Tag Archive for: National Institutes of Health
NIH awards $1.8 million to trial pacemaker delivery system for children
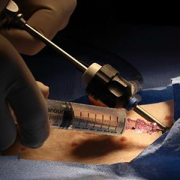
The PeriPath access port makes it possible for pacing and defibrillating leads to be placed in the smallest children through holes the size of a straw.
A $1.8 million Small Business Innovation Research (SBIR) grant from the National Institutes of Health (NIH) is funding the first clinical trial of a novel device called PeriPath. The device makes it possible for pacing and defibrillating leads (or wires) to be placed in the smallest children through holes the size of a straw, eliminating thoracotomy or sternotomy procedures for children who are too small for transvenous implantation.
Even the tiniest pacemakers and defibrillators on the market today aren’t small enough for infants and young children with heart rhythm abnormalities. Innovating smaller devices, including adapting current technology like the Medtronic Micra for pediatric use, is a good start but won’t be enough to eliminate some of the challenges for these patients. When a newborn or young child needs any pacemaker or defibrillator, they face open chest surgery. Their arteries and veins are just too small for even the smallest size transvenous pacemaker catheter.
The research goal
Charles Berul, M.D., division chief of Cardiology and co-director of the Children’s National Heart Institute, partnered with engineers in the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Hospital to develop and test a first-of-its-kind minimally invasive pericardial access tool. The tool allows doctors to place pacing and defibrillation leads to the epicardial surface of the heart under direct visualization from an endoscope.
The team hypothesizes that this tool will allow for pacing and defibrillation therapy to be delivered through a single small port inserted through the skin that is about the size of a drinking straw.
Why it matters: Less pain, shorter and fewer surgeries
If successful, the device will eliminate the need for open chest surgery in patients who aren’t candidates for transvenous placement. The ability to place these leads percutaneously should:
- Reduce pain and infection risk.
- Decrease procedure times.
- Minimize surgery complications that arise from open surgery.
- Improve better visualization for pericardial punctures.
- Allow other novel therapies such as epicardial ablation or, in the future, even drug/gene delivery into the pericardial space.
Any implanted pacemaker or defibrillator must be replaced every 5-10 years. A young child in critical need of such devices could face surgeries 10 or more times to replace the device and/or leads.
Pre-clinical testing shows early data that this percutaneous approach is as safe and effective as an open surgical technique, although it remains in early-stage evaluation.
What’s next
The NIH SBIR funding will allow the research team to assess long-term safety and efficacy and commercialize the PeriPath tool. Next steps are to:
- Refine the design of PeriPath for production manufacturing, integrate testing protocols into a Quality Management System and conduct a pilot verification build. Success is defined as manufacturing production devices that pass 510(k) verification and validation testing.
- Demonstrate substantial equivalence to predicate trocars through performance and handling validation testing using PeriPath to implant an epicardial lead in a pediatric simulator. If successful, the team will demonstrate equivalence and obtain investigational device exception (IDE).
- In the latter part of the plan, to perform a first in human feasibility clinical study using PeriPath to implant a commercial pacemaker lead with institutional review board (IRB) approval in infants at Children’s National.
Bottom line
Dr. Berul says, “This research could have a transformative impact on current clinical practice by converting an open surgical approach to a minimally invasive percutaneous procedure.”
He also notes that while the study design focuses on the unique needs of infants and children with congenital heart disease – who are the primary focus of the device – the results of the trial may benefit thousands of adult patients who need pacemakers or defibrillators but who are not candidates for the transvenous placement.
Francis S. Collins, M.D., Ph.D. from NIH: The future of genomic medicine and research funding opportunities

Genomic medicine, diversity, equity and inclusion (DEI), a world post-COVID-19 and pediatric research funding were among the topics discussed during the “Special Fireside Chat” keynote lecture at the 2021 Children’s National Hospital Research, Education and Innovation Week.
Francis S. Collins, M.D., Ph.D., director at the National Institutes of Health (NIH), is well known for his landmark discoveries of disease genes and his leadership of the international Human Genome Project, which culminated in April 2003 with the completion of a finished sequence of the human DNA instruction book.
The President and CEO of Children’s National, Kurt Newman, M.D., joined Dr. Collins during the “Special Fireside Chat” keynote lecture. Dr. Newman posed several health care-related questions to Dr. Collins over the course of 30 minutes. Dr. Collins’s responses shed light on what it takes to advance various research fields focused on improving child health and develop frameworks that advocate for DEI in order to foster a more just society.
Q: You have been involved with genomic medicine since its inception. You discovered the gene causing cystic fibrosis and led the Human Genome project. What do you see as the future of genomic medicine, especially as it relates to improving child health?
A: Thank you for the question, Kurt. First, I wanted to say congratulations on your 150th anniversary. Children’s National Hospital has been such a critical component for pediatric research and care in the Washington, D.C., area, and at the national and international levels. We at the NIH consider it a great privilege to be your partner in many of the things that we can and are doing together.
Genomic medicine has certainly come a long way. The word genomics was invented in 1980, so we have not been at this for that long. Yet, the success of the Human Genome Project and the access to cost-effective tools for rapid DNA sequencing have made many things possible. It took a lot of effort, time and money to discover the gene that causes cystic fibrosis. Kurt, if you look at what we did, while it was rewarding, it was a challenging problem that occupied the hearts of the scientific community in 1980. Now, a graduate student at Children’s National that has access to DNA samples, a thermal cycler, a DNA sequencer and the internet could do in about a week what it took us a decade and with 50 people.
We have been able to rocket forward as far as identifying the genetic causes of 6,500 diseases, where we know precisely the molecular glitch responsible for those conditions. While most of those are rare diseases, it leads to the opportunity for immediate diagnosis, which used to be a long and troubled journey.
DNA sequencing has increasingly become an essential tool in newborns, especially when trying to sort out puzzling diagnosis for specific syndromes or phenotypes that are not immediately clear. Additionally, DNA sequencing significantly impacted clinical care in cancer because it made it possible to look at the mutations driving the malignancy and its genetic information that can lead to interventions. This approach is going forward in the next few years in ways that we can see now. Although I am a little reluctant to make predictions because I have to be careful about that, it may be possible to obtain complete genome sequences that can be yours for life and place them into the medical record to make predictions about future risks and choices about appropriate drugs. This path costs less than any imaging tests.
Q: The racial justice movement that was brought back to the forefront this past year has, once again, reaffirmed that this country has so much more work to do in order to end systemic racism. You have been at the forefront of promoting diversity, equity and inclusion in research and at the NIH. What do you and the NIH plan to do further DEI efforts in research and in general so that we can be a more just and equitable society?
