REI Week 2025 empowers the future in pediatric research and innovation
Children’s National Hospital hosted its fifteenth annual Research, Education and Innovation Week from March 31–April 4, 2025, bringing together clinicians, scientists, educators and innovators from across the institution to celebrate discovery and collaboration. This year’s theme, “Empowering the Future in Pediatric Research and Innovation with Equity, Technology and a Global Reach,” served as a call to action for advancing science that improves child health both locally and around the world.
Each day of the week-long event featured thought-provoking lectures — now available to watch — dynamic panel discussions, interactive workshops and vibrant poster sessions, all highlighting the diverse and interdisciplinary work taking place across Children’s National.
Centering the patient and the planet
REI Week began on Monday with a powerful keynote lecture from Lynn R. Goldman, MD, MS, MPH, Michael and Lori Milken dean of the Milken Institute School of Public Health at the George Washington University. In her talk, “Children: Uniquely vulnerable to climate-related threats,” Dr. Goldman underscored the urgent need to protect children from the environmental hazards of a changing climate and to integrate climate science into pediatric care and advocacy.
At mid-morning, Mary-Anne “Annie” Hartley, MD, PhD, MPH, director of the LiGHT Laboratory at École Polytechnique Fédérale de Lausanne, introduced the “MOOVE” platform — Massive Open Online Validation and Evaluation of clinical LLMs. Her talk demonstrated how artificial intelligence, when rigorously validated, has the potential to transform clinical decision-making and global health equity.
Monday’s final keynote, “Zinc and childhood diarrhea,” was presented by Christopher Duggan, MD, MPH, director of the Division of Nutrition at Harvard Medical School. Dr. Duggan highlighted the global health impact of zinc supplementation in reducing childhood mortality — a reminder that simple, evidence-based interventions can save millions of lives.
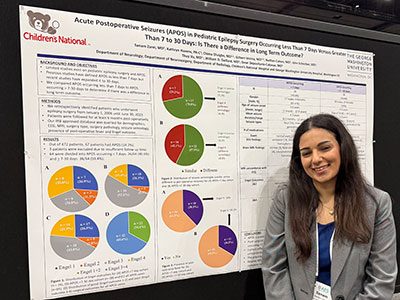
In that first day, the first poster session of the week showcased projects in adolescent medicine, global health, infectious diseases, oncology and more. The session reflected the full breadth of research taking place across Children’s National.
Ambroise Wonkam, MD, PhD, professor of genetic medicine at Johns Hopkins University, then delivered Tuesday’s Global Health Keynote Lecture, “Harnessing our common African genomes to improve health and equity globally.” His work affirmed that inclusive genomics is key to building a healthier world.
Later, the Global Health Initiative event and GCAF Faculty Seminar encouraged attendees to pursue collaborative opportunities at home and abroad, reflecting the growing global footprint of Children’s National research programs.
Transforming education and care delivery
On Wednesday, Larrie Greenberg, MD, professor emeritus of pediatrics, kicked off the day with a Grand Rounds keynote on educational transformation: “Shouldn’t teachers be more collaborative with their learners?” He followed with a CAPE workshop exploring the effectiveness of case-based learning.
The Nursing Sponsored Keynote Lecture by Vincent Guilamo-Ramos, PhD, MPH, LCSW, ANP-BC, PMHNP-BC, FAAN, explored “Redesigning the U.S. broken health system.” He offered an urgent and inspiring call to reimagine pediatric care by addressing social determinants of health.
In the Jill Joseph Grand Rounds Lecture, Deena J. Chisolm, PhD, director of the Center for Child Health Equity at Nationwide Children’s Hospital, challenged attendees to move beyond dialogue into action in her talk, “Health equity: A scream to a whisper?,” reminding researchers and clinicians that advocacy and equity must be foundational to care.
The day continued with a poster session spotlighting medical education, neonatology, urology and neuroscience, among other fields.
