Researchers publish first-ever atlas of cancerous mutations in histones
Leading genetic researchers at Children’s National Hospital have published the first pan-cancer atlas of key mutations that can drive molecular changes leading to tumors, creating a roadmap that could lead to new treatments for brain tumors and other cancers.
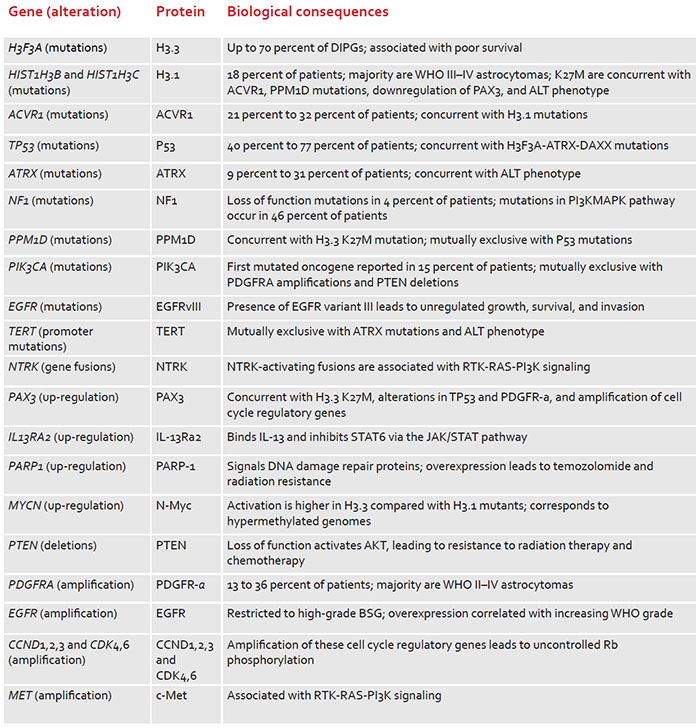
The research – published in npj Genomic Medicine – presents the first-of-its-kind atlas of histone mutations across pediatric, adolescent/young adult and adult cancers. The novel genetic work offers a framework allowing specific cancers to be redefined in the context of changes in histones, which are essential proteins that provide the structural support for chromosomes.
The big picture
“One of the major challenges that we face every day with pediatric, aggressive tumors, including pediatric high-grade gliomas, is that these tumors grow fast. Doctors often have to give patients 9 to 12 months from diagnosis,” said Javed Nazarian, Ph.D., scientific director of the Brain Tumor Institute at Children’s National and principal investigator at the Center for Genetic Medicine Research. “Children’s National has put together a team of clinicians that are truly devoted to finding a therapy for pediatric high-grade gliomas and aggressive pediatric brain tumors. Our dedicated team empowers translational research, from bench to bedside and reverse translation.”
In 2023, the American Cancer Society estimates that 9,910 children under age 15 will be diagnosed with cancer, making it the second leading cause of death among children. Because of treatment advances, 85% will survive, but many will be left with lifelong disabilities from their treatment. Nazarian and his team believe that identifying the underlying molecular alterations leading to cancers will be essential to finding new therapies that extend life expectancies and preserve quality of life.
The fine print
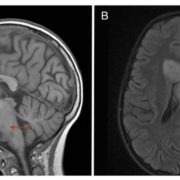
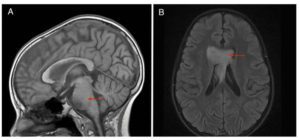
Histones are essential cellular structures, which prevent DNA from getting tangled. Nazarian and other researchers are investigating whether errors in histones could lead to cancers, including high-grade gliomas and other particularly sinister tumors that can strike young children. By mapping the mutations of the histone-encoding genes, Nazarian and his team believe they can find the drivers of tumors in many pediatric and adult cancers. In studying more than 12,000 tumors for the pan-cancer atlas, they cataloged patient ages, survival outcomes and tumor locations to reveal important trends among different cancers.
Overall, the team found that 11% of tumors had somatic histone mutations, with the highest rates observed among chondrosarcoma, a type of bone cancer (67%); pediatric high-grade glioma, a type of cancer that attacks glial cells in the brain and spinal cord (>60%); and lymphoma, a category of cancers in the lymph system (>30%).
“I think one implication of our study is that we are looking at the epigenomic changes of these mutations in a new light,” Nazarian said. “These mutations are not just specific to a particular tumor type, but they are indeed across a large spectrum of cancer types, and they come in different flavors that could potentially show a new avenue for treatments.”