Sudden blindness leads to unusual diagnosis

Suresh N. Magge, M.D., and his colleagues at Children’s National recently published the details of an unusual case of advanced moyamoya disease in the journal Stroke.
When Children’s National Health System Neurosurgeon Suresh N. Magge, M.D., met his new patient, the 16-year-old had suddenly lost her vision in both eyes.
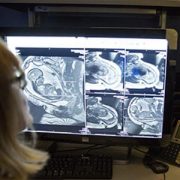
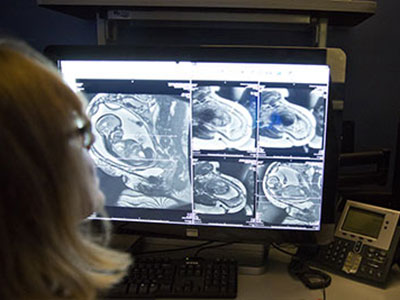
To discover the reason for this abrupt loss of vision, her doctors ran a battery of tests. An ophthalmologist found no problems with her eyes. Her optic nerves, which run signals generated from the eyes to the brain, also appeared to work normally. However, a computerized tomography scan and magnetic resonance imaging showed the unmistakable signs of a stroke in her occipital lobe, the portion of the brain responsible for interpreting signals relayed from the optic nerves.
“Her brain basically wasn’t seeing what her eyes saw,” Dr. Magge explains.
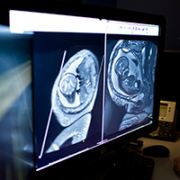
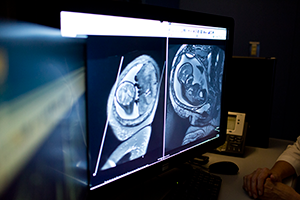
Delving deeper, her Children’s National care team found the reason why their young patient had suffered a stroke by using a cerebral angiogram, an imaging test that shows the blood vessels in and around the brain. The teen had moyamoya disease, a rare condition that causes blood vessels in the brain to narrow, often leading otherwise healthy adults and children to have strokes.
According to the National Institutes of Health, moyamoya is Japanese for “puff of smoke,” so named because of the telltale signs this condition presents on an angiogram. When arteries in the brain narrow, brain tissue becomes “thirsty” for more blood, Dr. Magge explains, leading its cells to produce chemicals that prompt new blood vessels to grow. These new collateral blood vessels often grow in a thin tangle that looks like smoke on an angiogram. Generally, however, they do not supply sufficient oxygenated blood to meet the brain’s needs, leaving it starved for oxygen. Eventually, the blood supply can get so low that patients suffer transient ischemic attacks, “mini-strokes” that temporarily deprive the brain tissue of oxygen, or full-blown strokes typically characterized by weakness, speech problems, facial paralysis or other problems.
Dr. Magge’s patient had little warning before her stroke occurred. The first major symptom that led her to seek medical attention was abrupt blindness, which Dr. Magge says is a highly unusual occurrence for a moyamoya diagnosis. That’s why he and colleagues decided to publish the details of her case as a teaching report April 14, 2017 in the journal Stroke.
The Children’s National co-authors wrote that once their patient was diagnosed with a stroke due to advanced moyamoya disease, with blood vessels severely narrowed throughout her brain, the first order of business was stabilizing her symptoms and making sure she did not have further strokes. Her blood pressure was stabilized, and she was started on aspirin therapy to decrease her risk of further strokes. She took time to recover as much as possible from her original stroke.
A few weeks later, Dr. Magge and his neurosurgery colleagues performed a type of surgery to revascularize – or restore blood flow – to areas of the brain that were still healthy but at risk of having subsequent strokes. The surgical procedure, known as pial synangiosis, reconfigures the brain’s blood vessels to make sure that these vulnerable areas of the brain have a sufficient blood supply.
Years later, Dr. Magge says, his patient is doing well, except for the original blindness, a permanent consequence of the stroke to her occipital lobe before her diagnosis. She has not had new strokes since the revascularization surgery. She will need aspirin therapy and periodic neurological checkups for the rest of her life, Dr. Magge explains, to make sure that the blood supply to her brain remains stable.
Children’s experts use a team approach to treat patients with complex care needs: Neurologists, neurosurgeons, intensivists, hematologists, anesthesiologists, neuroradiologists and nurses leverage their combined expertise with moyamoya disease to treat the complexities of this condition.
“A team approach is essential to deliver the best outcomes to children with life-changing diseases,” Dr. Magge says. “We try to help kids get back to living full and healthy lives.”