A: I appreciate you raising this, Kurt. Diversity, equity and inclusion (DEI) is an issue where everyone should be spending a lot of time, energy and passion. You are right. 2020 will be remembered for COVID-19. I also think it will be remembered for the things that occurred around the killing of George Floyd, and the recognition of the very foundation that is still infected by this terribly difficult circumstance of structural racism. I convened a group of about 75 deep thinkers about these issues, many of them are people of color from across the NIH’s different areas of activities. I asked the group to come forward with a bold set of proposals. This effort is how the program UNITE came together to work hard on this, which is now making recommendations that I intend to follow. We are determined to close that gap and pursue additional programs that will allow us to be more successful in recruiting and retaining minority groups, for example. We need to do something with our health disparity and research portfolio as well to ensure that we are not just looking around the edges of the causes for racial inequities. We are digging deeper into what the structural racism underpinnings are and what we can do about it. I am particularly interested in supporting research projects that test intervention and not just catalog the factors involved. We have been, at times, accused and maybe rightly so of being more academic about this, and, less kindly, we have been accused of admiring the problem of health disparities as opposed to acting on it. We are ready to act.
Q: COVID has affected us all in so many ways. Could you tell us what this past year has been like for you? Also, how is the NIH preparing for a soon-to-be post-COVID pandemic?
A: This is the time to contemplate the lessons learned as everyone knows that the last worst pandemic happened over a century ago. One thing that maybe will vex us going forward, which we already started to invest in a big way, is this whole long COVID syndrome, also referred to post-acute sequelae, to understand precisely the consequences and mechanisms like Multisystem Inflammatory Syndrome in Children (MIS-C). Before moving to the next pandemic, we must think about how we will help understand those who suffer from long COVID syndrome. As far as the broader lessons learn, Kurt, we must expect that there will be other pandemics because humans are interacting more with animals, so zoonosis is likely to emerge. We need to have a clear sense of preparation for the next one. For instance, we are working on this right now, but we need to have a stronger effort to develop small molecules of anti-viral drugs aimed at the major viral classes, so we do not have to start from scratch. We also need clinical trial networks warm all the time, ready to go and to learn how valuable public partnerships can be to get things done in a hurry.
Editor’s Note: The responses in this Q+A have been modified to fit the word count.
NIH grant funds development of pediatric feeding tube placement device
A new grant will help to finalize development of the Pediatric PUMA-G System, the world’s first and only ultrasound-based procedure for placing feeding tubes into the stomach.
Researchers at Children’s National Hospital have received grant funding from the National Institute of Diabetes and Digestive and Kidney Diseases, within the National Institutes of Health (NIH), to finalize development of the Pediatric PUMA-G System, the world’s first and only ultrasound-based procedure for placing feeding tubes into the stomach. The funding will also support the first clinical trial of this technology in pediatric patients.
“Children’s National was chosen because we have a strong record of innovating pediatric devices and surgical procedures through the Sheikh Zayed Institute and we have a busy clinical interventional radiology service,” says Karun Sharma, M.D., Ph.D., director of Interventional Radiology and associate director of clinical translation at the Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI) at Children’s National. “We are proud to be a part of this collaboration that will potentially help improve care of pediatric patients who cannot tolerate feeding by mouth.”
The feeding tubes are vital for children who cannot eat or swallow and require liquid nutrition (known as enteral feeding). Common feeding tube placement procedures for children may expose them to risks from invasive surgical tools or from ionizing radiation, which may lead to cancer in young patients at elevated rates. The PUMA-G System is less invasive and uses ultrasound to help physicians image the body during the procedure.
The grant, totaling $1.6M, will clinically evaluate the Pediatric PUMA-G System in collaboration with CoapTech, a biotechnology medical device company and two other premier pediatric medical centers — New York-Presbyterian Morgan Stanley Children’s Hospital and Children’s Hospital of Philadelphia.
Research team develops new and improved method for studying cardiac function

While researching how plastic affects heart function in sensitive populations, such as children born with congenital heart defects, Children’s National researcher Nikki Posnack, Ph.D., led a team that developed a new and improved, replicable method of performing simultaneous dual optical mapping to examine electrical activity and calcium for the study of cardiac function.
Since arriving at the Sheikh Zayed Institute for Pediatric Surgical Innovation, researcher Nikki Gillum Posnack, Ph.D., a principal investigator with the institute and assistant professor of pediatrics at the George Washington University School of Medicine and Health Sciences, has been focused on examining how exposure to plastic affects heart function in sensitive populations, such as children born with congenital heart defects. She performs optical mapping to conduct this research, but the industry standard approaches of either using dual cameras or sequential single cameras were cost prohibitive and technically challenging while also diminishing the quality of the imaging results.
Fast forward to July 2019 when Dr. Posnack and her team published “Plasticizer Interaction With the Heart” in the journal Arrhythmia and Electrophysiology, which used imaging techniques to reveal the impact of plastic chemicals on the electrical activity of the heart. Dr. Posnack’s laboratory has since expanded this technique and revealed a new replicable method of performing simultaneous dual optical mapping to examine electrical activity and calcium handling in the heart.
Sharing a new method for studying cardiac function
This groundbreaking method is itself the focus of a new BMC Biomedical Engineering journal article titled “Lights, camera, path splitter: a new approach for truly simultaneous dual optical mapping of the heart with a single camera.”
The article compares and contrasts the current standard for dual camera simultaneous configurations and single camera sequential configurations to Dr. Posnack’s new single camera simultaneous configuration.
Simultaneous dual mapping systems use two probes and dual dyes – one for electrical voltage and the other for calcium. While dual-dye combinations like Di-4-ANEPPS with Indo-1, Di-2-ANEPEQ and calcium green have been developed to separate fluorescence signals by emission, these dye combinations can have spectral overlap, creating the need for non-ideal emission bandpass to negate spectral overlap and/or the inclusion of a calcium probe with an inferior dissociation constant. Additionally, dual-sensor systems require proper alignment to ensure that fluorescence signals are being analyzed from the same tissue region on each individual detector, which could lead to erroneous results. The dual-camera optical setup is expensive, technically challenging and requires a large physical footprint that is often not feasible for basic science and teaching laboratories conducting critical research.
As an alternative, some researchers use a single camera configuration to sequentially image the voltage and calcium probes using excitation light patterning. This approach also has limitations. These single-sensor designs use dual-dye combinations that require two or more excitation light sources, but share a single emission band. Like the dual camera system, this platform design is also technically challenging since the different excitation light wavelengths require light source triggering, camera synchronization and frame interleaving. Due to timing coordination, decreased frame rates, excitation light ramp up/down times and shutter open/close times, single system setups require shorter exposure times compared to dual sensor setups, diminishing the signal-to-noise quality without offering the same temporal fidelity. There is a cost advantage to the single camera system, however, because the additional camera is often one of the most expensive components.
This new single camera, simultaneous dual optical mapping approach is the first multiparametric mapping system that simultaneously acquires calcium and voltage signals from cardiac preparations, using a commercially available optical path splitter, single camera and single excitation light. Using a large field of view sCMOS sensor that is faster and more sensitive, this configuration separates the two emission bands for voltage and calcium probes and simultaneously directs them to either sides of the single, large camera sensor. This protocol employs a commonly used dual-dye combination (RH237 and Rhod2-AM). In contrast, other protocols may require genetically-encoded indicators or fluorescent probes that are not yet commercially available.
