What’s Known
According to the Centers for Disease Control and Prevention, Zika viral transmission is occurring extensively throughout Central and South America. Like other mosquito-borne viruses, Zika virus can be passed by pregnant women to developing fetuses. Unlike these other viruses, Zika has been implicated in a growing number of cases of Brazilian infants born with microcephaly, a condition characterized by undersized heads and severe brain damage. The precise strategy that the Zika virus uses to elude the immune system and the reason why fetal brain cells are particularly vulnerable remain unknown.
What’s New
A 33-year-old Finnish woman was 11 weeks pregnant when she and her husband traveled on vacation to Mexico, Guatemala, and Belize in late November 2015. The pair was bitten by mosquitoes during their trip, particularly in Guatemala. One day after returning to their Washington, DC home, the woman got sick, experiencing eye pain, muscle pain, a mild fever, and a rash. A series of early ultrasounds showed no sign of microcephaly or brain calcifications. A fetal ultrasound at the 19th week and a fetal MRI at the 20th week, however, revealed severe brain damage.
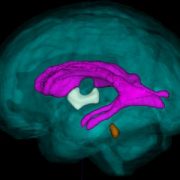
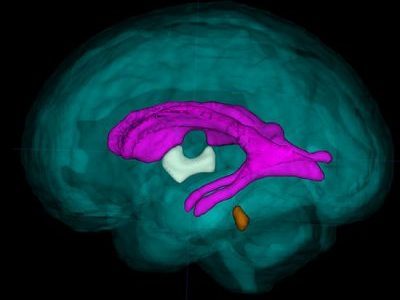
The brain of the 21-week-old aborted fetus weighed only 30 grams. Zika RNA, viral particles, and infectious virus were detected, and Zika virus isolated from the fetal brain remained infectious when tested. The concentration of virus was highest in the fetal brain, umbilical cord, and placenta. The mother remained infected with Zika virus at 21 weeks, some 10 weeks after her initial infection.
Questions for Future Research
- Could serial measurements and blood tests more accurately detect and, ultimately, predict fetal abnormalities following Zika virus infection?
- Why does the Zika virus replicate with ease within the womb?
- At which stage of pregnancy are fetuses most vulnerable?
- Which specific brain cells does Zika target?
Source: “Zika Virus Infection with Prolonged Maternal Viremia and Fetal Brain Abnormalities.” R.W. Driggers, C.Y. Ho, E.M. Korhonen, S. Kuivanen, A.J. Jääskeläinen, T. Smura, D.A. Hill, R. DeBiasi, G. Vezina, J. Timofeev, F.J. Rodriguez, L. Levanov, J. Razak, P. Iyengar, A. Hennenfent, R. Kennedy, R. Lanciotti, A. du Plessis, and O. Vapalahti. The New England Journal of Medicine. June 2, 2016.