Tag Archive for: MRI
Building “digital twins” to test complicated surgeries
Syed Anwar, Ph.D., is developing self-supervised algorithms for medical imaging.
Syed Anwar, Ph.D., joins the growing AI initiative in the Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI) at Children’s National Hospital with extensive research experience in machine learning and medical imaging from the University of Engineering and Technology in Taxila, Pakistan, the University of Sheffield, U.K., and the University of Central Florida through the Fulbright Scholars Program. At Children’s National, he’s grateful for the proximity between researchers and clinicians as he studies federated learning and works to build “digital twins” that allow medical teams to test complicated surgical and treatment plans on infants with disorders including Pierre Robin Sequence. This rare congenital birth defect is characterized by an underdeveloped jaw, backward displacement of the tongue and upper airway obstruction. Anwar works alongside Marius George Linguraru, D.Phil., M.A., M.Sc., principal investigator at SZI, and the Precision Medical Imaging Lab to increase AI capacity in all areas of pediatric care at the hospital.
Q: What is the focus of your research work?
A: The main theme is a digital twin. It’s an engineering innovation that people have been using for some time, especially in manufacturing and aviation. For example, you can create a digital simulation of an airplane with a flight simulator. Now, people are starting to use the power of data-driven digital twins for medical applications.
I’m working to create a digital twin for infants born with Pierre Robin Sequence, where they need to have surgical interventions for improving the structure of the bones in the jaws. It includes a lot of clinical approaches, including surgery and ways to address apnea and food intake.
There are multiple areas of clinical expertise involved. With a digital twin, we will have a digital representation of the patient, and the surgeon, the radiologist and other clinicians can experiment with a proposed intervention before actually touching the patient.

Syed Anwar, Ph.D., joins the growing AI initiative in the Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI) at Children’s National Hospital.
Q: How else are you using your engineering background in your research?
A: Another part of my work is federated learning, which is a type of machine learning. In artificial intelligence, we want big data as the starting point to train our deep learning models. When studying children, this is not always possible because we have smaller data sets.
Federated learning is a tool that helps in these situations. Data is kept at a local site. We train a model to learn from all that data at the different sites. One benefit is that we don’t need to share the data, which is very useful for preserving patient privacy. But you can still apply deep learning models and develop AI solutions using the distributed data for improved clinical outcomes.
Q: What do you see as the main hurdles you have to overcome?
A: For all medical data, and particularly for kids, the amount of data we see in a children’s hospital is small, particularly for rare diseases.
The second hurdle is good, quality labels. For example, if you are doing tumor segmentation, you still need to have some ground rules from a radiologist showing which part of the image is the tumor.
These challenges come together in another focus of my research – self-supervised learning, meaning we can train a machine to learn from the data itself, without the labels or ground rules. From a machine learning point of view, I am in the process of developing self-supervised algorithms for medical imaging and in general for medical data. It’s an amazing time to be in this research area and to enable the translation of AI driven solutions for clinical workflows.
Q: What excites you about being at Children’s National and working at SZI?
A: I come from an engineering background, and my research area has been medical imaging for some time, mainly magnetic resonance imaging. Before coming here, I was working at a university in Pakistan, teaching machine learning and conducting research related to medical imaging and biomedical signal processing. But I was missing strong connections with people caring for patients at the hospital.
AI algorithm that detects brain abnormalities could help cure epilepsy

A new AI algorithm can detect subtle brain abnormalities that cause epileptic seizures.
An artificial intelligence (AI) algorithm that can detect subtle brain abnormalities that cause epileptic seizures has been developed by a UCL-led team of international researchers, including Children’s National Hospital.
To do this, the team quantified features from MRI scans, such as how thick or folded the brain was at nearly 300,000 locations in each case.
They then trained the AI algorithm using examples labelled by expert radiologists as either a healthy brain or one with focal cortical dysplasia (FCD) based on their patterns and features.
The results, published in Brain, showed that in the main cohort of 538 patients, the algorithm was able to detect the FCD in 67% of cases.
