Kaliamurthy, M.D., addiction psychiatrist and director of the Addictions Program, spoke to CNN about the impact of drug addiction on teen health and the lack of resources available to treat opioid use disorder. (CNN)
3. Health panel urges interventions for children and teens with high BMI
Susma Vaidya, M.D., M.P.H., associate medical director of the IDEAL Clinic, shared her concerns about childhood obesity treatment recommendations issued today by a leading panel of independent U.S. health experts. (The Washington Post)
4. An Rx for food? Doctor’s offices offer groceries to those in need
Shideh Majidi, M.D., M.S.C.S., and Emily Frymark, clinical dietitian, spoke about how the food pharmacy, created in partnership with the Capital Area Food Bank, benefits patients with diabetes and other chronic conditions. (The Washington Post)
5. First patient begins newly approved sickle cell gene therapy
Kendric Cromer, a 12-year-old boy being treated at Children’s National Hospital, became the first person in the world with sickle cell disease to begin a commercially approved gene therapy that may cure the condition. “This is a big effort,” says David Jacobsohn, M.D., ScM, M.B.A. (The New York Times)
6. ‘We created this problem’: A pediatric surgeon on how gun violence affects children
Mikael Petrosyan, M.D., associate chief of General and Thoracic Surgery, discusses the stress medical staff face when treating young victims of gun violence. (NPR)
7. 7th grade boy rings bell after final round of chemotherapy
Landon, an 11-year-old patient, rang the bell at Children’s National Hospital with family, friends, doctors and nurses cheering after finishing his final round of chemotherapy. (The Today Show)
8. Study: One in three adolescents experience ‘period poverty’
Monika Goyal, M.D., M.S.C.E., pediatric emergency medicine specialist and co-director of the Center for Translational Research, emphasized the need for awareness in addressing period poverty in teenagers and young adults. (Healio)
9. The AI assurance labs are coming
Kolaleh Eskandanian, Ph.D., M.B.A., P.M.P., vice president and chief innovation officer, participates in a panel discussion covering AI data collection, associated risks, reliance and other topics related to artificial intelligence. (POLITICO)
10. First day of a ‘new life’ for a boy with sickle cell
Children’s National patient Kendric Cromer, 12, became one of the first children ever to be treated with a newly approved gene therapy that will free him from the sickle cell disease that has stolen his childhood. (The New York Times)
The best of 2024 from Innovation District
 2024 marked another groundbreaking year for Children’s National Hospital, showcasing remarkable advances across the spectrum of pediatric medicine, research and healthcare innovation. From pioneering surgical procedures to breakthrough artificial intelligence applications, the institution continued to push the boundaries of what’s possible in children’s healthcare. Read on for our list of the most popular articles we published on Innovation District in 2024.
2024 marked another groundbreaking year for Children’s National Hospital, showcasing remarkable advances across the spectrum of pediatric medicine, research and healthcare innovation. From pioneering surgical procedures to breakthrough artificial intelligence applications, the institution continued to push the boundaries of what’s possible in children’s healthcare. Read on for our list of the most popular articles we published on Innovation District in 2024.
1. Prenatal COVID exposure associated with changes in newborn brain
A study led by researchers at Children’s National Hospital showed that babies born during the COVID-19 pandemic have differences in the size of certain structures in the brain, compared to infants born before the pandemic. The findings suggest that exposure to the coronavirus and being pregnant during the pandemic could play a role in shaping infant brain development.
(3 min. read)
2. Children’s National Hospital again ranked among the best in the nation by U.S. News & World Report
