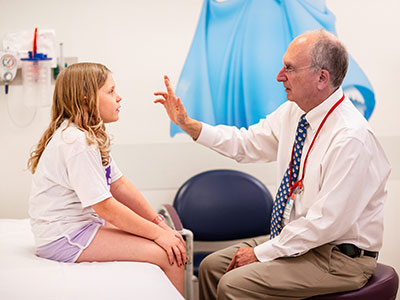
The hippobot is a mechanical horseback riding simulator that provides hippotherapy for children.
Cerebral palsy is the most common type of movement disorder in children, affecting 1 in 500 babies born each year. For these infants, learning to sit up, stand and walk can be a big challenge which often requires years of physical therapy to stretch and strengthen their muscles. A team led by Kevin Cleary, Ph.D., technical director of the Bioengineering Initiative at Children’s National Health System, and Sally Evans, M.D., director of Pediatric Rehabilitation Medicine at Children’s National, has created two new types of robotic therapy that they hope will make physical therapy more enjoyable and accessible for children.
Hippobot equine therapy simulator
One of the most effective types of therapy for children with cerebral palsy is hippotherapy, which uses horseback riding to rehabilitate children with neurological and musculoskeletal disabilities. The movement of horses helps riders with cerebral palsy improve endurance, balance and core strength, which in turns helps them gain the ability to sit without support. If a child with cerebral palsy does not master independent sitting early in life, he or she may never gain the ability to stand or walk. Unfortunately, many children never have the chance to experience hippotherapy due to geographical constraints and cost issues.
To increase patient access to hippotherapy, the bioengineering team (Reza Monfaredi, Ph.D.; Hadi Fooladi Talari, M.S.; Pooneh Roshani Tabrizi, Ph.D.; and Tyler Salvador, B.S.) developed the hippobot — a mechanical horseback riding simulator that provides hippotherapy for children ages 4 to 10 in the office setting. To create the hippobot, the researchers mounted a carousel horse on a six-degree of freedom commercial motion platform (the platform moves in the x, y and z directions and rotates about roll, pitch and yaw axes). They then programmed the platform to simulate a horse walking, trotting and cantering.
“Several experienced horse riders have tried the motion platform and commented that it gives a realistic feel,” says Dr. Cleary.
The team then incorporated optical tracking of the hippobot rider’s spine and pelvis to monitor their posture and created a virtual reality video display that simulates a horse moving down a pier. As other animals come towards the horse, the rider must lean right or left to avoid them. The trackers on their back show which way they are leaning and feed that information into the gaming system.
“We wanted to see how the patient’s spine reacts as the horse moves through different patterns, and if the patients get better at maintaining their posture over several sessions,” says Dr. Cleary.
To date the system has been used with several children with cerebral palsy under an IRB-approved study. All of the participants enjoyed riding the horse and came back for multiple sessions.
The hippobot system was developed in close collaboration with the Physical Medicine and Rehabilitation Division at Children’s National, including Olga Morozova, M.D., Justin Burton, M.D., and Justine Belschner, P.T.
Pedbot ankle rehabilitation system
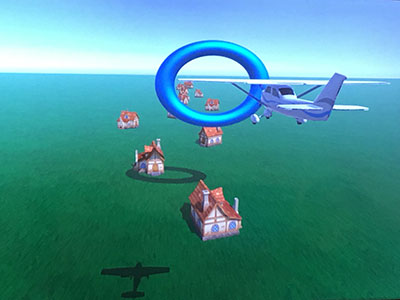
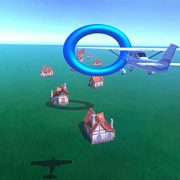
Patients use pedbot as an input device to pilot an airplane through a series of hoops. The level of the difficulty of the game can be easily adjusted based on the patient’s capability and physical condition.
More than half of children with cerebral palsy also have gait impairment as a result of excessive plantar flexion and foot inversion/eversion, or equinovarus/equinovalgus at their ankle and foot. To help these patients, Dr. Cleary’s team developed the pedbot — a small robot platform that enables better strengthening, motor control and range of motion in the ankle joint.
“Children with cerebral palsy have difficulty walking in part because they have trouble controlling their feet,” explains Dr. Evans. “Use of pedbot as part of therapy can help to give them increased control of their feet.”
Most ankle rehabilitation robots are limited in their movements, and have only one or two degrees of freedom, focusing on ankle dorsiflexion/plantarflexion and sometimes inversion/eversion. Pedbot is unique in that it has three degrees of freedom with a remote center of motion in the ankle joint area that allows it to move in ways other devices can’t.
The pedbot platform can move in three translational directions (x, y and z) and also rotate about three axes (the x, y and z axes). As an analogy, this is similar to the movement of a flight simulator. The system also includes motors and encoders at each axis and can be used in passive and active modes.
In both modes, the patient sits on a therapy chair with their foot strapped to the robotic device. In the passive mode, the therapist assists the patient in training motions along each axis. The robot can then repeat the motion under therapist supervision while incrementally increasing the range of motion as desired by the therapist.
For the active mode or “gaming” mode, the team developed a video game based on an airplane motif. Patients use pedbot as an input device to pilot an airplane through a series of hoops. The level of the difficulty of the game can be easily adjusted based on the patient’s capability and physical condition.
To date, four patients have participated in an IRB-approved clinical trial for the pedbot. All of the patients enjoyed the game and they were willing to continue to participate as suggested by a physiotherapist.
The pedbot team, in addition to the engineers mentioned above, includes Catherine Coley, P.T.; Stacey Kovelman, P.T.; and Sara Alyamani, B.A. In future work, they plan to expand the system to include electromyography measurements with Paola Pergami, M.D.,Ph.D. They also are planning to develop a low cost, 3D printed version for the home market so children can do Pedbot therapy every day.