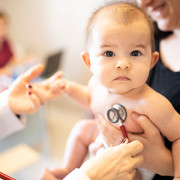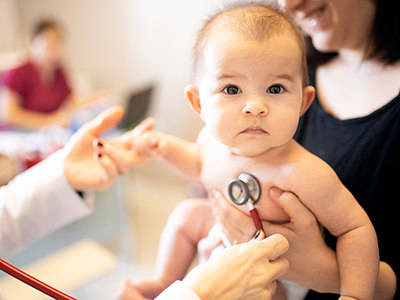
Despite early reports suggesting COVID-19 does not seriously impact children, a new study shows that children who contract COVID-19 can become very ill.
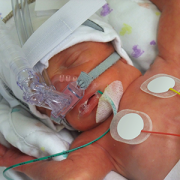
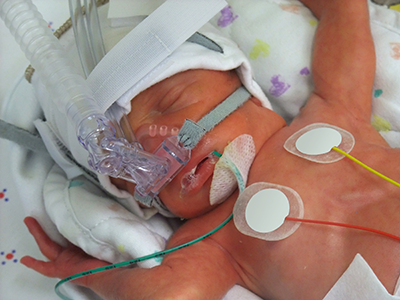
In contrast to the prevailing view that the novel coronavirus known as COVID-19 does not seriously impact children, a new study finds that children who contract the virus can become very ill—many of them critically so, according to physician researchers at Children’s National Hospital. Their results, published in the Journal of Pediatrics and among the first reports from a U.S. institution caring for children and young adults, shows differences in the characteristics of children who recovered at home, were hospitalized, or who required life support measures. These findings highlight the spectrum of illness in children, and could help doctors and parents better predict which pediatric patients are more likely to become severely ill as a consequence of the virus.
In late 2019, researchers identified a new coronavirus, known as SARS-CoV-2, which causes COVID-19. As the disease spread around the world, the vast majority of reports suggested that elderly patients bear the vast majority of the disease burden and that children are at less risk for either infection or severe disease. However, study leader Roberta DeBiasi, M.D., M.S., chief of the Division of Infectious Diseases at Children’s National, states that she and her colleagues began noticing an influx of children coming to the hospital for evaluation of a range of symptoms starting in mid-March 2020, who were tested and determined to be infected with COVID-19. One quarter of these children required hospitalization or life support.
“It was very apparent to us within the first several weeks of the epidemic that this was a very different situation than our colleagues on the West Coast of the US had described as their experience just weeks before,” DeBiasi says. “Right away, we knew that it was important for us to not only care for these sick children, but to examine the factors causing severe disease, and warn others who provide medical care to children.”
To better understand this phenomenon, she and her colleagues examined the medical records of symptomatic children and young adults who sought treatment at Children’s National for COVID-19 between March 15 and April 30, 2020. Each of these 177 children tested positive using a rapid assay to detect SARS-CoV-2 performed at the hospital. The researchers gathered data on each patient, including demographic details such as age and sex; their symptoms; whether they had any underlying medical conditions; and whether these patients were non-hospitalized, hospitalized, or required critical care.
The results of their analysis show that there was about an even split of male and female patients who tested positive for COVID-19 at Children’s National during this time period. About 25% of these patients required hospitalization. Of those hospitalized, about 75% weren’t considered critically ill and about 25% required life support measures. These included supplemental oxygen delivered by intubation and mechanical ventilation, BiPAP, or high-flow nasal cannula – all treatments that support breathing – as well as other support measures such as dialysis, blood pressure support and medications to treat infection as well as inflammation.
Although patients who were hospitalized spanned the entire age range, more than half of them were either under a year old or more than 15 years old. The children and young adults over 15 years of age, Dr. DeBiasi explains, were more likely to require critical care.
About 39% of all COVID-19 patients had underlying medical conditions, including asthma, which has been highlighted as a risk factor for worse outcomes with this infection. However, DeBiasi says, although underlying conditions were more common as a whole in hospitalized patients – present in about two thirds of hospitalized and 80% of critically ill – asthma didn’t increase the risk of hospitalization or critical illness. On the other hand, children with underlying neurological conditions, such as cerebral palsy, microcephaly, or global developmental delay, as well as those with underlying cardiac, hematologic, or oncologic conditions were significantly more likely to require hospitalization.
In addition, although early reports of COVID-19 suggested that fever and respiratory symptoms are hallmarks of this infection, Dr. DeBiasi and her colleagues found that fewer than half of patients had both concurrently. Those with mild, upper respiratory symptoms, such as runny nose, congestion, and cough were less likely to end up hospitalized than those with more severe respiratory symptoms, such as shortness of breath. The frequency of other symptoms including diarrhea, chest pain and loss of sense of smell or taste was similar among hospitalized and non-hospitalized patients.
Dr. DeBiasi notes that although other East Coast hospitals are anecdotally reporting similar upticks in pediatric COVID-19 patients who become seriously ill, it’s currently unclear what factors might account for differences from the less frequent and milder pediatric illness on the West Coast. Some factors might include a higher East Coast population density, differences between the genetic, racial and ethnic makeup of the two populations, or differences between the viral strains circulating in both regions (an Asian strain on the West Coast, and a European strain on the East Coast).
Regardless, she says, the good news is that the more researchers learn about this viral illness, the better prepared parents, medical personnel and hospitals will be to deal with this ongoing threat.
Other researchers from Children’s National who participated in this study include Xiaoyan Song, Ph.D., M.Sc., Meghan Delaney, D.O., M.P.H., Michael Bell, M.D. , Karen Smith, M.D., Jay Pershad, M.D., Emily Ansusinha, Andrea Hahn, M.D., M.S., Rana Hamdy, M.D., M.P.H., MSCE, Nada Harik, M.D., Benjamin Hanisch, M.D., Barbara Jantausch, M.D., Adeline Koay, MBBS, MS.c., Robin Steinhorn, Kurt Newman, M.D. and David Wessel, M.D.