Children’s National Hospital was ranked as a top hospital in the nation by the U.S. News & World Report 2024-25 Best Children’s Hospitals annual rankings. This marks the eighth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
(2 min. read)
3. Children’s National performs first ever HIFU procedure on patient with cerebral palsy
In January 2023, a team of multidisciplinary doctors performed the first case in the world of using bilateral high intensity focused ultrasound (HIFU) pallidotomy on Jesus, a 22-year-old patient with dyskinetic cerebral palsy. The procedure is part of a clinical trial led by Chima Oluigbo, M.D., pediatric neurosurgeon at Children’s National Hospital.
(3 min. read)
4. Novel ultrasound device gets FDA breakthrough designation with Children’s National support
A novel ultrasound device developed by Bloom Standard received the Food and Drug Administration’s valued breakthrough device designation with the help of Children’s National Hospital. The device that enables autonomous, hands-free ultrasound scans to be performed anywhere, by any user.
(2 min. read)
5. First-of-its-kind pilot study on the impacts of Lyme disease in pregnancy and infant development
Understanding the effects of Lyme disease on the developing fetal brain is essential to ensure timely prenatal and postnatal treatments to protect the fetus and newborn. In response to this need, Children’s National Hospital is leading a pilot study to establish the groundwork needed for a larger study to determine the effect of in utero exposure to Lyme disease on pregnancy and early childhood neurodevelopmental outcomes.
(3 min. read)
6. Earliest hybrid HLHS heart surgery kids thrive 5 years later
Five years ago, Cayden was born 6 weeks early weighing less than four pounds and at risk of dying from her critical congenital heart disease. Today, she’s a happy five-year-old. Early diagnosis of her hypoplastic right ventricle, double inlet left ventricle and critical coarctation of the aorta allowed for the team at Children’s National Hospital to create a careful plan for safe delivery and to offer an innovative hybrid HLHS surgical approach at the hospital within 24 hours after she was born.
(1 min. read)
7. Wayne J. Franklin, M.D., F.A.C.C., named senior vice president of the Children’s National Heart Center
Children’s National Hospital appointed Wayne J. Franklin, M.D., F.A.C.C., as the new senior vice president (SVP) of the Children’s National Heart Center. In this role, Dr. Franklin oversees the full spectrum of heart care services including cardiac imaging and diagnostics, interventional cardiology, electrophysiology, cardiac anesthesia, cardiac surgery and cardiac intensive care.
(2 min. read)
8. Artificial – and accelerated – intelligence: endless applications to expand health equity
By pioneering artificial intelligence (AI) innovation programs at Children’s National Hospital, Marius George Linguraru, D.Phil., M.A., M.Sc., and the AI experts he leads are ensuring patients and families benefit from a coming wave of technological advances. The team is teaching AI to interpret complex data that could otherwise overwhelm clinicians.
(4 min. read)
9. Evidence review: Maternal mental conditions drive climbing death rate in U.S.
Painting a sobering picture, a research team led by Children’s National Hospital culled years of data demonstrating that maternal mental illness is an under-recognized contributor to the death of new mothers. They called for urgent action to address this public health crisis.
(3 min. read)
10. Nathan Kuppermann, M.D., M.P.H., named chief academic officer and chair of Pediatrics
Children’s National Hospital appointed Nathan Kuppermann, M.D., M.P.H., as its new executive vice president, chief academic officer and chair of Pediatrics. In this role, Dr. Kuppermann oversees research, education and innovation for the Children’s National Research Institute as well as academic and administrative leadership in the Department of Pediatrics at George Washington University School of Medicine & Health Services.
(2 min. read)
11. First global clinical trial achieves promising results for hypochondroplasia
