Life-changing care: How Children’s National tackles pediatric cervical spine injuries
The Cervical Spine program at Children’s National Hospital is responsible for treating a range of conditions, including trauma, congenital abnormalities and tumors. These conditions can lead to instability or misalignment of the cervical spine. Some of the common symptoms of cervical spine injuries are motor and sensory deficits, incontinence and headaches. These symptoms can indicate underlying issues that need to be addressed promptly.
“There are unique challenges in pediatric cases due to anatomical differences. The cervical spines of children are more at risk for injury because of their developmental stage and structural characteristics,” says Matthew Oetgen, MD, MBA, chief of Orthopaedic Surgery and Sports Medicine at Children’s National.
The patient benefits
Children’s National offers a range of treatment options for cervical spine injuries, ranging from nonoperative care to surgical interventions. Nonoperative care options include immobilization with cervical collars or halos. These methods help stabilize the spine without surgery. Surgical interventions such as fixation and fusion are used to maintain alignment and decrease instability and are necessary for more severe cases.
“There are unique challenges in pediatric cases due to anatomical differences. The cervical spines of children are more at risk for injury because of their developmental stage and structural characteristics,” says Dr. Oetgen.
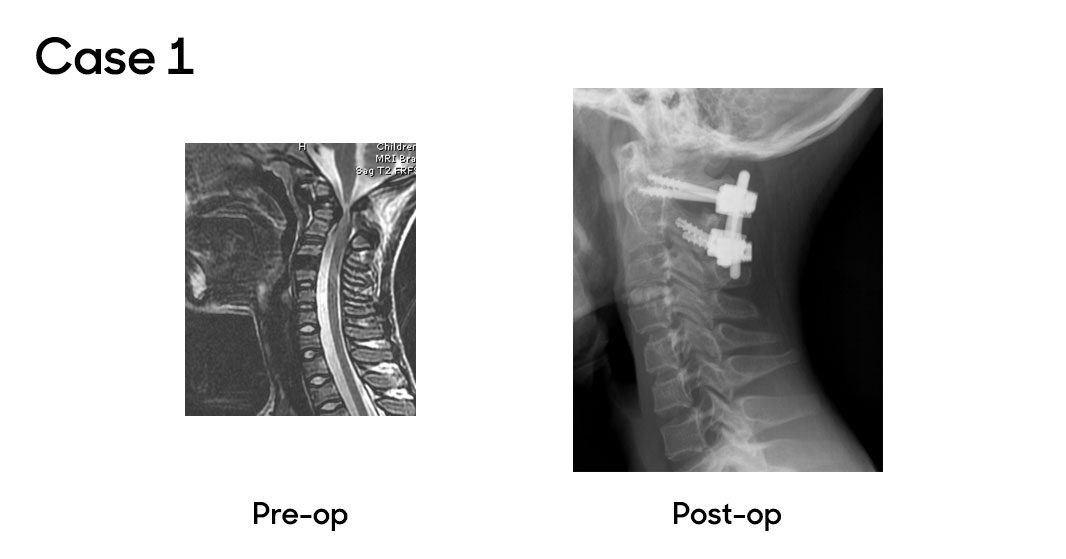
Case 1
In a recent case, a patient was referred to the orthopaedic clinic for a small scoliosis. Benjamin Martin, MD, associate chief of Clinical Operations and Affairs, reviewed the patient’s X-rays and found the spinal deformity minor and not concerning. However, he identified neurological signs during the patient’s physical exam, including mild weakness, unsteady gait, hyperreflexia and positive Babinski and clonus tests. An MRI revealed unstable os odontoideum with upper cervical stenosis and spinal cord compression causing myelomalacia. The patient was referred to Dr. Oetgen and John Myseros, MD, associate chief of Neurosurgery, who confirmed the diagnosis. The patient underwent a C1 decompression and C1-2 fusion using computer-aided navigation. The patient recovered well, achieving C1-2 fusion, improved neurological function and made a full recovery.
