Adjusting key protein could improve brain function in children with fetal alcohol syndrome
Researchers at Children’s National are testing whether controlling the level of apolipoprotein E could serve as an effective treatment for the poor neurobehavioral outcomes tied to fetal alcohol spectrum disorders.
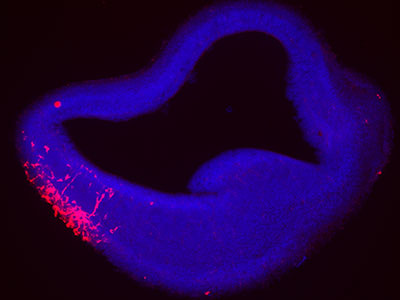
Reduced levels of a protein – called apolipoprotein E – are responsible for the lifelong cognitive and neurological symptoms in fetal alcohol spectrum disorders (FASD), according to a new study published in the journal Molecular Psychiatry. For the first time, researchers at Children’s National Hospital are testing whether controlling the level of this protein could serve as an effective treatment for the poor neurobehavioral outcomes tied to FASD, which is believed to affect roughly 5% of school-aged children.
Apolipoprotein E is controlled by a gene called APOE, a well-known risk gene for Alzheimer’s disease. It contains the instructions that guide how the connections between neurons in the brain strengthen or weaken over time. This process, known as synaptic plasticity, is crucial for learning and memory formation. In this study, plasma was collected from participants at two sites in Western Ukraine, along with information about drinking behavior reported by their mothers.
What this means
“In addition to Alzheimer’s risk, we found this gene is also a crucial contributor to cognitive problems in children with FASD,” said Kazue Hashimoto-Torii, Ph.D., a principal investigator at the Center for Neuroscience Research at Children’s National. “The new data shows that we understand the mechanism by which prenatal alcohol exposure causes a decrease of the APOE level in the brain. We will continue this work to help improve our understanding of FASD, hoping to replenish this important protein and improve outcomes for children with these disorders.”
FASD is an umbrella diagnosis for the physical and developmental challenges that face children who are exposed to alcohol in utero, including intellectual disability, delay in motor and language development and other neurological diagnoses. While alcohol alone is problematic to many aspects of development, evidence also suggests that genetic factors play a role. Only 4.3% of children with heavy alcohol exposure develop FASD, and twin studies have revealed that fraternal twins have different FASD outcomes compared to identical twins.
What’s next
The research team at Children’s National wanted to pinpoint places where genes could play a role and consider therapies. For the first time, researchers found that plasma levels of APOE were reduced in children with FASD, which strongly supports a potential target for therapy. In addition, their genomics study found a variant of APOE increases the risk of cognitive problems in subjects who were prenatally exposed to alcohol, especially those of African ancestry.
“We found that providing a drug that activates the APOE receptor rescued learning deficits and anxiety in pre-clinical models,” said Masaaki Torii, Ph.D., principal investigator at the Center for Neuroscience Research. “The implications offer an exciting glimpse into possible therapies for some of the neurological harms associated with prenatal alcohol exposure and FASD.”
Read the full manuscript – Reduction of APOE accounts for neurobehavioral deficits in fetal alcohol spectrum disorders – in Molecular Psychiatry.