Medical device competition announces six winners to share in $250K
October 2, 2020

“Congratulations to all ten companies who competed in this year’s finals for developing pioneering innovations that can provide much-needed medical device advancements for children,” says Kolaleh Eskandanian, Ph.D., MBA, PMP, vice president and chief innovation officer at Children’s National Hospital and principal investigator of NCC-PDI.
The National Capital Consortium for Pediatric Device Innovation (NCC-PDI) announces six winners chosen in the annual “Make Your Medical Device Pitch for Kids!” competition. Focused on innovations in cardiovascular, NICU, and orthopaedic and spine devices, areas of critical need where innovation can significantly improve children’s healthcare, ten finalists competed on Wednesday, September 30 in a virtual pitch event for a share of $250,000 grants funded by the Food and Drug Administration (FDA).
A panel of judges with clinical, business, and regulatory expertise selected the winners based on the potential patient benefit and commercial viability of these pediatric devices. n the last decade, only 24% of Class III life-saving devices approved by FDA were for pediatric use – and most of those were for children over 12. Less than 4% were labeled for pediatric patients ages 0-2 years old and the number of approved devices is even lower for neonatal patients.
This year’s pediatric device innovation awardees are:
- Adipomics, Inc. – Cambridge, MA – COVI-SPOT, a one-step COVID-19 diagnosis on the spot;
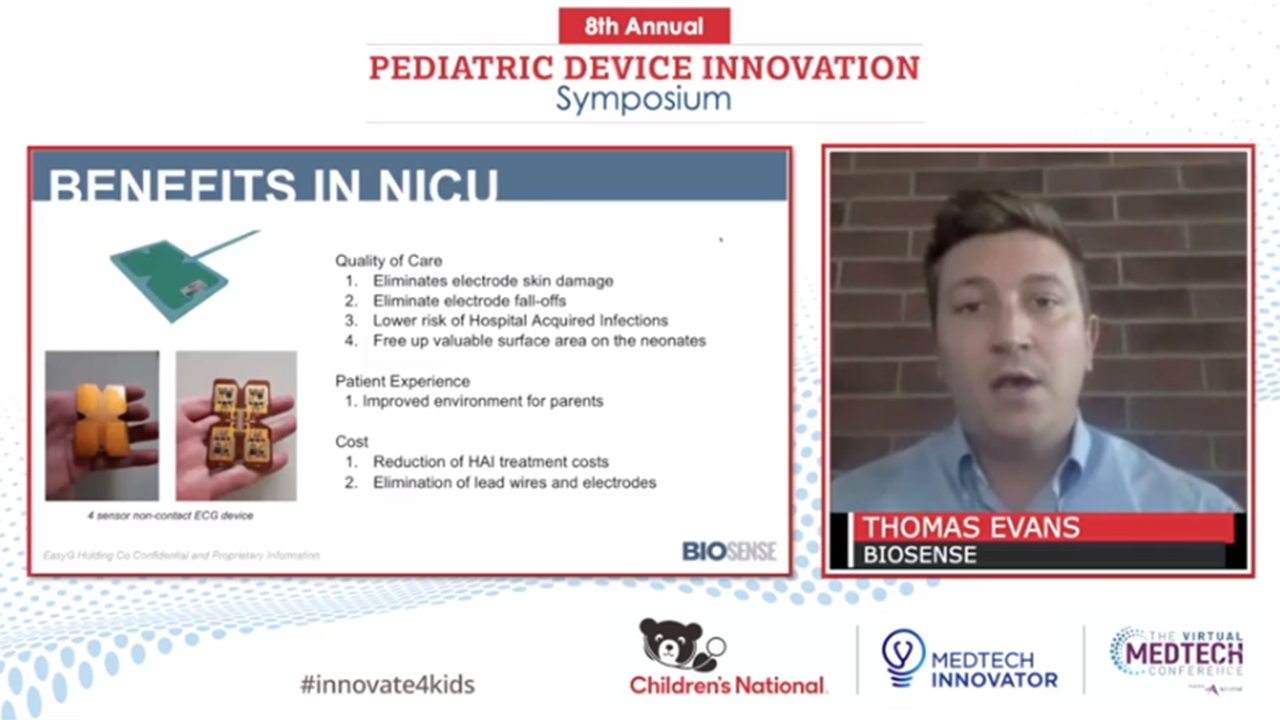
- BioSense – Beverly Hills, MI – the first working non-contact EKG for constant, non-invasive monitoring;
- Innara Health – Olathe, KS – a biofeedback device to help newborns and premature infants improve feeding readiness and promote the neuro and physiological aspects of feeding;
- Navi Medical Technologies – Melbourne, Australia – a non-invasive device to provide real-time feedback of central line tip location using ECG;
- Novonate – South San Francisco, CA – a device that secures and protects the umbilical catheter insertion site for neonates in intensive care;
- Renata Medical – Costa Mesa, CA – a growth stent for treatment of congenital narrowed lesions in neonatal patients.
“Congratulations to all ten companies who competed in this year’s finals for developing pioneering innovations that can provide much-needed medical device advancements for children,” says Kolaleh Eskandanian, Ph.D., MBA, PMP, vice president and chief innovation officer at Children’s National Hospital and principal investigator of NCC-PDI. “We thank the FDA for making these awards possible. Although more incentives for pediatric device development are needed, it is encouraging to witness the progress we have made since the inception of the PDC program.”

“For far too long, pediatric specialists have been manipulating adult medical devices to create solutions for children’s bodies because it’s the only available option,” said Kurt Newman, M.D., president and CEO of Children’s National Hospital. “Children need and deserve devices that are conceived and designed with their biology and future in mind.”
The ten pediatric device finalists were selected from a field of 30 semi-finalists who competed in March 2020. Leading up to the September 2020 pitch competition, the finalists participated in a unique pediatric accelerator track powered by MedTech Innovator, one of the industry’s leading nonprofit startup accelerators, and supported by the pediatric expertise of Children’s National Hospital.
NCC-PDI is one of five consortia in the FDA’s Pediatric Device Consortia Grant Program created to support the development and commercialization of medical devices for children and is led by the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Hospital and the A. James Clark School of Engineering at the University of Maryland, with support from partners MedTech Innovator, BioHealth Innovation and design firm Archimedic.
“For far too long, pediatric specialists have been manipulating adult medical devices to create solutions for children’s bodies because it’s the only available option,” said Kurt Newman, M.D., president and CEO of Children’s National Hospital. “Children need and deserve devices that are conceived and designed with their biology and future in mind. I’m proud that the annual NCC-PDI symposium and pitch competition is spurring pediatric device innovation. The companies that were highlighted this year are creating solutions that will help children to lead healthier lives and grow up stronger.”
The pitch competition took place as part of the 8th Annual Pediatric Device Innovation Symposium presented by Children’s National Hospital as part of The MedTech Conference by AdvaMed.
To watch the complete pitch competition, click here.