Lab led by Zhe Han, Ph.D., receives $1.75 million from NIH

A new four-year NIH grant will enable Zhe Han, Ph.D., to carry out the latest stage in the detective work to determine how histone-modifying genes regulate heart development and the molecular mechanisms of congenital heart disease caused by these genetic mutations.
The National Institutes of Health (NIH) has awarded $1.75 million to a research lab led by Zhe Han, Ph.D., principal investigator and associate professor in the Center for Genetic Medicine Research, in order to build models of congenital heart disease (CHD) that are tailored to the unique genetic sequences of individual patients.
Han was the first researcher to create a Drosophila melanogaster model to efficiently study genes involved in CHD, the No.1 birth defect experienced by newborns, based on sequencing data from patients with the heart condition. While surgery can fix more than 90 percent of such heart defects, an ongoing challenge is how to contend with the remaining cases since mutations of a vast array of genes could trigger any individual CHD case.
In a landmark paper published in 2013 in the journal Nature, five different institutions sequenced the genomes of more than 300 patients with CHD and their families, identifying 200 mutated genes of interest.
“Even though mutations of these genes were identified from patients with CHD, these genes cannot be called ‘CHD genes’ since we had no in vivo evidence to demonstrate these genes are involved in heart development,” Han says. “A key question to be answered: How do we efficiently test a large number of candidate disease genes in an experimental model system?”
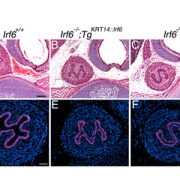
In early 2017, Han published a paper in Elife providing the answer to that lingering question. By silencing genes in a fly model of human CHD, the research team confirmed which genes play important roles in development. The largest group of genes that were validated in Han’s study were histone-modifying genes. (DNA winds around the histone protein, like thread wrapped around a spool, to become packed into a higher-level structure.)
The new four-year NIH grant will enable Han to carry out the next stage of the detective work to determine precisely how histone-modifying genes regulate heart development. In order to do so, his group will silence the function of histone-modifying genes one by one, to study their function in the fly heart development and to identify the key histone-modifying genes for heart development. And because patients with CHD can have more than one mutated gene, he will silence multiple genes simultaneously to determine how those genes work in partnership to cause heart development to go awry.
By the end of the four-year research project, Han hopes to be able to identify all of the histone-modified genes that play pivotal roles in development of the heart in order to use those genes to tailor make personalized fly models corresponding to individual patient’s genetic makeup.
Parents with mutations linked to CHD are likely to pass heart disease risk to the next generation. One day, those parents could have an opportunity to sequence their genes to learn the degree of CHD risk their offspring face.
“Funding this type of basic research enables us to understand which genes are important for heart development and how. With this knowledge, in the near future we could predict the chances of a baby being born with CHD, and cure it by using gene-editing approaches to prevent passing disease to the next generation,” Han says.