The team validated the utility of the approach by performing high-speed simultaneous dual imaging with sufficient signal-to-noise ratio for calcium and voltage signals and specificity of emission signals with negligible cross-talk. Demonstrating the need for simultaneous electrical and calcium sensors, they found that when ventricular tachycardia is induced, there is spatially discordant calcium alternans present in different regions of the heart even when the electrical alternans remain concordant.
Having eliminated the second camera as well as the need for multiple excitation light sources, light pattering and frame interleaving, this system is more cost effective, simpler, and can be easily setup by various types of researchers, not just those with engineering backgrounds.
With a limited research budget and a background in physiology, Dr. Posnack worked collaboratively with her post-doctoral fellow Rafael Jaimes III, an engineer in the Sheikh Zayed Institute for Pediatric Surgical Innovation, to develop a cost-effective system that would enable her to truly study the effects of plastics on the heart.
Multidisciplinary approach
“We’re fortunate to have a multidisciplinary team in the Sheikh Zayed Institute so that I could work with an engineer to develop the technology and system we needed to propel our research,” said Dr. Posnack. “There are so many researchers who have the science background, but not necessarily the technical aptitude, and they get stymied in their research, so we’re proud that this paper will help other researchers replicate the system to study cardiac function.”
The research paper was funded by a grant from the National Institutes of Health as well as support from the Children’s Research Institute, Children’s National Heart Institute and the Sheikh Zayed Institute for Pediatric Surgical Innovation.
The applications for this optical mapping system are significant and Dr. Posnack has been consulted by other research teams looking to implement it in their labs. Additionally, Dr. Posnack has collaborated with several neuroscience teams at Children’s National Hospital, including one that is investigating the effects of hypoxia on brain and heart development, and another that is interested in using image modalities and data processing to analyze calcium as an indicator of neuron firing.
Dr. Posnack continues to use this new dual optical mapping system to further her research as she anticipates the publication of a new article about age-dependent changes in cardiac electrophysiology and calcium handling.
Tailored T-cell therapies neutralize viruses that threaten kids with PID
Tailored T-cells specially designed to combat a half dozen viruses are safe and may be effective in preventing and treating multiple viral infections, according to research led by Children’s National Hospital faculty.
Catherine Bollard, M.B.Ch.B., M.D., director of the Center for Cancer and Immunology Research at Children’s National and the study’s senior author, presented the teams’ findings Nov. 8, 2019, during a second-annual symposium jointly held by Children’s National and the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH). Children’s National and NIAID formed a research partnership in 2017 to develop and conduct collaborative clinical research studies focused on young children with allergic, immunologic, infectious and inflammatory diseases. Each year, they co-host a symposium to exchange their latest research findings.
According to the NIH, more than 200 forms of primary immune deficiency diseases impact about 500,000 people in the U.S. These rare, genetic diseases so impair the person’s immune system that they experience repeated and sometimes rare infections that can be life threatening. After a hematopoietic stem cell transplantation, brand new stem cells can rebuild the person’s missing or impaired immune system. However, during the window in which the immune system rebuilds, patients can be vulnerable to a host of viral infections.
Because viral infections can be controlled by T-cells, the body’s infection-fighting white blood cells, the Children’s National first-in-humans Phase 1 dose escalation trial aimed to determine the safety of T-cells with antiviral activity against a half dozen opportunistic viruses: adenovirus, BK virus, cytomegalovirus (CMV), Epstein-Barr virus (EBV), Human Herpesvirus 6 and human parainfluenza-3 (HPIV3).
Eight patients received the hexa-valent, virus-specific T-cells after their stem cell transplants:
- Three patients were treated for active CMV, and the T-cells resolved their viremia.
- Two patients treated for active BK virus had complete symptom resolution, while one had hemorrhagic cystitis resolved but had fluctuating viral loads in their blood and urine.
- Of two patients treated prophylactically, one developed EBV viremia that was treated with rituximab.
Two additional patients received the T-cell treatments under expanded access for emergency treatment, one for disseminated adenoviremia and the other for HPIV3 pneumonia. While these critically ill patients had partial clinical improvement, they were being treated with steroids which may have dampened their antiviral responses.
“These preliminary results show that hexaviral-specific, virus-specific T-cells are safe and may be effective in preventing and treating multiple viral infections,” says Michael Keller, M.D., a pediatric immunologist at Children’s National and the lead study author. “Of note, enzyme-linked immune absorbent spot assays showed evidence of antiviral T-cell activity by three months post infusion in three of four patients who could be evaluated and expansion was detectable in two patients.”
In addition to Drs. Bollard and Keller, additional study authors include Katherine Harris M.D.; Patrick J. Hanley Ph.D., assistant research professor in the Center for Cancer and Immunology; Allistair Abraham, M.D., a blood and marrow transplantation specialist; Blachy J. Dávila Saldaña, M.D., Division of Blood and Marrow Transplantation; Nan Zhang Ph.D.; Gelina Sani BS; Haili Lang MS; Richard Childs M.D.; and Richard Jones M.D.
###
Children’s National-NIAID 2019 symposium presentations
“Welcome and introduction”
H. Clifford Lane, M.D., director of NIAID’s Division of Clinical Research
“Lessons and benefits from collaboration between the NIH and a free-standing children’s hospital”
Marshall L. Summar, M.D., director, Rare Disease Institute, Children’s National
“The hereditary disorders of PropionylCoA and Cobalamin Metabolism – past, present and future”
Charles P. Venditti, M.D., Ph.D., National Human Genome Research Institute Collaboration
“The road(s) to genetic precision therapeutics in pediatric neuromuscular disease: opportunities and challenges”
Carsten G. Bönnemann, M.D., National Institute of Neurological Disorders and Stroke
“Genomic diagnostics in immunologic diseases”
Helen Su, M.D., Ph.D., National Institute of Allergy and Infectious Diseases
“Update on outcomes of gene therapy clinical trials for X-SCID and X-CGD and plans for future trials”
Harry Malech, M.D., National Institute of Allergy and Infectious Diseases
“Virus-specific T-cell therapies: broadening applicability for PID patients”
Catherine Bollard, M.D., Children’s National
“Using genetic testing to guide therapeutic decisions in Primary Immune Deficiency Disease”
Vanessa Bundy, M.D., Ph.D., Children’s National
Panel discussion moderated by Lisa M. Guay-Woodford, M.D.
Drs. Su, Malech, Bollard and Bundy
Morgan Similuk, S.C.M., NIAID
Maren Chamorro, Parent Advocate
“Underlying mechanisms of pediatric food allergy: focus on B cells
Adora Lin, M.D., Ph.D., Children’s National
“Pediatric Lyme outcomes study – interim update”
Roberta L. DeBiasi, M.D., MS, Children’s National
“Molecular drivers and opportunities in neuroimmune conditions of pediatric onset”
Elizabeth Wells, M.D., Children’s National
$5M in federal funding to help patients with urea cycle disorders

Andrea L. Gropman, M.D.: We have collected many years of longitudinal clinical data, but with this new funding now we can answer questions about these diseases that are meaningful on a day-to-day basis for patients with urea cycle disorders.