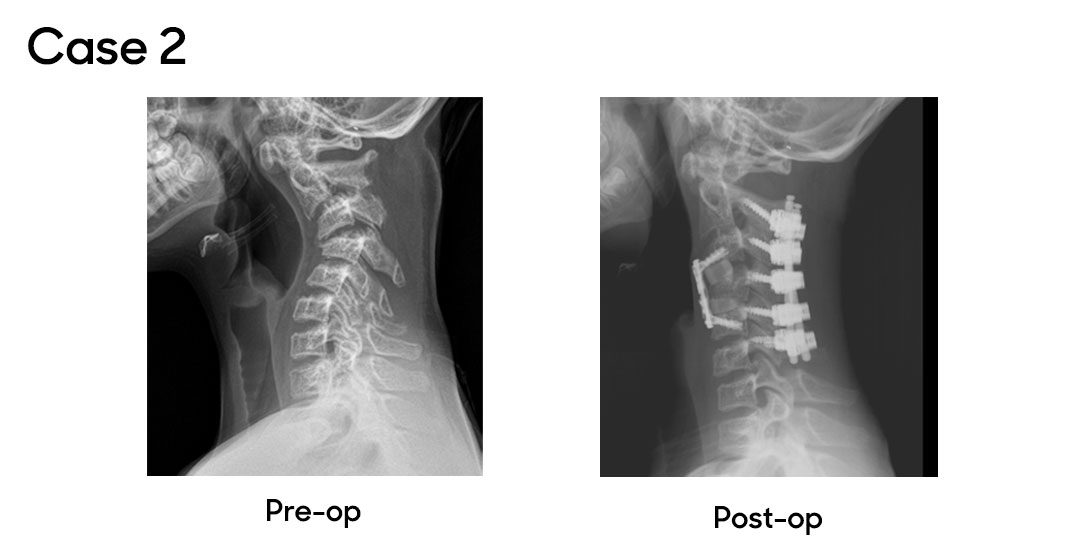
Case 2
In another case, a patient with progressive cervical kyphosis was referred to Dr. Oetgen. Diagnosed with medulloblastoma in 2015, the patient had undergone multiple cervical spinal surgeries elsewhere, resulting in post-laminectomy kyphosis causing pain, fatigue and restricted activity. This complex case was managed collaboratively by Drs. Myseros and Oetgen. The patient first underwent halo gravity traction to address the contracted deformity, followed by a combination surgery: anterior discectomy and fusion at C3-4 and C4-5, and posterior cervical fusion from C2-C6. The patient recovered well, showing significant improvement in alignment, comfort and activity levels.
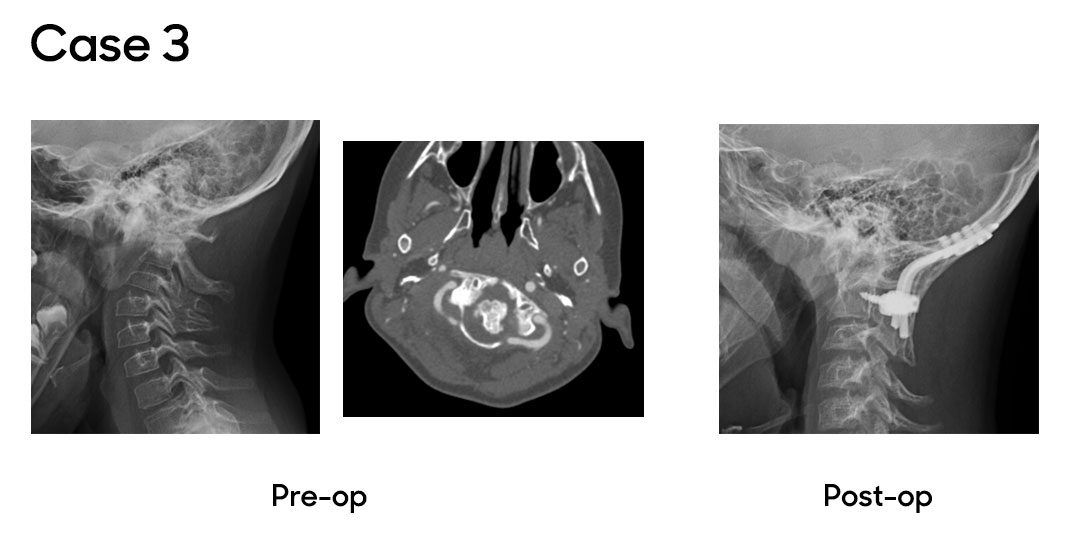
Case 3
In the final case, a patient with Lowe Syndrome was referred after four months of neck pain, stiffness and a rotational deformity of the head, limiting motion and causing discomfort. An MRI and CT scan revealed chronic atlanto-axial rotatory instability, a rare deformity in children. Due to the severity and duration of the patient’s symptoms, Dr. Oetgen and the neurosurgical team recommended an occiput-to-C2 posterior decompression, realignment and fusion. The procedure was successful, and the patient has since returned to full activity with normal alignment and no symptoms.
Key takeaway
The importance of early intervention in cervical spine injuries is key to preventing long-term neurological deficits. It’s important to stay current on screenings and seek timely treatment to maintain neurological function.
Read more about our advances in Orthopaedic Surgery and Sports Medicine.