“We put an emphasis on creating an AI algorithm that was interpretable and could help doctors make decisions. Showing doctors how the Multicentre Epilepsy Lesion Detection project (MELD) algorithm made its predictions was an essential part of that process,” said Mathilde Ripart, research assistant at UCL and the study’s co-first author.
Around 1% of the population have epilepsy and, of these, 20-30% do not respond to medications.
“We are excited to collaborate with MELD on ways to improve the treatment of pharmacoresistant epilepsy,” said Nathan Cohen, M.D., neurologist at Children’s National Hospital and co-author of the study. “This advanced imaging platform is open source and demonstrates the benefit of team science at the broadest scale.”
In children who have had surgery to control their epilepsy, FCD is the most common cause, and in adults it is the third most common cause.
Additionally, of patients who have epilepsy that have an abnormality in the brain that cannot be found on MRI scans, FCD is the most common cause.
You can read the full UCL press release here.
Early versus late MRI in newborn brain injury
A single magnetic resonance imaging (MRI) performed in the first week after birth is adequate to assess brain injury and offer prognostic information in newborn infants with hypoxic ischemic encephalopathy (HIE) treated with therapeutic hypothermia, according to a new study published in The Journal of Pediatrics.
A collaborative team of neonatology, neurology and neuroradiology experts from Children’s National Hospital that included Gilbert Vezina, M.D., Taeun Chang, M.D., and An N. Massaro, M.D., came together to evaluate the agreement in brain injury findings between early and late MRI in newborn infants with hypoxic ischemic encephalopathy (HIE) treated with therapeutic hypothermia. The team then compared the ability of early versus late MRI to predict early neurodevelopmental outcomes.
This was a prospective longitudinal study of 49 patients with HIE who underwent therapeutic hypothermia and had MRI performed at both <7 and ≥7 days of age. MRIs were reviewed by an experienced neuroradiologist and assigned brain injury severity scores according to established systems. Scores for early and late MRIs were assessed for agreement using the kappa statistic. The ability of early and late MRI scores to predict death or developmental delay at 15-30 months of age was assessed by logistic regression analyses.
The results of the study found agreement between the early and late MRI was substantial to near perfect (k>0.75, p<0.001) across MRI scoring systems. In cases of discrepant scoring, early MRI was more likely to identify severe injury when compared with late MRI. Early MRI scores were more consistently predictive of adverse outcomes compared with late MRI.
Read the full study in The Journal of Pediatrics.
Predicting risk for infantile spasms after acute symptomatic neonatal seizures
Infantile spasms (IS) is a severe epilepsy in early childhood. Early treatment of IS provides the best chance of seizure remission and favorable developmental outcome.
Taeun Chang, M.D., director of the Neonatal Neurology and Neurocritical Care Program at Children’s National Hospital, participated in a study with other national pediatric experts which aimed to develop a prediction rule to accurately predict which neonates with acute symptomatic seizures will develop IS.
The group of researchers found that multiple potential predictors were associated with IS, including Apgar scores, EEG features, seizure characteristics, MRI abnormalities and clinical status at hospital discharge. The final model born from this work included three risk factors: (a) severely abnormal EEG or ≥3 days with seizures recorded on EEG, (b) deep gray or brainstem injury on MRI and (c) abnormal tone on discharge exam.
The significance of these findings is that IS risk after acute symptomatic neonatal seizures can be stratified using commonly available clinical data. No child without risk factors, vs >50% of those with all three factors, developed IS. This risk prediction rule may be valuable for clinical counseling as well as for selecting participants for clinical trials to prevent post‐neonatal epilepsy. This tailored approach may lead to earlier diagnosis and treatment and improve outcomes for a devastating early life epilepsy.
Read the full study in Epilepsia.
Maternal mental health alters structure and biochemistry of developing fetal brain
Even when pregnant women have uncomplicated pregnancies and high socioeconomic status, when they experience elevated anxiety, stress or depression these prenatal stressors can alter the structure of the developing fetal brain and disrupt its biochemistry, according to Children’s National Hospital research published online Jan. 29, 2020, in JAMA Network Open.