An international research consortium co-led by Andrea L. Gropman, M.D., at Children’s National Hospital has received $5 million in federal funding as part of an overall effort to better understand rare diseases and accelerate potential treatments to patients.
Urea cycle disorder, one such rare disease, is a hiccup in a series of biochemical reactions that transform nitrogen into a non-toxic compound, urea. The six enzymes and two carrier/transport molecules that accomplish this essential task reside primarily in the liver and, to a lesser degree, in other organs.
The majority of patients have the recessive form of the disorder, meaning it has skipped a generation. These kids inherit one copy of an abnormal gene from each parent, while the parents themselves were not affected, says Dr. Gropman, chief of the Division of Neurodevelopmental Pediatrics and Neurogenetics at Children’s National. Another more common version of the disease is carried on the X chromosome and affects boys more seriously that girls, given that boys have only one X chromosome.
Regardless of the type of urea cycle disorder, when the urea cycle breaks down, nitrogen converts into toxic ammonia that builds up in the body (hyperammonemia), particularly in the brain. As a result, the person may feel lethargic; if the ammonia in the bloodstream reaches the brain in high concentrations, the person can experience seizures, behavior changes and lapse into a coma.
Improvements in clinical care and the advent of effective medicines have transformed this once deadly disease into a more manageable chronic ailment.
“It’s gratifying that patients diagnosed with urea cycle disorder now are surviving, growing up, becoming young adults and starting families themselves. Twenty to 30 years ago, this never would have seemed conceivable,” Dr. Gropman says. “We have collected many years of longitudinal clinical data, but with this new funding now we can answer questions about these diseases that are meaningful on a day-to-day basis for patients with urea cycle disorders.”
In early October 2019, the National Institutes of Health (NIH) awarded the Urea Cycle Disorders Consortium for which Dr. Gropman is co-principal investigator a five-year grant. This is the fourth time that the international Consortium of physicians, scientists, neuropsychologists, nurses, genetic counselors and researchers has received NIH funding to study this group of conditions.
Dr. Gropman says the current urea cycle research program builds on a sturdy foundation built by previous principal investigators Mendel Tuchman, M.D., and Mark Batshaw, M.D., also funded by the NIH. While previous rounds of NIH funding powered research about patients’ long-term survival prospects and cognitive dysfunction, this next phase of research will explore patients’ long-term health.
Among the topics they will study:
Long-term organ damage. Magnetic resonance elastrography (MRE) is a state-of-the-art imaging technique that combines the sharp images from MRI with a visual map that shows body tissue stiffness. The research team will use MRE to look for early changes in the liver – before patients show any symptoms – that could be associated with long-term health impacts. Their aim is spot the earliest signs of potential liver dysfunction in order to intervene before the patient develops liver fibrosis.
Academic achievement. The research team will examine gaps in academic achievement for patients who appear to be underperforming to determine what is triggering the discrepancy between their potential and actual scholastics. If they uncover issues such as learning difficulties or mental health concerns like anxiety, there are opportunities to intervene to boost academic achievement.
“And if we find many of the patients meet the criteria for depression or anxiety disorders, there are potential opportunities to intervene. It’s tricky: We need to balance their existing medications with any new ones to ensure that we don’t increase their hyperammonemia risk,” Dr. Gropman explains.
Neurologic complications. The researchers will tap continuous, bedside electroencephalogram, which measures the brain’s electrical activity, to detect silent seizures and otherwise undetectable changes in the brain in an effort to stave off epilepsy, a brain disorder that causes seizures.
“This is really the first time we will examine babies’ brains,” she adds. “Our previous imaging studies looked at kids and adults who were 6 years and older. Now, we’re lowering that age range down to infants. By tracking such images over time, the field has described the trajectory of what normal brain development should look like. We can use that as a background and comparison point.”
In the future, newborns may be screened for urea cycle disorder shortly after birth. Because it is not possible to diagnose it in the womb in cases where there is no family history, the team aims to better counsel families contemplating pregnancy about their possible risks.
Research described in this post was underwritten by the NIH through its Rare Diseases Clinical Research Network.
Children’s National/NIH team competes in #IDbugbowl
IDBugBowl team member Maria Susana Rueda-Altez, M.D., hopes her knowledge of infectious diseases common to Peru, like dengue virus, will give her team an advantage.
It’s a bird. It’s a plane. No, it’s an infectious agent that zipped past country borders, infecting international passengers who shared the same commercial aircraft as a person who had symptomatic illness.
The buzzer rings. And the correct answer is: What is severe acute respiratory syndrome?
This fall, a combined team from Children’s National in Washington, D.C. and the National Institutes of Health (NIH) will compete against three other teams testing their collective infectious disease knowledge through IDBugBowl, a Jeopardy-style quiz geared toward fellows, residents and medical students. The competition is held during IDWeek2019. “From anaplasmosis to Zika, any topic is fair game,” according to organizers.
“BugBowl has become so popular that the IDWeek 2019 program committee carved out a separate time for the contest to ensure it would not conflict with any other symposia,” says Roberta L. DeBiasi, M.D., MS, chief of the Division of Pediatric Infectious Diseases at Children’s National. “On a day-to-day basis, we all contend with serious infectious diseases that have the potential to jeopardize human health. However, this event helps to expand knowledge among the general public in a fun and engaging way.”
The Children’s National/NIH team participating in the Oct. 5 trivia contest includes:
- Kevin Lloyd, M.D., third-year pediatrics resident
- Maria Susana Rueda-Altez, M.D., third-year pediatrics resident
- Kanal Singh, M.D., fellow, adult infectious diseases at the National Institutes of Health (NIH) and
- Alexandra Yonts, M.D., fellow, pediatric infectious diseases at Children’s National
Even though she has little formal training in infectious diseases, team member Dr. Rueda-Altez says: “One thing I have in my favor is that I’m from Peru. We’re used to seeing infectious diseases that are less common elsewhere, including tuberculosis and hantavirus.”
And while disease-carrying mosquitoes aren’t abundant at Peru’s higher altitudes, closer to sea level and in its rain forests, infected mosquitoes spread chikungunya, dengue, malaria and Zika, she adds.
Take this quiz to test your infectious disease knowledge.
$2M from NIH to extract meaningful data from CRISPR screens

Protein-coding genes comprise a mere 1% of DNA. While the other 99% of DNA was once derided as “junk,” it has become increasingly apparent that some non-coding genes enable essential cellular functions.
Wei Li, Ph.D., a principal investigator in the Center for Genetic Medicine Research at Children’s National in Washington, D.C., proposes to develop statistical and computational methods that sidestep existing hurdles that currently complicate genome-wide CRISPR/Cas9 screening. The National Institutes of Health has granted him $2.23 million in funding over five years to facilitate the systematic study of genes, non-coding elements and genetic interactions in various biological systems and disease types.
Right now, a large volume of screening data resides in the public domain, however it is difficult to compare data that is stored in one library with data stored at a different library. Over the course of the five-year project, Li aims to:
- Improve functional gene identification from CRISPR screens.