Researchers from Children’s National Hospital presented findings from the first clinical trial of the medication vosoritide for children with hypochondroplasia – a rare genetic growth disorder. During the phase 2 trial, researchers found vosoritide increased the growth rate in children with hypochondroplasia, allowing them to grow on average an extra 1.8 cm per year.
(2 min. read)
12. Pioneering research center aims to revolutionize prenatal and neonatal health
Since its establishment in July 2023, the Center for Prenatal, Neonatal & Maternal Health Research at Children’s National Hospital has gained recognition through high-impact scientific publications, featuring noteworthy studies exploring the early phases of human development.
(3 min. read)
Neighborhood disadvantage alters brain networks in unborn babies
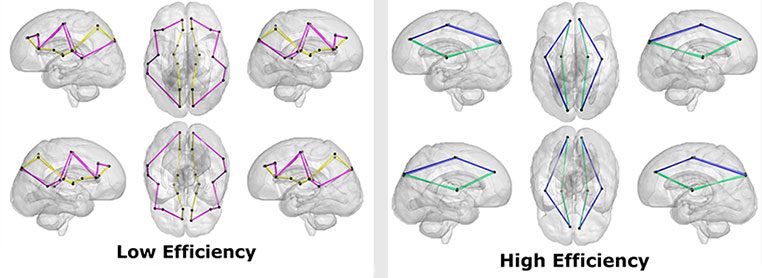 Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the latest edition of The Journal of Pediatrics, underscore the need to support young families from a baby’s earliest days.
Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the latest edition of The Journal of Pediatrics, underscore the need to support young families from a baby’s earliest days.
According to this new research from the Center for Prenatal, Neonatal & Maternal Health Research at Children’s National Hospital, exposure to neighborhood disadvantage lessens the functional integration of neural networks in the fetal brain, as seen on functional MRIs of healthy babies. The center compared the brains of 68 healthy babies on 79 scans. Researchers then mapped details about neural activity to a “social vulnerability index” from the Centers for Disease Control and Prevention, which indicates proximity to environmental stressors.
“We specifically looked at brain architecture to see how easily information flows between different regions,” said Kevin Cook, Ph.D., research faculty at the center and the manuscript’s first author. “To do this, we used graph theory, which borrows concepts from social network theory. It’s widely applied in computer science to understand how information flows within groups, and neuroscience has adapted it to study how information travels within the brain.”
What we found
Dr. Cook said researchers focused on the three metrics:
- Path length, which measures how many stops information needs to make along its way through the brain.
- Global efficiency, which measures the overall efficiency of the entire brain’s network.
- Small-world propensity, which describes how the brain’s network is organized and indicates how well the brain is organized into smaller, efficient networks.
As social vulnerability increased, the research team found global efficiency decreased, meaning the brain’s neural network was less efficient. The path lengths were also longer in children with greater neighborhood disadvantage, reinforcing the global efficiency findings.
The fine print
The under-development and over-development of fetal brains may contribute to neurological disorders, such as autism, epilepsy and other conditions of interest to researchers. Yet science’s understanding of how the brain matures in utero is still limited.
In this study, researchers found a notable difference related to age. At the youngest gestational ages, path lengths are longer, and both global efficiency and small world propensity are lower. As the fetus gets closer to term, path length and global efficiency show rapid maturation and less advantaged fetuses catch up to their peers who have greater advantages.
Researchers saw the same findings for small-world propensity, but the maturation didn’t stop. These unborn babies overshot their peers and had greater small-world propensity, suggesting their brains are divided into a greater number of smaller networks than their advantaged peers.
“We believe that length and global efficiency are on a trend to overshoot,” Dr. Cook said. “These findings are notable because they agree with what we know about older children and adolescents. Greater disadvantage is associated with hyper- or over-maturation of the brain. Our findings suggest that this may be starting even before birth.”
What’s ahead
While still early, this research improves the understanding of how environmental complexities can impact an unborn baby. Catherine Limperopoulos, Ph.D., director of the research center, which opened in 2023, said this work will be foundational as they continue to study the impact of a child’s environment on development.