The Children’s National research findings “have enormous scientific, clinical and public health implications,” Charles A. Nelson III, Ph.D., Boston Children’s Hospital, writes in a companion editorial.
“Previously we found that 65% of pregnant women who received a diagnosis of fetal congenital heart disease had elevated levels of stress. It’s concerning but not surprising that pregnant women who wonder if their baby will need open heart surgery would feel stress,” says Catherine Limperopoulos, Ph.D., director of the Center for the Developing Brain at Children’s National and the study’s senior author. “In this latest study, we ran the same panel of questionnaires and were surprised to find a high proportion of otherwise healthy pregnant women whose unborn babies are doing well also report high levels of stress.”
Anxiety and depression are the most common mental health problems during pregnancy. To learn more about the implications for the developing fetal brain, the Children’s National research team recruited 119 healthy volunteers with low-risk pregnancies from obstetric clinics in Washington, D.C., from Jan. 1, 2016, to April 17, 2019. The women’s mean age was 34.4 years old. All were high school graduates, 83% were college graduates, and 84% reported professional employment.
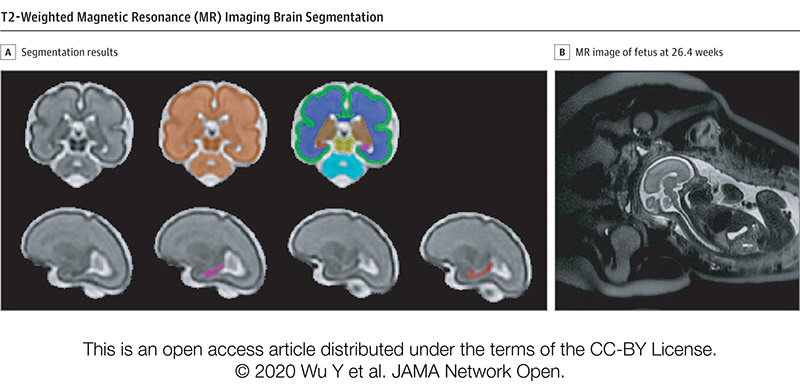
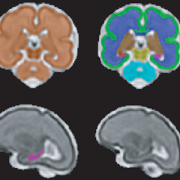
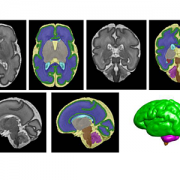
T2-Weighted Magnetic Resonance (MR) Imaging Brain Segmentation. Segmentation results of total brain (orange), cortical gray matter (green), white matter (blue), deep gray matter (brown), brainstem (yellow), cerebellum (light blue), left hippocampus (purple) and right hippocampus (red) on a 3-Dimensional reconstructed T2-weighted MR image of a fetus at 26.4 gestational weeks. The hippocampus plays a central role in memory and behavioral inhibition and contains high concentrations of corticosteroid receptors and, thus, this brain region is sensitive to stress. Credit: JAMA Network Open.
The team performed 193 fetal brain magnetic resonance imaging (MRI) sessions between 24-40 weeks gestation and measured the volume of the total fetal brain as well as the cortical gray matter, white matter, deep gray matter, cerebellum, brainstem and hippocampus volumes. On the same day as their MRI visit, the pregnant women completed validated questionnaires to measure maternal stress, anxiety and depression, answering questions such as “how do you feel right now,” “how do you generally feel” as well as the degree of stressful feelings they experienced the month prior.