- Develop new analyses algorithms for screens targeting non-coding elements.
- Study genetic interactions from CRISPR screens targeting gene pairs.
Ultimately, Li’s work will examine a range of disease types. Take cancer.
“There is abundant information already available in the public domain, like the Project Achilles from the Broad Institute. However, no one is looking to see what is going in inside these tumors,” Li says. “Cancer is a disease of uncontrolled cell growth that makes tumors grow faster.”
Li and colleagues are going to ask which genes control this process by looking at genes that hit the brakes on cell growth as well as genes that pump the gas.
“You knock out one gene and then look: Does the cell grow faster or does it grow more slowly? If the cell grows more slowly, you know you are knocking out a gene that has the potential to stop tumor growth. If cells are growing faster, you know that you’re hitting genes that suppress cancer cell growth.”
In a nutshell, CRISPR (clustered regularly interspaced short palindromic repeats) screens knock out different genes and monitor changes in corresponding cell populations. When CRISPR first became popular, Li decided he wanted to do something with the technology. So, as a Postdoc at Harvard, he developed comprehensive computational algorithms for functional screens using CRISPR/Cas9.
To reach as many people as possible, he offered that MAGeCK/MAGeCK-VISPR software free to as many researchers as possible, providing source code and offering internet tutorials.
“So far, I think there are quite a lot of people using this. There have been more than 40,000 software downloads,” he adds. “It’s really exciting and revolutionary technology and, eventually, we hope the outcomes also will be exciting. We hope to find something really helpful for cancer patients.”
Research reported in this publication was supported by the National Human Genome Research Institute of the National Institutes of Health under award number R01HG010753.
Researchers receive $2.5M grant to optimize brain development in babies with CHD
Children’s National Health System researchers Richard Jonas, M.D., Catherine Bollard, M.B.Ch.B., M.D., and Nobuyuki Ishibashi, M.D., have been awarded a $2.5 million, three-year grant from the National Institutes of Health (NIH) to conduct a single-center clinical trial at Children’s National. The study will involve collaboration between the Children’s National Heart Institute, the Center for Cancer and Immunology Research, the Center for Neuroscience Research and the Sheikh Zayed Institute for Pediatric Surgical Innovation.
The goal of the study will be to optimize brain development in babies with congenital heart disease (CHD) who sometimes demonstrate delay in the development of cognitive and motor skills. This can be a result of multiple factors including altered prenatal oxygen delivery, brain blood flow and genetic factors associated with surgery including exposure to the heart lung machine.
The award will be used to complete three specific aims of a Phase 1 safety study as described in the NIH grant:
- Aim 1: To determine the safety and feasibility of delivering allogeneic bone marrow derived mesenchymal stromal cell (BM-MSC) during heart surgery in young infants less than 3 months of age using the heart lung machine. The optimal safe dose will be determined.
- Aim 2: To determine the impact of MSC infusion on brain structure using advanced neuroimaging and neurodevelopmental outcomes.
- Aim 3: To determine differences in postoperative inflammatory and patho-physiological variables after MSC delivery in the infant with CHD.
“NIH supported studies in our laboratory have shown that MSC therapy may be extremely helpful in improving brain development in animal models after cardiac surgery,” says Dr. Ishibashi. “MSC infusion can help reduce inflammation including prolonged microglia activation that can occur during surgery that involves the heart lung machine.”
In addition the researchers’ studies have demonstrated that cell-based intervention can promote white matter regeneration through progenitor cells, restoring the neurogenic potential of the brain’s own stem cells that are highly important in early brain development.
The Phase 1 clinical trial is being implemented in two stages beginning with planning, regulatory documentation, training and product development. During the execution phase, the trial will focus on patient enrollment. Staff from the Cellular Therapy Laboratory, led by director Patrick Hanley, Ph.D., manufactured the BM-MSC at the Center for Cancer and Immunology Research, led by Dr. Bollard. The Advanced Pediatric Brain Imaging Laboratory, led by Catherine Limperopoulos, Ph.D., will perform MR imaging.
The phase 1 safety study will set the stage for a phase 2 effectiveness trial of this highly innovative MSC treatment aimed at reducing brain damage, minimizing neurodevelopmental disabilities and improving the postoperative course in children with CHD. The resulting improvement in developmental outcome and lessened behavioral impairment will be of enormous benefit to individuals with CHD.
Antonio R. Porras, Ph.D., awarded prestigious NIH grant for craniosynostosis modeling, career advancement

Antonio R. Porras, Ph.D., is a staff scientist in the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Health System.
Antonio R. Porras, Ph.D., a staff scientist in the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Health System, has received the prestigious Pathway to Independence Award from the National Institutes of Health (NIH). This award funds Dr. Porras’ research for the next five years, enabling him to develop two bone growth models that will better inform clinicians treating patients with craniosynostosis and help to optimize outcomes. Also referred to as the K99/R00 grant, this NIH award is for researchers who are either in the postdoctoral/residency period or who are early career investigators. It is designed to transition them from mentored positions to independent, tenure-track or equivalent faculty positions so that they can launch competitive research careers.
Marius George Linguraru, D.Phil., M.A., M.Sc., a principal investigator in the Sheikh Zayed Institute for Pediatric Surgical Innovation, is Dr. Porras’ primary mentor on this research project along with co-mentors Robert Keating, M.D., division chief of neurosurgery at Children’s National, and Maximilian Muenke, M.D., chief in the Medical Genetics Branch at the National Human Genome Research Institute.
Dr. Porras has taken a research interest in craniosynostosis, the early fusion of one or more cranial sutures that may lead to craniofacial malformations and brain growth constraints during childhood. With this NIH K99/R00 award, Dr. Porras will employ his expertise in computer science, biomedical engineering, quantitative imaging and statistical modeling to create a personalized computational predictive model of cranial bone growth for subjects without cranial pathology and for patients with craniosynostosis. Dr. Porras will also quantify the coupled growth patterns of the cranial bones and the brain using an existing brain growth model.
Affecting one in 2,100 to 2,500 live births, craniosynostosis complications can result in elevated intra-cranial pressure and subsequent impaired brain growth. While treatable, optimal outcomes are stymied by subjectivity in the evaluation of cranial malformations and prediction of cranial bone development. There are currently no personalized clinical tools available to predict healthy or pathological cranial growth and no objective techniques to optimize the long-term outcome of treatment for patients with craniosynostosis.
Gustavo Nino, M.D., honored with national award from American Thoracic Society

Gustavo Nino, M.D., a pulmonologist who directs the Sleep Medicine program at Children’s National, was honored by the American Thoracic Society with The Robert B. Mellins, M.D. Outstanding Achievement Award in recognition of his contributions to pediatric pulmonology and sleep medicine.
“I am humbled and pleased to be recognized with this distinction,” says Dr. Nino. “This national award is particularly special because it honors both academic achievements as well as research that I have published to advance the fields of pediatric pulmonology and sleep medicine.”
