Novel ultrasound device gets FDA breakthrough designation with Children’s National support
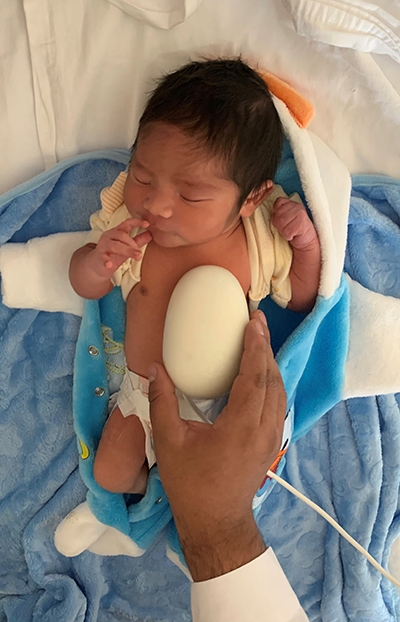
The Bloom Standard device enables autonomous, hands-free ultrasound scans to be performed anywhere, by any user.
A novel ultrasound device developed by Bloom Standard received the Food and Drug Administration’s valued breakthrough device designation with the help of Children’s National Hospital and support provided through an FDA-funded grant that established the Alliance for Pediatric Device Innovation (APDI), formerly branded as the National Capital Consortium for Pediatric Device Innovation (NCC-PDI). The grant funding, clinical expertise and regulatory guidance demonstrate the hospital’s leadership in pediatric medical device innovation and its commitment to supporting critically needed advancements.
“In many ways, Children’s National has been a key resource as we continue this journey from concept to commercialization,” said Annamarie Saarinen, co-founder of Bloom Standard and the mother of an infant who was diagnosed with a critical heart defect days after her birth.
Why we’re excited
Children’s National Innovation Ventures and APDI leaders saw the value in the Bloom Standard innovation: a device that enables autonomous, hands-free ultrasound scans to be performed anywhere, by any user. The FDA granted the innovation its breakthrough device designation to help streamline the regulatory process so that patients and healthcare providers have more timely access to devices.
Bloom Standard can potentially save lives and improve outcomes for newborns and babies with cardiac and respiratory issues. The technology can eliminate the delay that often occurs for infants who need ultrasound imaging because their medical location lacks the technology. Bloom Standard’s portability and ease of use can potentially reduce mortality and poor outcomes tied to delayed detection of lung and cardiac conditions. It has potential cost savings, too.
Children’s National leads the way
Children’s National supported this novel medical technology for children by engaging in multiple steps through the regulatory process:
- Grant funding: Bloom Standard participated in pediatric pitch competitions in 2020 and 2022 and each time, because of the merits of the device, was awarded grant funding that helped to support its progression.
- Regulatory support: Children’s National pediatrician Francesca Joseph, M.D., brings a wealth of expertise navigating the FDA’s regulatory processes and currently service as co-investigator and core regulatory expert for the pediatric device consortium led by Children’s National. Saarinen called Dr. Joseph “an invaluable resource,” explaining that her input and support has been very significant in Bloom Standard’s understanding and navigation of FDA processes. “For a small device company like ours, she has been a lifeline,” Saarinen said.
Saarinen says she is grateful for the continuing relationship with Children’s National as her company enters its next phase on the journey to commercialization.
Children’s National Vice President and Chief Innovation Officer Kolaleh Eskandanian, Ph.D., MBA, PMP, who directs the FDA-funded Alliance for Pediatric Device Development, says that nurturing and supporting life-saving technologies like Bloom Standard is important because the device addresses a dire unmet need in the pediatric space, especially in settings with fewer resources and in public health emergency situations. “Advancements in children’s medical devices continue to lag behind those of adults, and we must use our research and innovation infrastructure to help close that gap and to influence policy changes.”