Long-term outcomes are key measure of CHD care quality and safety
Short-term outcomes data may be one benchmark of the quality and safety found in a pediatric cardiac surgery program, but according to the team at Children’s National Hospital, it shouldn’t be the sole factor in how referring physicians and families make decisions about where to seek care.
Instead, physicians and families need more information about what to expect for a child’s entire lifespan. To meet this need, the Division of Cardiac Surgery at Children’s National has launched the Children’s National Cardiac Outcomes Registry (CNCOR), a first-of-its-kind database that captures outcomes from 15-plus years of congenital heart surgical repairs for specific congenital heart conditions.
What it means
Today, most pediatric heart programs publicly share their outcomes based on benchmarks defined by the Society for Thoracic Surgeons (STS). For cardiac surgery, the data points are focused on the short-term outcomes for various procedures, ranging from 30 days to one year after surgery.
However, as surgical procedures have been refined and multi-disciplinary teams continually improve their knowledge of how to treat children with these congenital heart defects, more children with these conditions are living far beyond those time frames. One metric, operative mortality for “index cases” defined by the STS (excluding VAD and ECMO), measures how many children having heart surgery with the cardiopulmonary bypass die during surgery or within the 30 days after surgery.
Recently at Children’s National, the Cardiac Surgery, Cardiac Intensive Care Unit, Cardiology and Cardiac Anesthesia teams had a remarkable year with zero operative mortality, or no deaths, for the types of cases included in this benchmark. While it’s impossible to expect zero mortality to continue indefinitely when caring for children with the most severe and complex heart conditions — as is the case at Children’s National — the national average operative mortality for these procedures is closer to 3%.
Yves d’Udekem, M.D., Ph.D., chief of Cardiac Surgery at Children’s National, says that this achievement is not one he expected to “ever see in his lifetime” and is a testament to the teams’ dedication to providing the highest quality care every day. However, he also noted, “Achieving such impressive statistics for short-term measures has never been, is not, and will never be my objective. We also need to look beyond it. What happens to this patient 5, 10 or 15 years down the line? That’s what we want to know.”
Children’s National leads the way
The CNCOR collects long-term data from 20 years of cardiac surgical procedures at Children’s National. This data has allowed Children’s National to create a series of charts that show predicted long-term outcomes for specific congenital heart conditions.
The charts are now available in a new Cardiac Surgery Long-Term Outcomes section on the website, organized by specific types of congenital heart disease, including:
- Tetralogy of Fallot
- Aortic arch coarctation
- Atrioventricular septal defects
- Transposition of the great arteries
For most conditions, data is available showing long-term mortality as well as re-operation or re-intervention rates for children who underwent these procedures at Children’s National, going as far back as the year 2000. Eventually, the database might expand to include indicators such as exercise capacity and neurological outcomes.
What’s next
The launch of the CNCOR is the start of efforts by the team at Children’s National to provide what Dr. d’Udekem calls “proper” benchmarking based on the whole life of a patient rather than a single event in time.
If more institutions collect and report this data and make it available, patients, families and practitioners who refer to cardiac surgery will have a true lifetime perspective of what a surgical procedure at a specific institution may mean for a child.
























 Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.



 A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
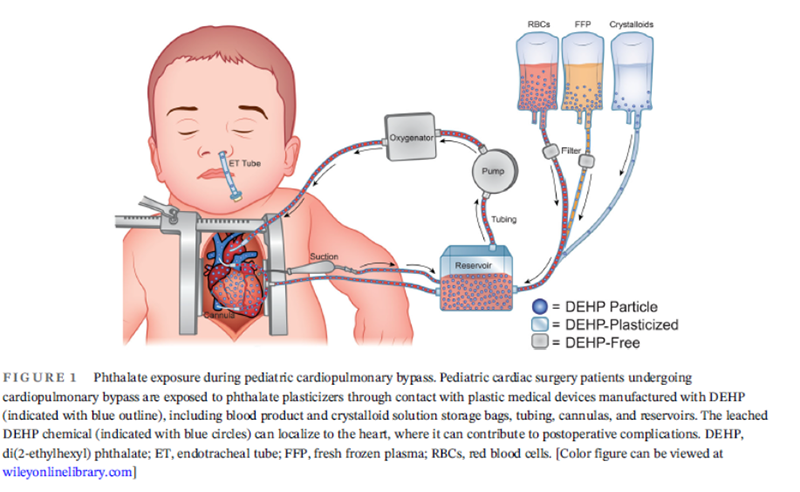
 A new study published in
A new study published in 

 Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its
Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its 