After completing a mentored career development award (K Award) from the National Institutes of Health (NIH), Dr. Nino established an independent research program at Children’s National funded by three different NIH R-level grants, an R01 research project grant; an R21 award for new, exploratory research; and an R4 small business/technology transfer award to stimulate research innovation.
The research team Dr. Nino leads has made important contributions to developing novel models to study the molecular mechanisms of airway epithelial immunity in newborns and infants. He also has pioneered the use of computer-based lung imaging tools and physiological biomarkers to predict early-life respiratory disease in newborns and infants.
Dr. Nino has published roughly 60 peer-review manuscripts including in the “Journal of Allergy and Clinical Immunology,” the “European Respiratory Journal,” and the “American Journal of Respiratory and Critical Care Medicine,” the three top journals in the field of respiratory medicine. He has been invited to chair sessions about sleep medicine during meetings held by the Pediatric Academic Societies, American College of Chest Physicians and the American Thoracic Society (ATS).
Dr. Nino also has served as NIH scientific grant reviewer of the Lung Cellular and Molecular Immunology Section; The Infectious, Reproductive, Asthma and Pulmonary Conditions Section; and The Impact of Initial Influenza Exposure on Immunity in Infants NIH/National Institute of Allergy and Infectious Diseases Special Emphasis Panel.
In addition to his research and academic contributions, over the past five years Dr. Nino has led important clinical and educational activities at Children’s National and currently directs the hospital’s Sleep Medicine program, which has grown to become one of the region’s largest programs conducting more than 1,700 sleep studies annually.
He has developed several clinical multidisciplinary programs including a pediatric narcolepsy clinic and the Advanced Sleep Apnea Program in collaboration with the Division of Ear, Nose and Throat at Children’s National. In addition, Dr. Nino started a fellowship program in Pediatric Sleep Medicine accredited by the Accreditation Council for Graduate Medical Education in collaboration with The George Washington University and has served as clinical and research mentor of several medical students, pediatric residents and fellows.
Steven Hardy, Ph.D., awarded prestigious NIH grant for sickle cell research, career development

Steven Hardy, Ph.D., a pediatric psychologist in the Center for Cancer and Blood Disorders at Children’s National, has been awarded a K23 Mentored Patient-Oriented Research Career Development Award by the National Heart, Lung, and Blood Institute (NHLBI) in recognition of his progress toward a productive, independent clinical research career. National Institutes of Health (NIH) Mentored Career Development Awards are designed to provide early career investigators with the time and support needed to focus on research and develop new research capabilities that will propel them to lead innovative studies in the future.
Dr. Hardy, who has worked at Children’s National since 2013, specializes in the emotional, behavioral and cognitive aspects of children’s health, with a particular emphasis on evaluating and treating psychological difficulties among children with cancer or sickle cell disease. With the K23 award, he will receive nearly $700,000 over a five-year period, which will provide him with an intensive, supervised, patient-oriented research experience. The grant will support Dr. Hardy’s time to conduct research, allow him to attend additional trainings to enhance research skills, and fund a research project titled “Trajectory of Cognitive Functioning in Youth with Sickle Cell Disease without Cerebral Infarction.”
Many children with sickle cell disease (SCD) also have intellectual challenges which stem from two primary pathways – stroke and other disease-related central nervous system effects. While stroke is a major complication of SCD, the majority of children with SCD have no evidence of stroke but may still exhibit cognitive functioning challenges related to their disease. Such cognitive difficulties have practical implications for the 100,000 individuals in the SCD, as 20-40% of youth with SCD repeat a grade in school and fewer than half of adults with SCD are employed. Dr. Hardy’s project will focus on understanding the scope and trajectory of cognitive difficulties in children with SCD without evidence of stroke, as well as the mechanisms that precipitate disease-related cognitive decline. The study will characterize temporal relationships between biomarkers of SCD severity and changes in cognitive functioning to inform future development of risk stratification algorithms to predict cognitive decline. Armed with the ability to predict cognitive decline, families will have additional information to weigh when making decisions and providers will be better able to intervene and tailor treatment.
$2M NIH grant for treating disease linked to APOL1

Children’s researcher Zhe Han, Ph.D., has received a $2 million award from the National Institutes of Health (NIH) to study new approaches to treat kidney disease linked to inheriting Apolipoprotein L1 (APOL1) risk alleles. These risk alleles are particularly common among persons of recent African descent, and African Americans are disproportionately affected by the increased risk in kidney disease associated with these risk alleles.
Han, an associate professor in Children’s Center for Genetic Medicine Research, has established a leading research program that uses the fruit fly Drosophila as a model system to study how genetic mutations lead to disease.
“Drosophila is a very basic model, but studies in the fly have led to major breakthroughs in understanding fundamental biological processes that underlie health and disease in humans,” Han says. “Since coming to Children’s National five years ago, I have focused a significant part of my research studying particular fly cells called nephrocytes that carry out many of the important roles of human kidney glomeruli, units within the kidney where blood is cleaned. Working together with clinician colleagues here, we have demonstrated that these Drosophila cells can be used to very efficiently study different types of renal disease caused by genetic mutations.”
The APOL1 risk alleles are genetic variants, termed G1 and G2, found almost exclusively in people of African ancestry and can lead to a four-fold higher risk of end-stage kidney disease, the last of five stages of chronic kidney disease. Exactly how inheriting these risk alleles increases the risk of kidney disease remains an unanswered question and the focus of considerable research activity. Han’s laboratory has developed a Drosophila model of APOL1-linked renal disease by producing the G1 and G2 forms of APOL1 specifically in nephrocytes. This led to defects in fly renal cells that strikingly overlap with disease-associated changes in experimental model and human kidney cells expressing APOL1 risk alleles.
The new NIH award will fund large-scale screening and functional testing to identify new treatment targets and new drugs to treat kidney disease linked to APOL1. Using a genetic screening approach, Han’s lab will identify nephrocyte “modifier” genes that interact with APOL1 proteins and counter the toxic effects of risk-associated G1 and G2 variants.
The team also will identify nephrocyte genes that are turned on or off in the presence of APOL1 risk alleles, and confirm that such “downstream” APOL1-regulated genes are similarly affected in experimental model and human kidney cells. The potential of the newly identified “modifier” and “downstream” genes to serve as targets of novel therapeutic interventions will be experimentally tested in fly nephrocytes in vivo and in cultured mammalian kidney cells.
Finally, the Drosophila model will be used as a drug screening platform for in vivo evaluation of positive “hits” from a cell-based APOL1 drug screening study in order to identify compounds that are most effective with the fewest side effects.
“These types of studies can be most efficiently performed in Drosophila,” Han adds. “They take advantage of the speed and low cost of the fly model system and the amazing array of well-established, sophisticated genetic tools available for the fly. Using this model to elucidate human disease mechanisms and to identify new effective therapies has truly become my research passion.”