“These findings have important implications for understanding how status and disadvantage may have a cumulative effect on fetal brain development,” Dr. Limperopoulos said. “We must study and consider how to conceptualize the impact of socioenvironmental disadvantage in communities to better care for children and work to improve outcomes.”
Read the full study – “Greater Neighborhood Disadvantage Is Associated with Alterations in Fetal Functional Brain Network Structure” – in The Journal of Pediatrics.
Children’s National again ranked among the best in the nation by U.S. News & World Report
 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2024-25 Best Children’s Hospitals annual rankings. This marks the eighth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2024-25 Best Children’s Hospitals annual rankings. This marks the eighth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
This year, U.S. News ended ordinal rankings on its Honor Roll. Instead of assigning a numerical rank from 1 to 10, all hospitals on the Honor Roll will be recognized as having attained the highest standards of care in the nation.
In addition, Children’s National tied for #1 pediatric hospital in the Mid-Atlantic region, which includes New York, New Jersey, Delaware, Pennsylvania, the District of Columbia, West Virginia and Virginia. It’s also best in the Mid-Atlantic in Neonatology.
For the fourteenth straight year, Children’s National ranked in 10 specialty services. New this year, U.S. News included behavioral health as a service line in the rankings. Since it’s the first year, there are no ordinal rankings for behavioral health, but the Children’s National program was named one of the top 50 programs in the country.
“In my first year here, I witnessed what makes Children’s National so special — our commitment to collaboration, empowering one another, and charting a bold path forward for pediatric care,” said Michelle Riley-Brown, MHA, FACHE, president and chief executive officer of Children’s National. “I’m proud U.S. News again recognized Children’s National as one of the top in the nation and the highest-ranked pediatric hospital in D.C., Maryland and Virginia. Together, we’ll continue to push the boundaries of care, research and innovation to make a difference for those who matter most — the kids.”
The annual rankings are the most comprehensive source of quality-related information on U.S. pediatric hospitals and recognizes the nation’s top 50 pediatric hospitals based on a scoring system developed by U.S. News.
“For nearly two decades, U.S. News has published Best Children’s Hospitals to empower the parents and caregivers of children with complex medical needs,” said Ben Harder, chief of health analysis and managing editor at U.S. News. “Children’s hospitals appearing on the U.S. News Honor Roll have a track record of delivering unparalleled specialized care.”
The bulk of the score for each specialty service is based on quality and outcomes data. The process includes a survey of relevant specialists across the country, who are asked to list hospitals they believe provide the best care for patients with the most complex conditions.
The Children’s National specialty services that U.S. News ranked in the top 10 nationally are:
- Neonatology (No. 2), led by Billie Lou Short, M.D.
- Neurology and Neurosurgery (No. 5), led by William D. Gaillard, M.D., and Robert F. Keating, M.D.
- Cancer (No. 5), led by Eugene Hwang, M.D.
- Diabetes and Endocrinology (No. 7), led by Andrew Dauber, M.D., MMSc.
- Gastroenterology and GI surgery (No. 8), led by Ian Leibowitz, M.D., Marc Levitt, M.D., Anthony Sandler, M.D., and Timothy Kane, M.D.
- Nephrology (No. 8), led by Marva Moxey-Mims, M.D.
- Orthopedics (No. 9), led by Matthew Oetgen, M.D., M.B.A.
The other four specialties ranked among the top 50 are Behavioral Health, Cardiology and Heart Surgery, Pulmonology and Lung Surgery, and Urology.
Think tank improves care for Malawian children with seizures, epilepsy
Posters at the REI Week 2025 Monday, March 31 poster session.