Of the pregnant women in the study:
- 27% tested positive for stress
- 26% tested positive for anxiety
- 11% tested positive for depression
- Maternal anxiety and stress were associated with increased fetal cortical gyrification
- Elevated maternal depression was associated with decreased creatine and choline levels in the fetal brain
- Maternal stress scores decreased with increasing gestational age, while anxiety and depression did not
“We report for the first time that maternal psychological distress may be associated with increased fetal local gyrification index in the frontal and temporal lobes,” says Yao Wu, Ph.D., a research associate working with Limperopoulos at Children’s National and the study’s lead author. “We also found an association with left fetal hippocampal volume, with maternal psychological distress selectively stunting the left hippocampal volumetric growth more than the right. And elevated maternal depression was associated with decreased creatine and choline levels in the fetal brain,” Wu adds.
Late in pregnancy – at the time these women were recruited into the cohort study – the fetal brain grows exponentially and key metabolite levels also rise. Creatine facilitates recycling of adenosine triphosphate, the cell’s energy currency. Typically, levels of this metabolite rise, denoting rapid changes and higher cellular maturation; creatine also is known to support cognitive function. Choline levels also typically rise, marking cell membrane turnover as new cells are generated and support memory, mental focus and concentration.
“These women were healthy, and of high socioeconomic status and educational level, leading us to conclude that the prevalence of prenatal maternal psychological distress may be underestimated,” Limperopoulos adds. “While stress is an everyday reality for most of us, this is different because elevated stress during pregnancy can alter fetal brain programming. Our findings underscore the critical need to universally screen all pregnant women for prenatal psychological distress, even young mothers whose pregnancies wouldn’t otherwise raise red flags.”
In addition to Limperopoulos and Wu, Children’s National study co-authors include Yuan-Chiao Lu, Ph.D., research associate; Marni Jacobs, Ph.D., biostatistician; Subechhya Pradhan, Ph.D., research faculty; Kushal Kapse, MS, staff engineer; Li Zhao, Ph.D., research faculty; Nickie Niforatos-Andescavage, M.D., neonatologist; Gilbert Vezina, M.D., director of the neuroradiology program; and Adré J. du Plessis, M.B.Ch.B., director, Fetal Medicine Institute. Research coordinators Catherine Lopez, MS, Kathryn Lee Bannantine, BSN, and Jessica Lynn Quistorff, MPH, assisted with subject recruitment.
Financial support for the research described in this post was provided by the National Institutes of Health under grant No. RO1 HL116585-01 and the Thrasher Research Fund under Early Career award No. 14764.
Journal Reference:
Yao Wu, Yuan-Chiao Lu, Marni Jacobs, Subechhya Pradhan, Kushal Kapse, Li Zhao, Nickie Niforatos-Andescavage, Gilbert Vezina, Adré J. du Plessis, Catherine Limperopoulos. “Association of prenatal maternal psychological distress with fetal brain growth, metabolism and cortical maturation,” JAMA Network Open, 3(1): e1919940, 2020
False negatives: Delayed Zika effects in babies who appeared normal at birth

Colombian infants exposed to Zika virus in the womb showed neurodevelopmental delays as toddlers, despite having “normal” brain imaging and head circumference at birth, a finding that underscores the importance of long-term neurodevelopmental follow-up for Zika-exposed infants.
Colombian infants exposed to Zika virus in the womb showed neurodevelopmental delays as toddlers, despite having “normal” brain imaging and head circumference at birth, a finding that underscores the importance of long-term neurodevelopmental follow-up for Zika-exposed infants, according to a cohort study published online Jan. 6, 2020, in JAMA Pediatrics.
“These infants had no evidence of Zika deficits or microcephaly at birth. Neurodevelopmental deficits, including declines in mobility and social cognition, emerged in their first year of life even as their head circumference remained normal,” says Sarah B. Mulkey, M.D. Ph.D., a fetal/neonatal neurologist at Children’s National Hospital and the study’s first author. “About one-third of these newborns who underwent postnatal head ultrasound had nonspecific imaging results, which we believe are the first published results finding a link between subtle brain injuries and impaired neuromotor development in Zika-exposed children.”
The multi-institutional research group led by Children’s National enrolled pregnant women in Atlántico Department, which hugs the Caribbean coast of Colombia, who had been exposed to Zika, and performed a series of fetal magnetic resonance images (MRI) and ultrasounds as their pregnancies progressed.