$2 million NIH grant to study nephrotic syndrome

A Children’s researcher has received a $2 million grant from the National Institutes of Health (NIH) to study nephrotic syndrome in Drosophila, a basic model system that has revealed groundbreaking insights into human health. The award for Zhe Han, Ph.D., an associate professor in Children’s Center for Genetic Medicine Research, is believed to be the first ever NIH Research Project grant (R01) to investigate glomerular kidney disease using Drosophila. Nephrotic syndrome is mostly caused by damage of glomeruli, so it is equivalent to glomerular kidney disease.
“Children’s National leads the world in using Drosophila to model human kidney diseases,” Han says.
In order to qualify for the five-year funding renewal, Han’s lab needed to successfully accomplish the aims of its first five years of NIH funding. During the first phase of funding, Han established that nephrocytes in Drosophila serve the same functions as glomeruli in humans, and his lab created a series of fly models that are relevant for human glomerular disease.
“Some 85 percent of the genes known to be involved in nephrotic syndrome are conserved from the fly to humans. They play similar roles in the nephrocyte as they play in the podocytes in human kidneys,” he adds.
Pediatric nephrotic syndrome is a constellation of symptoms that indicate when children’s kidneys are damaged, especially the glomeruli, units within the kidney that filter blood. Babies as young as 1 year old can suffer proteinuria, which is characterized by too much protein being released from the blood into the urine.
“It’s a serious disease and can be triggered by environmental factors, taking certain prescription medicines or inflammation, among other factors. Right now, that type of nephrotic syndrome is mainly treated by steroids, and the steroid treatment works in many cases,” he says.
However, steroid-resistant nephrotic syndrome occurs primarily due to genetic mutations that affect the kidney’s filtration system: These filters are either broken or the protein reabsorption mechanism is disrupted.
“When genetics is to blame, we cannot turn to steroids. Right now there is no treatment. And many of these children are too young to be considered for a kidney transplant,” he adds. “We have to understand exactly which genetic mutation caused the disease in order to develop a targeted treatment.”
With the new funding, Han will examine a large array of genetic mutations that cause nephrotic syndrome. He’s focusing his efforts on genes involved in the cytoskeleton, a network of filaments and tubules in the cytoplasm of living cells that help them to maintain shape and carry out important functions.
“Right now, we don’t really understand the cytoskeleton of podocytes – highly specialized cells that wrap around the capillaries of the glomerulus – because podocytes are difficult to access. To change a gene requires time and considerable effort in other experimental models. However, changing genes in Drosophila is very easy, quick and inexpensive. We can examine hundreds of genes involving the cytoskeleton and see how changing those genes affect kidney cell function,” he says.
Han’s lab already found that Coenzyme Q10, one of the best-selling nutrient supplements to support heart health also could be beneficial for kidney health. For the cytoskeleton, he has a different targeted medicine in mind to determine whether Rho inhibitors also could be beneficial for kidney health for patients with certain genetic mutations affecting their podocyte cytoskeleton.
“One particular aim of our research is to use the same strategy as we employed for the Coq2 gene to generate a personalized fly model for patients with cytoskeleton gene mutations and test potential target drugs, such as Rho inhibitors.” Han added. “As far as I understand, this is where the future of medicine is headed.”
$3 million NIH grant to study APOL1 and HIV synergy

Zhe Han, Ph.D., (pictured) and Patricio E. Ray, M.D., have received a $3 million, five-year grant from the National Institutes of Health to study the mechanisms behind APOL1 and HIV nephropathies in children, using a combination of Drosophila models, cultured human podocytes and a preclinical model.
Two Children’s researchers have received a $3 million, five-year grant from the National Institutes of Health (NIH) to study the mechanisms of APOL1 and HIV nephropathies in children, using a combination of Drosophila models, cultured human podocytes and a preclinical model.
The APOL1 genetic variants G1 and G2, found almost exclusively in people of African ancestry, lead to a four-fold higher risk of end-stage kidney disease. HIV infection alone also increases the risk of kidney disease but not significantly. However, HIV-positive people who also carry the APOL1 risk alleles G1 or G2 are about 30 times more likely to develop HIV-nephropathy (HIVAN) and chronic kidney disease.
For more than 25 years, Children’s pediatric nephrology program has studied HIV/renal diseases and recently developed Drosophila APOL1-G0 and G1 transgenic lines. That pioneering research suggests that HIV-1 acts as a “second hit,” precipitating HIV-renal disease in children by infecting podocytes through a mechanism that increases expression of the APOL1-RA beyond toxic thresholds.
With this new infusion of NIH funding, labs led by Zhe Han, Ph.D., and Patricio E. Ray, M.D., will determine the phenotype of Drosophila Tg lines that express APOL1-G0/G1/G2 and four HIV genes in nephrocytes to assess how they affect structure and function. The teams also will determine whether APOL1-RA precipitates the death of nephrocytes expressing HIV genes by affecting autophagic flux.
“Our work will close a critical gap in understanding about how HIV-1 interacts with the APOL1 risk variants in renal cells to trigger chronic kidney disease, and we will develop the first APOL1/HIV transgenic fly model to explore these genetic interactions in order to screen new drugs to treat these renal diseases,” says Dr. Ray, a Children’s nephrologist.
While a large number of people from Africa have two copies of APOL1 risk alleles, they do not necessarily develop kidney disease. However, if a patient has two copies of APOL1 risk alleles and is HIV-positive, they almost certainly will develop kidney disease.

“Our work will close a critical gap in understanding about how HIV-1 interacts with the APOL1 risk variants in renal cells to trigger chronic kidney disease, and we will develop the first APOL1/HIV transgenic fly model to explore these genetic interactions in order to screen new drugs to treat these renal diseases,” says Dr. Ray, a Children’s nephrologist.
“Many teams want to solve the puzzle of how APOL1 and HIV synergize to cause kidney failure,” says Han, associate professor in Children’s Center for Genetic Medicine Research. “We are in the unique position of combining a powerful new kidney disease model system, Drosophila, with long-standing human podocyte and HIVAN studies.”
The team hypothesizes that even as an active HIV infection is held in check by powerful new medicines, preventing the virus from proliferating or infecting new cells, HIV can act as a Trojan horse by making the human cells it infects express HIV protein.
To investigate this hypothesis, the team will create a series of fly models, each expressing a major HIV protein, and will test the genetic interaction between these HIV genes with APOL1. Similar studies also will be performed using cultured human podocytes. Identified synergy will be studied further using biochemical and transcription profile analyses.
“Drosophila is a basic model system, but it has been used to make fundamental discoveries, including genetic control of how the body axes is determined and how the biological clock works – two studies that led to Nobel prizes,” Han adds. “I want to use the fly model to do something close to human disease. That is where my research passion lies.”
Marva Moxey-Mims, M.D., named NephCure Kidney International scientific adviser

Marva Moxey-Mims, M.D., chief of the Division of Nephrology at Children’s National Health System, has been named to the Scientific Advisory Board for NephCure Kidney International, a non-profit that aims to accelerate research for rare forms of nephrotic syndrome.
Dr. Moxey-Mims and two additional scientific advisers were selected for their commitment to improving care for patients with glomerular disease, diseases that impair kidney function by attacking blood cleaning units within the kidney.