Panelists discuss innovation during REI Week 2025.

Global Health Initiative community engagement event during REI Week 2025.

Chris Rees presents his REI Week 2025 lecture.

Nathan Kuppermann listens to a presenter during the REI Week 2025 Tuesday, April 1, poster session.

Michelle Riley-Brown, Nathan Kuppermann, Catherine Bollard and Naomi Luban on stage during the REI Week 2025 awards ceremony.

Brandy Salmon presents on innovation programs at Virginia Tech during the REI Week 2025 Innovation Day.

Catherine Bollard listens to a presenter during the REI Week 2025 Monday, March 21 poster session.

Ambroise Wonkman poses for a picture with Children’s National staff.

Tanzeem Choudhury presenting during REI Week 2025.

Global health leaders from Children’s National Hospital joined Malawian clinical experts in a two-day think tank to improve the care of children with seizures in the southern African nation. Details were published recently in a perspective in the American Journal of Tropical Medicine and Hygiene.
“The meeting was eye-opening,” said Douglas Postels, M.D., a pediatric neurologist and faculty member with the Center for Translational Research at Children’s National. “Outcomes are changed when we collaborate directly and work through the best solutions for clinical care in low-resource settings. You must weigh what clinics need, what they can implement, and the impact on public health.”
The big picture
Dr. Postels does research and provides clinical care at Queen Elizabeth Central Hospital (QECH), the largest public hospital in Malawi. “Queen’s” is the only medical setting in a country of over 20 million people where an electroencephalogram (EEG) and specialty pediatric neurology services are available. QECH is an essential — and often overtaxed — component of neurological care in Malawi. Cerebral malaria, epilepsy and seizures from birth asphyxia drive the need for neurological evaluations.
EEG is available at QECH for all hospitalized children and is principally used to evaluate for subclinical seizures in those who are comatose. One of three trained technicians acquires the EEG, which is electronically sent to the United States for interpretation. Under best-case scenarios, the turnaround time between EEG collection and interpretation from a neurologist in the U.S. is about two hours. However, many obstacles can slow results, including misaligned schedules across time zones, internet outages and other complications.
With the support of the Center for Translational Research, Dr. Postels brought five U.S.-based colleagues to Africa to meet with Malawian clinicians to better understand local clinical care needs. A think tank of U.S. and Malawian clinicians worked together to consider research directions and priorities for neurology care and EEG services. Their conversation focused extensively on integrating technology in the places where it will be most clinically useful.
The fine print
Dr. Postels’ research group aims to aid in the clinical care of Malawian children with epilepsy and to make EEG accessible outside of Queen’s hospital. They hope to eventually create a point-of-care EEG system, linking a method of EEG acquisition that requires little training to apply with a software-based interpretation system.
Expanding the use of EEG in Malawi and throughout Africa has numerous clinical applications. Malawian clinicians hoped that EEG could be made available at their local hospitals and folded into a more comprehensive care algorithm for children at risk of seizures — “one aspect of a ‘packet of care’ that included better overall understanding of seizures, criteria for administration of antiseizure medications, and transfer to the central hospital,” according to the journal article.
Malawian clinicians were also interested in using EEG to detect increased intracranial pressure after head trauma, as neuroimaging services are nearly as rare as EEG in Malawi. One pediatrician also wanted to use EEG to guide the treatment of neonatal seizures, which are common in children with birth asphyxia, and to use EEG as a prognostic biomarker.
“We hope to empower Malawian clinicians with clinical research skills and experiences so that they can lead and direct future discussions with patients, parents and stakeholders to advance neurologic care and research in their country,” Dr. Postels and his team wrote.
To dive deeper, read the complete perspective — “Establishing Priorities for Epilepsy Care and Electroencephalogram Use in Low-Resource Settings” — in the American Journal of Tropical Medicine and Hygiene.
New philanthropic support from the United Arab Emirates furthers research breakthroughs and care

