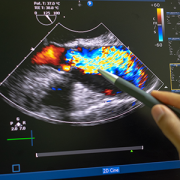
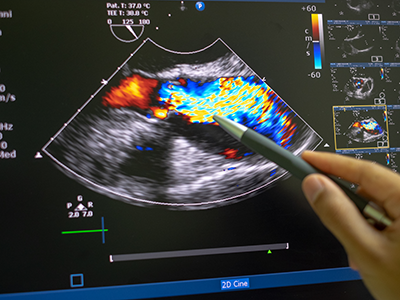
Novel AI platform matches cardiologists in detecting rheumatic heart disease
Artificial intelligence (AI) has the potential to detect rheumatic heart disease (RHD) with the same accuracy as a cardiologist, according to new research demonstrating how sophisticated deep learning technology can be applied to this disease of inequity. The work could prevent hundreds of thousands of unnecessary deaths around the world annually.
Developed at Children’s National Hospital and detailed in the latest edition of the Journal of the American Heart Association, the new AI system combines the power of novel ultrasound probes with portable electronic devices installed with algorithms capable of diagnosing RHD on echocardiogram. Distributing these devices could allow healthcare workers, without specialized medical degrees, to carry technology that could detect RHD in regions where it remains endemic.
RHD is caused by the body’s reaction to repeated Strep A bacterial infections and can cause permanent heart damage. If detected early, the condition is treatable with penicillin, a widely available antibiotic. In the United States and other high-income nations, RHD has been almost entirely eradicated. However, in low- and middle-income countries, it impacts the lives of 40 million people, causing nearly 400,000 deaths a year.
“This technology has the potential to extend the reach of a cardiologist to anywhere in the world,” said Kelsey Brown, M.D., a cardiology fellow at Children’s National and co-lead author on the manuscript with Staff Scientist Pooneh Roshanitabrizi, Ph.D. “In one minute, anyone trained to use our system can screen a child to find out if their heart is demonstrating signs of RHD. This will lead them to more specialized care and a simple antibiotic to prevent this degenerative disease from critically damaging their hearts.”
The big picture

The new AI system combines the power of novel ultrasound probes with portable electronic devices installed with algorithms capable of diagnosing RHD on echocardiogram.
Millions of citizens in impoverished countries have limited access to specialized care. Yet the gold standard for diagnosing RHD requires a highly trained cardiologist to read an echocardiogram — a non-invasive and widely distributed ultrasound imaging technology. Without access to a cardiologist, the condition may remain undetected and lead to complications, including advanced cardiac disease and even death.
According to the new research, the AI algorithm developed at Children’s National identified mitral regurgitation in up to 90% of children with RHD. This tell-tale sign of the disease causes the mitral valve flaps to close improperly, leading to backward blood flow in the heart.
Beginning in March, Craig Sable, M.D., interim division chief of Cardiology, and his partners on the project will implement a pilot program in Uganda incorporating AI into the echo screening process of children being checked for RHD. The team believes that a handheld ultrasound probe, a tablet and a laptop — installed with the sophisticated, new algorithm — could make all the difference in diagnosing these children early enough to change outcomes.
“One of the most effective ways to prevent rheumatic heart disease is to find the patients that are affected in the very early stages, give them monthly penicillin for pennies a day and prevent them from becoming one of the 400,000 people a year who die from this disease,” Dr. Sable said. “Once this technology is built and distributed at a scale to address the need, we are optimistic that it holds great promise to bring highly accurate care to economically disadvantaged countries and help eradicate RHD around the world.”
Children’s National Hospital leads the way
To devise the best approach, two Children’s National experts in AI — Dr. Roshanitabrizi and Marius George Linguraru, D.Phil., M.A., M.Sc., the Connor Family Professor in Research and Innovation and principal investigator in the Sheikh Zayed Institute for Pediatric Surgical Innovation — tested a variety of modalities in machine learning, which mimics human intelligence, and deep learning, which goes beyond the human capacity to learn. They combined the power of both approaches to optimize the novel algorithm, which is trained to interpret ultrasound images of the heart to detect RHD.
Already, the AI algorithm has analyzed 39 features of hearts with RHD that cardiologists cannot detect or measure with the naked eye. For example, cardiologists know that the heart’s size matters when diagnosing RHD. Current guidelines lay out diagnostic criteria using two weight categories — above or below 66 pounds — as a surrogate measure for the heart’s size. Yet the size of a child’s heart can vary widely in those two groupings.
“Our algorithm can see and make adjustments for the heart’s size as a continuously fluid variable,” Dr. Roshanitabrizi said. “In the hands of healthcare workers, we expect the technology to amplify human capabilities to make calculations far more quickly and precisely than the human eye and brain, saving countless lives.”
Among other challenges, the team had to design new ways to teach the AI to handle the inherent clinical differences found in ultrasound images, along with the complexities of evaluating color Doppler echocardiograms, which historically have required specialized human skill to evaluate.
“There is a true art to interpreting this kind of information, but we now know how to teach a machine to learn faster and possibly better than the human eye and brain,” Dr. Linguraru said. “Although we have been using this diagnostic and treatment approach since World War II, we haven’t been able to share this competency globally with low- and middle-income countries, where there are far fewer cardiologists. With the power of AI, we expect that we can, which will improve equity in medicine around the world.”



 The hospital will advocate for the unique needs of children as part of nationwide network working to accelerate transformative health solutions.
The hospital will advocate for the unique needs of children as part of nationwide network working to accelerate transformative health solutions.
 Children’s National Hospital was awarded nearly $7.5 million in a five-year grant to continue its leadership of an FDA-funded pediatric device consortium. Building upon a decade of previous consortium leadership, the new consortium is Alliance for Pediatric Device Innovation (APDI) and features a new and expanded roster of partners that reflects its added focus on providing pediatric innovators with expert support on evidence generation, including the use of real-world evidence (RWE), for pediatric device development.
Children’s National Hospital was awarded nearly $7.5 million in a five-year grant to continue its leadership of an FDA-funded pediatric device consortium. Building upon a decade of previous consortium leadership, the new consortium is Alliance for Pediatric Device Innovation (APDI) and features a new and expanded roster of partners that reflects its added focus on providing pediatric innovators with expert support on evidence generation, including the use of real-world evidence (RWE), for pediatric device development.






 Children’s National Hospital is joining a team of global health researchers to use large language models (LLMs) like ChatGPT to help Kenyan youth learn about their health and adopt lifestyles that may prevent cancer, diabetes and other non-communicable diseases.
Children’s National Hospital is joining a team of global health researchers to use large language models (LLMs) like ChatGPT to help Kenyan youth learn about their health and adopt lifestyles that may prevent cancer, diabetes and other non-communicable diseases.