Novel pediatric pacemaker shows safety, effectiveness for fragile infants in multi-center study
A pacemaker modified in a novel way to work better for the smallest children, including newborns, is safe and effective to stabilize heart rhythms for at least two years, according to a study published in Circulation: Arrhythmia and Electrophysiology, a journal of the American Heart Association.
The Kim family, whose daughter received this device, was featured on NBC Nightly News in May 2024.
What it means
The study includes the findings from 29 infants who received the novel pediatric pacemaker, which is no bigger than a AAA battery, at multiple institutions in the United States. A majority of them (79%) were born premature, weighing less than five pounds (2.3 kg).
The devices remain stable, with effective pacing, normal electrical parameters and battery longevity aligned with projections for up to two years. This design and application provides a viable alternative to standard-size generators and addresses a vital unmet need for these small patients. In fact, though the study includes data from the first 29 cases, the number of children who have received these devices across the United States today has doubled to nearly 60.
The specially modified pediatric-sized implantable pacemaker includes a Medtronic Micra sub-assembly that connects to an epicardial lead. While this makes the leadless pacemaker into one that uses leads, the resulting device is significantly smaller than any commercially available pacemaker previously on the market in the U.S.
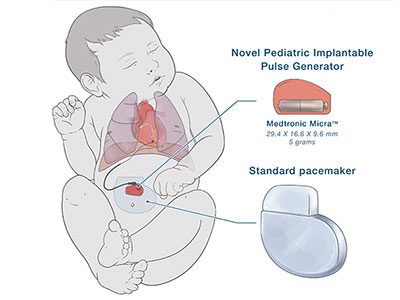
The novel pediatric implantable pulse generator is about a quarter of the size of a traditional pacemaker.
Why it matters
“The need for an urgent permanent pacemaker in newborns is quite rare, but when needed, it is often an emergency,” said lead author Charles Berul, MD, a cardiologist and electrophysiologist at Children’s National Hospital in a press release from the American Heart Association. “Babies who were very small often cannot get a permanent pacemaker and must undergo multiple temporary pacing wires or other techniques in the hopes of getting them big enough to undergo a standard pacemaker placement.”
Dr. Berul also notes that a smaller pacemaker may also help frail elderly patients and be a better choice for some children and adults.
What’s next: Better delivery
Innovating smaller devices is a good start. However, when a newborn or young child needs any pacemaker or defibrillator, they face open chest surgery. Their arteries and veins are just too small for even the smallest size transvenous pacemaker catheter.
Dr. Berul and engineers in the Sheikh Zayed Institute for Pediatric Surgical Innovation are testing a first-of-its-kind minimally invasive pericardial access tool. This tool allows for pacing and defibrillation therapy that can be delivered through a single small port inserted through the skin.
Read the study, Multicenter Results of a Novel Pediatric Pacemaker in Neonates and Infants, in Circulation: Arrhythmia and Electrophysiology, a journal of the American Heart Association.














 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the 

















 Clinicians caring for children are often left to rely on off-label devices and medications approved for adults, especially during public health crises, national disasters and other emergencies. To address this critical gap, Children’s National Hospital is launching a 10-year partnership with the federal Biomedical Advanced Research and Development Authority (BARDA) — valued at $1.5 million per year, with the possibility of an additional funding boost of up to $515 million.
Clinicians caring for children are often left to rely on off-label devices and medications approved for adults, especially during public health crises, national disasters and other emergencies. To address this critical gap, Children’s National Hospital is launching a 10-year partnership with the federal Biomedical Advanced Research and Development Authority (BARDA) — valued at $1.5 million per year, with the possibility of an additional funding boost of up to $515 million.






 The Children’s National Research Institute released its
The Children’s National Research Institute released its 