Posters and pathways to progress
Throughout the week, poster sessions highlighted cutting-edge work across dozens of pediatric disciplines. These sessions gave attendees the opportunity to engage directly with investigators and reflect on the shared mission of discovery across multiple disciplines, including:
- Neuroscience and Neuroprotection – Novel approaches to brain injury treatment and neurodevelopmental disorders.
- Genetics and Precision Medicine – Advances in understanding genetic predisposition and innovative therapies.
- Health Disparities and Global Health – Strategies to bridge gaps in pediatric healthcare access worldwide.
- Innovative Technology in Pediatrics – Breakthroughs in AI, bioinformatics and medical device development.
Honoring excellence across Children’s National
The REI Week 2025 Awards Ceremony celebrated outstanding contributions in research, mentorship, education and innovation. The winners in each category were:
POSTER SESSION AWARDS
Basic & Translational Research
Faculty: Benjamin Liu, PhD
“Genetic Conservation and Diversity of SARS-CoV-2 Envelope Gene Across Variants of Concern”
Faculty: Steve Hui, PhD
“Brain Metabolites in Neonates of Mothers with COVID-19 Infection During Pregnancy”
Faculty: Raj Shekhar, PhD
“StrepApp: Deep Learning-Based Identification of Group A Streptococcal (GAS) Pharyngitis”
Post docs/Fellows/Residents: Dae-young Kim, PhD
“mhGPT: A Lightweight Domain-Specific Language Model for Mental Health Analysis”
Post docs/Fellows/Residents: Leandros Boukas, MD, PhD
“De Novo Variant Identification From Duo Long-Read Sequencing: Improving Equitable Variant Interpretation for Diverse Family Structures”
Staff: Naseem Maghzian
“Adoptive T Lymphocyte Administration for Chronic Norovirus Treatment in Immunocompromised Hosts (ATLANTIC)”
Graduate Students: Abigail Haffey
“Synergistic Integration of TCR and CAR T Cell Platforms for Enhanced Adoptive Immunotherapy in Brain Tumors”
High School/Undergraduate Students: Medha Pappula
“An ADHD Diagnostic Interface Based on EEG Spectrograms and Deep Learning Techniques”
Clinical Research
Faculty: Folasade Ogunlesi, MD
“Poor Air Quality in Sub-Saharan Africa is Associated with Increase Health Care Utilization for Pain in Sickle Cell Disease Patients”
Faculty: Ayman Saleh, MD
“Growth Parameters and Treatment Approaches in Pediatric ADHD: Examining Differences Across Race”
Post docs/Fellows/Residents: Nicholas Dimenstein, MD, MPH
“Pre-Exposure Prophylaxis (PrEP) Eligibility in the Pediatric Emergency Department”
Staff: Tayla Smith, MPH
“The Public Health Impact of State-Level Abortion and Firearm Laws on Health Outcomes”
Graduate Students: Natalie Ewing
“Patterns of Bacteriuria and Antimicrobial Resistance in Patients Presenting for Primary Cloacal Repair: Is Assisted Bladder Emptying Associated with Bacteriuria?”
