AATS Valve Symposium hones techniques for heart valve repair over replacement in congenital heart disease
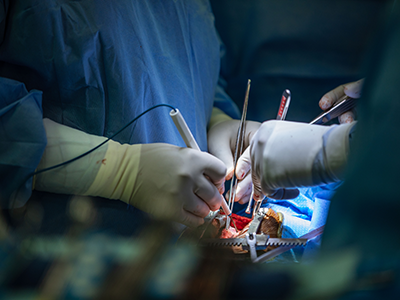
Dr. d’Udekem and the cardiac surgeons at Children’s National are well-versed in many of the most delicate techniques required to successfully complete repair procedures.
Yves d’Udekem, MD, PhD, chief of Cardiac Surgery at Children’s National Hospital, recently partnered with Emile Bacha, MD, president of the American Association for Thoracic Surgery (AATS), to offer the Congenital Valve Symposium in conjunction with the AATS 106th Annual Meeting.
The session created an opportunity for pediatric and adult cardiac surgeons to review case studies and surgical procedure videos as well as share tips and techniques for valve repair in children and adults with congenital heart disease. More than 250 people attended. This seminar is an example of the educational effort from leading cardiac surgeons to increase successful valve repair procedures and reduce the need for mechanical valve replacements, which can have challenging long-term side effects for this population.
Why it matters
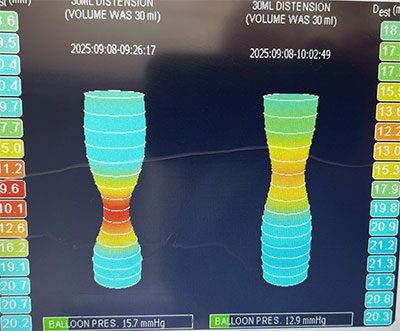
The AATS notes that the most frequently performed valve procedure in the pediatric population is a valve replacement, where a malfunctioning valve is removed and replaced with a mechanical one. However, research suggests that repairing an existing, living tissue valve may be the preferable strategy for long-term survival of children with congenital heart disease. The Division of Cardiac Surgery at Children’s National emphasizes repair techniques rather than replacement whenever possible.
The hold-up in the field
The delicate nature of the valves and the need for specific training in a variety of effective repair techniques has helped valve replacement remain the primary approach in pediatric cardiac surgery. The Children’s National team recently wrote, “Valve repair techniques in pediatric patients are frequently more intricate and less readily standardized compared to their adult counterparts due to the presence of complex and often dysplastic morphologies, consequently, a diverse range of surgical techniques is often necessitated within the pediatric population.”
Children’s National leads the way
There is a growing body of evidence for valve repair over replacement and the success of recent novel procedures such as partial heart transplants that replace malfunctioning valves with living valve tissue from a donor heart. Dr. d’Udekem and the cardiac surgeons at Children’s National are well-versed in many of the most delicate techniques required to successfully complete repair procedures. But successfully performing them at a handful of institutions is only the start of changing the standard of care.
“As valve repair becomes a fundamental part of our armamentarium in pediatric cardiac surgery, we should focus on organizing teaching of the younger generations of surgeons,” they wrote.
The AATS Congenital Valve Symposium is the most recent example of how the entire division participates in knowledge sharing and educational exchange to improve surgical care for all children with congenital heart disease. In addition to events, the team frequently welcomes cardiac surgery fellows and visiting surgeons to the operating rooms at Children’s National to study techniques. They also serve as authors and co-authors for a steady stream of peer-reviewed studies with detailed professional illustrations of techniques, tips and tricks.
See the scientific program for the recent AATS Congenital Valve Symposium to learn more about the presentations, topics and speakers.




 The division of pediatric cardiac surgery at
The division of pediatric cardiac surgery at 









 Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.
Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.



 In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.
In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.









 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.


