Safeguarding fetal brain health in pregnancies complicated by CHD

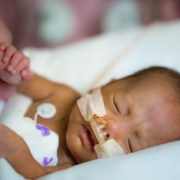
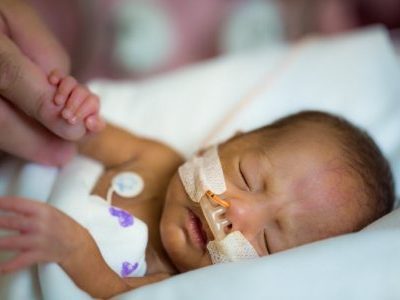
During the last few weeks of pregnancy, certain regions of the fetal brain experience exponential growth but also are more vulnerable to injury during that high-growth period.
Yao Wu, Ph.D., a research postdoctoral fellow in the Developing Brain Research Laboratory at Children’s National Health System, has received a Thrasher Research Fund early career award to expand knowledge about regions of the fetal brain that are vulnerable to injury from congenital heart disease (CHD) during pregnancy.
CHD, the most common birth defect, can have lasting effects, including overall health issues; difficulty achieving milestones such as crawling, walking or running; and missed days at daycare or school, according to the Centers for Disease Control and Prevention. Brain injury is a major complication for infants born with CHD. Catherine Limperopoulos, Ph.D., director of Children’s brain imaging lab, was the first to provide in vivo evidence that fetal brain growth and metabolism in the third trimester of pregnancy is impaired within the womb.
“It remains unclear which specific regions of the fetal brain are more vulnerable to these insults in utero,” Limperopoulos says. “We first need to identify early brain abnormalities attributed to CHD and understand their impact on infants’ later behavioral and cognitive development in order to better counsel parents and effectively intervene during the prenatal period to safeguard brain health.”
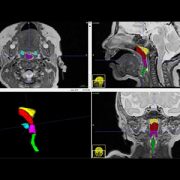
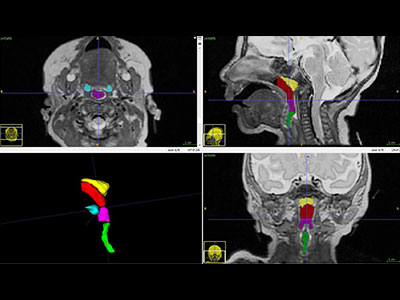
During the last few weeks of pregnancy, certain regions of the fetal brain experience exponential growth but also are more vulnerable to injury during that high-growth period. The grant, $26,749 over two years, will underwrite “Brain Development in Fetuses With Congenital Heart Disease,” research that enables Wu to utilize quantitative, non-invasive magnetic resonance imaging (MRI) to compare fetal brain development in pregnancies complicated by CHD with brain development in healthy fetuses of the same gestational age.Wu will leverage quantitative, in vivo 3-D volumetric MRI to compare overall fetal and neonatal brain growth as well as growth in key regions including cortical grey matter, white matter, deep grey matter, lateral ventricles, external cerebrospinal fluid, cerebellum, brain stem, amygdala and the hippocampus.
The research is an offshoot of a prospective study funded by the National Institutes of Health that uses advanced imaging techniques to record brain growth in 50 fetuses in pregnancies complicated by CHD who need open heart surgery and 50 healthy fetuses. MRI studies are conducted during the second trimester (24 to 28 weeks gestational age), third trimester (33 to 37 weeks gestational age) and shortly after birth but before surgery. In addition, fetal and neonatal MRI measurements will be correlated with validated scales that measure infants’ and toddlers’ overall development, behavior and social/emotional maturity.
“I am humbled to be selected for this prestigious award,” Wu says. “The findings from our ongoing work could be instrumental in identifying strategies for clinicians and care teams managing high-risk pregnancies to optimize fetal brain development and infants’ overall quality of life.”