His Highness Sheikh Mohamed bin Zayed Al Nahyan, President of the United Arab Emirates (right) visited Children’s National in September 2024.
Continuing a 30-year partnership that has yielded 82 U.S. patents and countless medical breakthroughs for kids and their families, the Government of the United Arab Emirates (UAE) has strengthened its transformational commitment to Children’s National Hospital with a new $35 million donation focused on prenatal, neonatal and maternal health.
The announcement of the new gift comes after a recent visit to the hospital by His Highness Sheikh Mohamed bin Zayed Al Nahyan, President of the United Arab Emirates (UAE), who met with Emirati families and patients receiving care at Children’s National Hospital.
The investment is the latest chapter of a longstanding philanthropic partnership between the UAE and Children’s National. Each year, more than 100 Emirati families travel to Children’s National for advanced pediatric care and life-saving treatments.
This latest investment will bolster various strategic health initiatives, including within the hospital’s Center for Prenatal, Neonatal & Maternal Health Research and the Zickler Family Prenatal Pediatrics Institute.
Researchers in the Center for Prenatal, Neonatal & Maternal Health Research are focused on the role of perinatal factors — including maternal stress, anxiety and depression — on the developing brain of the child. Studies also are revealing the impact of congenital anomalies such as heart disease and acquired conditions such as maternal infection with COVID-19 or Zika virus. New approaches to prenatal and postnatal care promise to optimize long-term outcomes of many hospitalized babies.
“Children in the Washington, D.C., area and across the world benefit greatly from the breakthroughs that have emerged from the incredible decades-long partnership between the UAE and Children’s National,” said Michelle Riley-Brown, President and CEO of Children’s National. “I am deeply grateful for the UAE’s most recent gift. The contribution will positively impact children and families and support the teams of researchers and specialists who dedicate their lives to developing innovative medical care.”
Key milestones
The UAE helped to establish the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National in 2009. Today, the Sheikh Zayed Institute (SZI) has grown into a world-class, self-sustaining research center receiving more than 80% of its funding from grants and outside sources.
This platform for invention is advancing autonomous, robotic surgery. The institute’s researchers believe pediatric surgical outcomes will improve if the precision and delicacy of a robot are incorporated into procedures such as gallbladder removal. SZI is also propelling the use of artificial intelligence to improve pediatric medicine and expand health equity. One example is a deep learning algorithm that uses hand-held ultrasounds to detect early signs of rheumatic heart disease, which kills nearly 400,000 people worldwide each year.
“The lives and health of countless children and families in the Washington area, in the UAE and around the world have been transformed by our partnership,” said Yousef Al Otaiba, the UAE Ambassador to the United States. “Our continued support promises even more breakthrough innovations in pediatric medicine.”
The UAE also supported the opening of the Children’s National Research & Innovation Campus through a 2019 commitment. The campus represents the first pediatric innovation hub of its kind, where scientists, inventors, caregivers, patients’ families and health authorities come together to advance pediatric health.
The Children’s National Rare Disease Institute and Center for Genetic Medicine Research are two of the teams housed at the campus. Together, they are pioneering care for children in the Washington region and abroad as an international referral site for rare disorders. Two examples of their research endeavors include: next-generation genomic testing to better understand how differences in genetic material can affect human health and identifying biochemical analytes.
The UAE opened a medical office in Washington, D.C., in 1991. Since then, thousands of Emirati patients have visited Children’s National for life-changing care for conditions such as congenital heart disease, neurological disorders and cancer. The hospital is currently treating 40 Emirati patients.
“Having our child treated at Children’s National means accessing specialized pediatric care from a renowned institution dedicated to children’s health,” said Hamad Alnuaimi, an Emirati father of a Children’s National patient. “It provides us with confidence and reassurance that our son is receiving the best possible medical attention from experts who understand and prioritize the unique needs of children. For the UAE to have a strong relationship with Children’s National signifies a valuable connection that enhances pediatric healthcare in our country. This partnership allows us to benefit from advanced treatments, medical innovations, and expertise that might otherwise be inaccessible. It represents a commitment to improving the health and well-being of children through international collaboration.”
Children’s National Hospital the 2024 American Academy of Pediatrics meeting
Children’s National Hospital-affiliated participants will attend this year’s American Academy of Pediatrics National Conference and Exhibition. The meeting will take place in Orlando, Florida from September 27 to October 1. You will find a mini schedule of sessions below.
Children’s National partnership addresses postpartum depression