During her tenure at the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health, Dr. Moxey-Mims launched the Chronic Kidney Disease in Children Cohort Study, a prospective study to investigate chronic kidney disease risk factors and outcomes, and helped launch the Cure Glomerulonephropathy Network, a multi-site study with the overarching aim to advance the diagnosis and care of patients with four different glomerular diseases.
“I am truly honored to join this distinguished group of scientific advisers and look forward to leveraging our combined strengths and research knowledge in order to deliver cures for kidney diseases faster,” says Dr. Moxey-Mims.
Innovative urologist Michael Hsieh takes unbeaten path

For an elementary school art project, Michael H. Hsieh, M.D., Ph.D., was instructed to fashion a coat of arms out of clay. In addition to panels for truth, justice and Taiwan, in the shield’s M.D. panel, a snake twists around a rod, like the staff for Asclepius, a Greek god associated with healing.
Children’s urologist Michael H. Hsieh, M.D., Ph.D., knew from age 10 that he would become a doctor. Proof is at his parents’ home. For an elementary school art project, students were instructed to fashion a coat of arms out of clay. In addition to panels for truth, justice and Taiwan, in the shield’s M.D. panel, a snake twists around a rod, like the staff for Asclepius, a Greek god associated with healing.
“I liked science. When I can use it to help patients, that is very rewarding,” says Dr. Hsieh, the first doctor in his family.
These days, Dr. Hsieh’s Twitter profile serves as a digital coat of arms, describing him as “tinker, tailor,” #UTI #biologist, epithelial #immunologist, helminthologist and #urologist.
Tinker/tailor is shorthand for the mystery drama, “Tinker Tailor Solider Spy,” he explains, adding that the “tinker” part also refers “to the fact that I am always questioning things, and science is about experimentation, trying to seek answers to questions.”
While still in medical school during a rotation Dr. Hsieh saw a bladder operation on a young child and thought it was “amazing.” That experience in part inspired Dr. Hsieh to become a urologist and bladder scientist. His training in immunology and study of the bladder naturally led him to study urinary tract infections and parasitic worms that affect the urinary tract. In addition, thanks to R01 funding from the National Institutes of Health (NIH), Dr. Hsieh is co-principal investigator with Axel Krieger, University of Maryland, and Jin U. Kang, Johns Hopkins, on a project to develop imaging robots for supervised autonomous surgery on soft tissue.
The $1 million in NIH funding pushes the boundaries on amazing by using multi-spectral imaging technology and improved techniques to reduce surgical complications.
Anastomosis is a technique used by surgeons to join one thing to another, whether it’s a vascular surgeon suturing blood vessels, an orthopedic surgeon joining muscles or a urologist stitching healthy parts of the urinary tract back together. Complications can set in if their stitching is too tight, prompting scar tissue to form, or too loose, letting fluid seep out.
“The human eye can see a narrow spectrum of electromagnetic radiation. These multi-spectral imaging cameras would see across greater set of wavelengths,” he says.
The project has three aims: figuring out the best way to place sutures using multi-spectral imaging, accurately tracking soft tissue as they model suturing and comparing the handicraft of a robot against anastomosis hand-sewn by surgeons.
“I like challenges, and I like new things. I am definitely not interested in doing permutations of other people’s work,” Dr. Hsieh explains. “I would much rather go on a path that hasn’t been tread. It is more difficult in some ways, but on a day-to-day basis, I know I am making a contribution.”
In another innovative research project, Dr. Hsieh leveraged a protein secreted by a parasitic worm, Schistosoma haematobium, that suppresses inflammation in hosts as a new therapeutic approach for chemotherapy-induced hemorrhagic cystitis, a form of inflammation of the bladder.
Watching his first surgery nearly 30 years ago, he had no idea robots might one day vie to take over some part of that complicated procedure, or that parasite proteins could be harnessed as drugs. However, he has a clear idea which innovations could be on the horizon for urology in the next three decades.
“My hope is 30 years from now, we will have a solid UTI vaccine and more non-antibiotic therapies. UTIs are the second-most common bacterial infection in childhood and, in severe cases, can contribute to kidney failure,” he says.
Globally, parasitic worms pose an ongoing challenge, affecting more than 1 billion worldwide – second only to malaria. People persistently infected by schistosome worms fail to reach their growth potential, struggle academically and lack sufficient energy for exercise or work.
“There is a feeling that the infection prevalence might be decreasing globally, but not as quickly as everyone hopes. In 30 years perhaps with more mass drug administration and additional drugs – including a vaccine – we’ll have it close to eliminated globally. It would become more like polio, casting a slim shadow with small pockets of infection here or there, rather than consigning millions to perpetual poverty.”
Growth disorder study starts by analyzing DNA
The National Institutes of Health has awarded Andrew Dauber, M.D., MMSc, the chief of endocrinology at Children’s National Health System, a five-year grant that will allow four pediatric health systems to compile and study clinical and genetic markers of severe pediatric growth disorders.
The study will use the electronic health records of large health systems combined with DNA samples from dozens of children, with the goal of enabling endocrinologists to detect children with previously undiagnosed severe genetic growth disorders.
“If you’re a pediatrician treating an 8-year-old patient who has stopped growing, the first thing you’ll want to do is determine the underlying cause, which could be due to many factors including a genetic mutation,” says Dr. Dauber. “There are many reasons why children grow poorly and it is often very difficult to figure out what is causing the problem. However, the various causes may be treated quite differently and may alert us to other medical issues that we need to watch out for. We need to be able to identify clues from the patient’s clinical presentation that may point us to the right diagnosis.”
Dr. Dauber and endocrinology researchers from Children’s National Health System, Cincinnati Children’s Hospital Medical Center, Boston Children’s Hospital and The Children’s Hospital of Philadelphia will use electronic health records to identify children who likely have rare genetic growth disorders. They will then use cutting-edge DNA sequencing technologies, whole exome sequences, to identify novel genetic causes of severe growth disorders. Patients with growth hormone resistance, resistance to insulin-like growth factor 1 (IGF-I) and severe short stature inherited from a single parent will be recruited for the initial phases of the study.
“It’s rare to find patients meeting criteria for each of these subgroups, which is why it’s critical to work collaboratively across institutions,” says Dr. Dauber. “This type of genetic sorting and sharing brings us closer to identifying new markers for severe or treatment-resistant growth disorders, which will help alert pediatricians and parents to potential risks earlier on in a child’s life.”
In addition to assessing genetic markers for short stature, the endocrinologists will conduct pilot studies of targeted interventions, such as IGF-I therapy in patients with mutations in the growth hormone pathway, based on these genetic underpinnings.
“Ideally, by identifying markers of severe growth disorders first, we’ll be able to provide targeted treatments and therapies later on to help patients throughout their lifespan,” adds Dr. Dauber.
Typical treatments for atypical growth patterns include growth hormone or less commonly insulin-like growth factor, or IGF-1, for short stature and hormone-inhibiting treatments for precocious puberty.
The multicenter clinical trial is funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), under grant Ro1HD093622, and runs through June 30, 2023.