Graduate Students: Manuela Iglesias, MS
“Exploring the Relationship Between Child Opportunity Index and Bayley-III Scores in Young Children”
High School/Undergraduate Students: Nicholas Lohman
“Preliminary Findings: The Efficacy, Feasibility and Acceptability of Group Videoconference Cognitive Behavioral Therapy with Exposure and Response Prevention for Treating Obsessive-Compulsive Disorder Among Children and Young People”
Community-Based Research
Faculty: Sharon Shih, PhD
“Assessing Pediatric Behavioral Health Access in DC using Secret Shopper Methodology”
Post docs/Fellows/Residents: Georgios Sanidas, MD
“Arrested Neuronal Maturation and Development in the Cerebellum of Preterm Infants”
Staff: Sanam Parwani
“Intersectionality of Gender and Sexuality Diversity in Autistic and Non-Autistic Individuals”
Graduate Student: Margaret Dearey
“Assessing the Burden of Period Poverty for Youth and Adolescents in Washington, DC: A Pilot Study”
Quality and Performance Improvement
Faculty: Nichole L. McCollum, MD
“A Quality Improvement Study to Increase Nurse Initiated Care from Triage and Improve Timeliness to Care”
Post docs/Fellows/Residents: Hannah Rodriguez, MD
“Reducing Unnecessary Antibiotic Use in a Level IV NICU”
Staff: Amber K. Shojaie, OTD, OTR/L
“Implementing Dynamic Axilla Splints in a Large Burn Patient”
MENTORSHIP AWARDS
Basic Science Research
Conrad Russell Y. Cruz, MD, PhD
Clinical Research
Bench to Bedside Research
Ioannis Koutroulis, MD, PhD, MBA
ELDA ARCE TEACHING SCHOLAR AWARD
SUZANNE FEETHAM NURSING RESEARCH SUPPORT AWARD
Eileen P. Engh, PhD RN
“Rare Disease Organization Lifecycle” Role in Helping Parents with Everyday Life Information Seeking and Connection (RDO-HELIX)
EXPLORATIONS IN GLOBAL HEALTH PILOT AWARDS
Launchpad Awards
Mi Ran Shin, MD, MPH
“Establishing Interdisciplinary Rehabilitation for Birth and Burn Injuries in Ethiopia”
Susan Harvey, MSN, CPNP-AC
“Implementation of Sickle Cell Pilot Program in Ndhiwa Sub County, Kenya”
Meleah Boyle, PhD, MPH
“Understanding and Addressing Environmental Sustainability to Protect the Health of the Children’s National and Global Communities”
Eiman Abdulrahman, MD
“Research Capacity Building to Improve Pediatric Emergency and Critical Care in Ethiopia”
Pilot Awards
Alexander Andrews, MD
“EEG as a Diagnostic and Prognostic Marker in Severe Pediatric Malaria, Blantyre Malawi”
Daniel Donoho, MD & Timothy Singer, MD
“Feasibility Study of a Novel Artificial Intelligence-Based Educational Platform to Improve Neurosurgical Operative Skills in Tanzania”
Hasan Syed, MD
“Bridging the Gap an Educational Needs Assessment for Pediatric Neurosurgery Training in Pakistan”
Sofia Perazzo, MD & Lamia Soghier, MD, MEd, MBA
“QI Mentorship to Improve Pediatric Screening and Follow-up in Rural Argentina”
Benjamin Liu, PhD
“AI-Empowered Real-Time Sequencing Assay for Rapid Detection of Schistosomiasis in Senegal”
Rae Mittal, MD
“Assessment and Enhancement of Proficiency in Emergency Child Neurology Topics for Post-Graduate Emergency Medicine Trainees in India”
Innovation Day ignites bold thinking
Thursday, REI Week shifted to the Children’s National Research & Innovation Campus for Innovation Day, a celebration of how bold ideas and collaborative culture can accelerate progress in pediatric medicine.
Brandy Salmon, PhD, associate vice president of Innovation and Partnerships at Virginia Tech, opened the day with “The Alchemy of Innovation,” focusing on how institutions can build a culture that fuels transformative partnerships.
A multidisciplinary panel discussion moderated by Nathan Kuppermann, MD, MPH, and Catherine Bollard, MBChB, MD, featured Nehal Mehta, MD, Julia Finkel, MD, Kevin Cleary, PhD, Ioannis Koutroulis, MD, PhD, MBA, Francesca Joseph, MD and Patrick Hanley, PhD, who shared how innovation can be advanced and promoted, especially as a core institutional priority.
A shared vision for the future
REI Week 2025 reaffirmed the values that define Children’s National: a commitment to excellence, collaboration and equity in pediatric research and care. As discoveries continue to emerge from our hospital and our research campuses, the connections built and ideas sparked during this week will help shape the future of pediatric health — locally and globally.