Research shows one in eight new moms will experience sadness, hopelessness and feelings of overwhelm in the first year after having a baby, a statistic that is even higher for those with newborns receiving emergency services.
Physician researchers at Children’s National Hospital have partnered with Assistant Professor Niyousha Hosseinichimeh, Ph.D., in Virginia Tech’s College of Engineering to introduce screenings for postpartum depression.
This is all part of a four year $1.8 million grant from the Agency for Healthcare Research and Quality (AHRQ) that funds a Perinatal Mood and Anxiety Patient Safety Lab. The goal is to improve mental health screening for parents through referral and treatment, combining medical expertise with systems engineering to advance healthcare.
Research shows one in eight new moms will experience sadness, hopelessness and feelings of overwhelm in the first year after having a baby, a statistic that is even higher for those with newborns receiving emergency services.
Q: How will this partnership drive research forward in postpartum depression?
A: “In this collaboration with Children’s National, we are looking at the parents of children who are being seen in the neonatal intensive care unit (NICU) and emergency department. In a pediatric setting, there aren’t services or providers who can treat the parents or adults of the children receiving treatment, but data shows that these caregivers are more likely to need mental health services,” Hosseinichimeh said. “Right now, the screening rate is low, and we want to understand how we can improve the screening and referral process to provide better mental healthcare for adults in these settings.
Q: What does the public need to know about postpartum depression?
A: “After admission to our NICU, 45% of parents screen positive for depression. I can’t think of any other disorder or disease that screens positive at 45%. Similarly, mothers of infants in our emergency department screened positive at 27%. This can’t be ignored,” said Lamia Soghier, M.D., neonatologist and associate division chief for NICU operations at Children’s National.
Pandemic stress in pregnant mothers may affect anxiety regions of babies’ brains

The research from Children’s National Hospital provides mounting evidence that children of the pandemic, even those far too young to understand it, need ongoing assessments of developmental or mental health support later in life.
A critical part of the brain linked to risks for anxiety later in life – the left amygdala – was significantly smaller by volume in babies of mothers who reported stress during the COVID-19 pandemic, according to a new manuscript published in JAMA Network Open.
The right hippocampus, which governs spatial, visual and verbal memories, and the white matter were also reduced in children whose mothers reported stress.
The research from Children’s National Hospital provides mounting evidence that children of the pandemic, even those far too young to understand it, need ongoing assessments of developmental or mental health support later in life.
“Looking ahead, we want to use this information – and studies with similar findings – to empower pregnant mothers to request support to mitigate their stress, especially in the event of another global health crisis,” said Nickie Andescavage, M.D., a neonatologist and principal investigator at the Center for Prenatal, Neonatal & Maternal Health Research. “We also want to make sure babies born during COVID-19 get the services that they need in life if they develop anxiety or other mental health disorders.”
The fine print
Researchers at the center used magnetic resonance imaging (MRI) to compare the brains of 103 babies born between 2014 and 2019 prior to the pandemic to 59 born between 2020 and 2022. Mothers who had COVID-19 or other complications in their pregnancies were excluded. The babies underwent MRI imaging while in utero and again soon after delivery.
The mothers were evaluated for stress and anxiety, using the Spielberger State-Train Anxiety Inventory and other evidence-based scoring measures. Pre-pandemic, 21% of mothers reported elevated symptoms of anxiety; in the pandemic cohort, that number jumped to nearly 62%.
Their babies’ brains were also changed, as regions widely understood to control emotion and anxiety displayed smaller volumes on MRI imaging. Given the global impact of the pandemic and universal reports of mental distress worldwide, the potential impact of these findings may impact an entire generation of children born during the pandemic. The team is just beginning to unravel the medical significance.
What’s next
Catherine Limperopoulos, Ph.D., director of the Center for Prenatal, Neonatal & Maternal Health Research, said understanding the impact of stress is vital in supporting the healthy development of young children. Current studies are underway at her center to tease apart the role of stress in prenatal development and examine its long-term impact on development, including cognition, behavior and mental health.
“We all know that being pregnant can be quite stressful, and there are certain times of collective stress that can provide us windows to understand how the body and mind manage it,” Dr. Limperopoulos said. “At our center, we care deeply about the health of mothers and babies, and our researchers plan to continue investigating the role of stress in development to continue building data to show that mental health must be a greater priority.”
This study – “Prenatal maternal psychological distress during the COVID-19 pandemic and newborn brain development” – was supported by the National Institutes of Health, the Intellectual and Developmental Disabilities Research Center, and the A. James & Alice B. Clark Foundation. You can read the full study in JAMA Network Open.