Babies with congenital heart disease display disrupted brain function before birth

In their study, the team at Children’s National Hospital found that specific brain regions become especially vulnerable to injury around 24 weeks of pregnancy when developing babies begin to have high energy demands and rapid neurovascular changes.
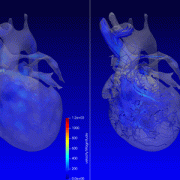
For the first time, researchers have found that babies born with congenital heart disease (CHD) have alterations to the emerging functional connectivity of their brains in utero. The changes are related to the subtype of their CHD and their oxygen status before they have lifesaving surgery to treat their cardiac malformation, according to new findings published in the American Heart Association’s Circulation Research.
In their study, the team at Children’s National Hospital found that specific brain regions become especially vulnerable to injury around 24 weeks of pregnancy when developing babies begin to have high energy demands and rapid neurovascular changes. That leaves certain parts of the brain, including the brainstem, more susceptible to injury from cardiac complications and poor circulation.
“We used a special type of magnetic resonance imaging to safely study the brains of these unborn babies, and we found that they have weakened connectivity in the deep grey structures, which are responsible for sensation, movement, alertness and other core functions,” said Josepheen De Asis-Cruz, M.D., Ph.D., assistant professor at the Developing Brain Institute at Children’s National and an author of the study. “This offers an important clue in utero to the type of care the babies will eventually need when they are born.”
The big picture
In the past decade, the survival rate for fetuses with CHD has greatly improved. About 80% of cases – even some of the most high-risk heart defects – can be successfully treated or palliated with surgery and survive. Yet Dr. Cruz said researchers are finding that the rates of poor neurodevelopmental outcomes are about the same. That’s why she and her colleagues are looking at what precisely may be injuring the brains of these newborns prior to surgery, offering a possible roadmap to interventions.
The fine print
The research team studied 107 healthy, low-risk pregnancies and 75 pregnancies known to be complicated by CHD. They used functional connectivity magnetic resonance imaging (fcMRI) to examine the emerging connections of the brains of unborn babies, given fcMRI’s unique ability to query the brain in a resting state when a patient is unable to respond to tasks. They also studied the oxygen saturation levels of the babies after they were born and then mapped all of this information to the type of CHD that they were diagnosed with.
“Our findings indicate that the compromised connectivity in the brains of CHD patients before delivery is linked to hypoxia after birth,” Cruz said. “There were important differences in the low- and high-risk CHD groups. Babies born with transposition of the great arteries or hypoplastic left heart syndrome – two of the most high-risk diagnoses – have notable changes in their brain function, which could someday be used as biomarkers to guide their care.”
What’s ahead
Researchers at Children’s National are working together, using a variety of modalities, to move toward precision imaging in utero to help predict a child’s neurodevelopmental outcomes. The ultimate goal: better interventions sooner.
“This work is foundational. As we fine-tune more techniques to identify babies at risk, we can understand how environmental, genetic and epigenetic factors impact brain development and guide care decisions,” said Catherine Limperopoulos, Ph.D., director of the Center for Prenatal, Neonatal & Maternal Health Research and a senior author of the paper. “We can imagine a day where we can offer pregnant mothers highly detailed and personalized information about their unborn baby, and individualized interventions that lead to healthier lifetimes.”