By elevating voices from the bedside to the bench, with the support of the executive sponsors Nathan Kuppermann, MD, MBChB, Catherine Bollard, MBChB, MD, Kerstin Hildebrandt, MSHS, Linda Talley, MS, RN, NE-BC and David Wessel, MD, REI Week demonstrated that we must embrace the community in all aspects of our work. Because we know that there are answers we can only get from the patients that we serve—and we need to be their voice.
Research, Education & Innovation Week will be back next year on April 13-17, 2026.












 For more than four decades, Children’s National Hospital has been at the forefront of neurofibromatosis (NF) care, research and treatment. In 2007, Dan and Jennifer Gilbert furthered this mission by establishing the
For more than four decades, Children’s National Hospital has been at the forefront of neurofibromatosis (NF) care, research and treatment. In 2007, Dan and Jennifer Gilbert furthered this mission by establishing the 

 The Divisions of Neurology and Neurosurgery at Children’s National Hospital are proud to host the 35th Annual Pediatric Neurology Update course.
The Divisions of Neurology and Neurosurgery at Children’s National Hospital are proud to host the 35th Annual Pediatric Neurology Update course.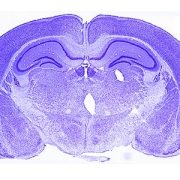













 In 2024, Children’s National Hospital continued to make remarkable strides across diverse areas of pediatric medicine, from groundbreaking technological innovations to critical health advocacy. The following compilation showcases ten significant stories that demonstrate the breadth and depth of the hospital’s impact, as featured in major national news outlets including NBC Nightly News, CNN, The Washington Post, The New York Times, NPR, The Today Show, Healio, and POLITICO. Delve into our 2024 news highlights for more.
In 2024, Children’s National Hospital continued to make remarkable strides across diverse areas of pediatric medicine, from groundbreaking technological innovations to critical health advocacy. The following compilation showcases ten significant stories that demonstrate the breadth and depth of the hospital’s impact, as featured in major national news outlets including NBC Nightly News, CNN, The Washington Post, The New York Times, NPR, The Today Show, Healio, and POLITICO. Delve into our 2024 news highlights for more.
 2024 marked another groundbreaking year for Children’s National Hospital, showcasing remarkable advances across the spectrum of pediatric medicine, research and healthcare innovation. From pioneering surgical procedures to breakthrough artificial intelligence applications, the institution continued to push the boundaries of what’s possible in children’s healthcare. Read on for our list of the most popular articles we published on Innovation District in 2024.
2024 marked another groundbreaking year for Children’s National Hospital, showcasing remarkable advances across the spectrum of pediatric medicine, research and healthcare innovation. From pioneering surgical procedures to breakthrough artificial intelligence applications, the institution continued to push the boundaries of what’s possible in children’s healthcare. Read on for our list of the most popular articles we published on Innovation District in 2024.




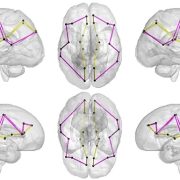
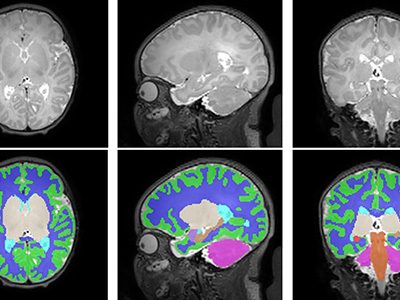
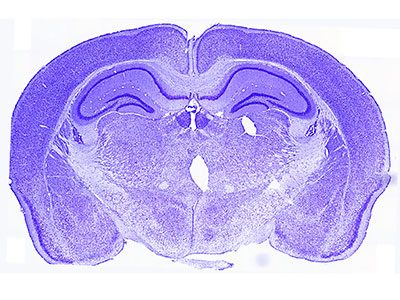
 Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the
Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the 


 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the