Even though their mothers had laboratory-confirmed Zika infections, 77 out of 82 of their offspring were born with no sign of congenital Zika syndrome, a constellation of birth defects that includes severe brain abnormalities, eye problems and congenital contractures, and 70 underwent additional testing of neurodevelopment during infancy. These apparently normal newborns were born between Aug. 1, 2016, and Nov. 30, 2017, at the height of the Zika epidemic, and had normal head circumference.
When they were 4 to 8 months or 9 to 18 months of age, the infants’ neurodevelopment was evaluated using two validated tools, the Warner Initial Developmental Evaluation of Adaptive and Functional Skills (a 50-item test of such skills as self-care, mobility, communication and social cognition) and the Alberta Infant Motor Scale (a motor examination of infants in prone, supine, sitting and standing positions). Some infants were assessed during each time point.
Women participating in the study were highly motivated, with 91% following up with appointments, even if it meant traveling hours by bus. In addition to Children’s National faculty traveling to Colombia to train staff how to administer the screening instruments, videotaped assessments, MRIs and ultrasounds were read, analyzed and scored at Children’s National. According to the study team, the U.S. scoring of Alberta Infant Motor Scale tests administered in Colombia is also unprecedented for a research study and offers the potential of remote scoring of infants’ motor skill maturity in regions of the world where pediatric specialists, like child neurologists, are lacking.
“Normally, neurodevelopment in infants and toddlers continues for years, building a sturdy neural network that they later use to carry out complex neurologic and cognitive functions as children enter school,” Dr. Mulkey adds. “Our findings underscore the recommendations by the Centers for Disease Control and Prevention (CDC) that all infants exposed to Zika in the womb undergo long-term follow-up, providing an opportunity to intervene earlier.”
An accompanying editorial by CDC staffers concurs, saying the study reported “intriguing data” that add “to the growing evidence of the need for long-term follow-up for all children with Zika virus exposure in utero to ensure they receive the recommended clinical evaluations even when no structural defects are identified at birth.”
In addition to Dr. Mulkey, study co-authors include Margarita Arroyave-Wessel, MPH, Dorothy I. Bulas, M.D., chief of Diagnostic Imaging and Radiology, JiJi Jiang, MS, Stephanie Russo, BS, Robert McCarter, ScD, research section head, design and biostatistics, Adré J. du Plessis, M.B.Ch.B., MPH, chief of the Division of Fetal and Transitional Medicine, and co-Senior Author, Roberta L. DeBiasi, MD, MS, chief of the Division of Pediatric Infectious Diseases, all of Children’s National; Colleen Peyton, PT, DPT, of Northwestern University; Yamil Fourzali, M.D., of Sabbag Radiologos, Barranquilla, Colombia; Michael E. Msall, M.D., of University of Chicago Comer Children’s Hospital; and co-Senior Author, Carlos Cure, M.D., BIOMELab, Barranquilla, Colombia.
Funding for the research described in this post was provided by the Thrasher Research Fund, the National Institutes of Health under award Nos. UL1TR001876 and KL2TR001877, and the Leadership Education in Neurodevelopmental and Related Disorders Training Program under grant HRSA/MCHB T73 MC11047.
Novel approach to detect fetal growth restriction

Morphometric and textural analyses of magnetic resonance imaging can point out subtle architectural deviations associated with fetal growth restriction during the second half of pregnancy, a first-time finding that has the promise to lead to earlier intervention.
Morphometric and textural analyses of magnetic resonance imaging (MRI) can point out subtle architectural deviations that are associated with fetal growth restriction (FGR) during the second half of pregnancy. The first-time finding hints at the potential to spot otherwise hidden placental woes earlier and intervene in a more timely fashion, a research team led by Children’s National Hospital faculty reports in Pediatric Research.
“We found reduced placental size, as expected, but also determined that the textural metrics are accelerated in FGR when factoring in gestational age, suggesting premature placental aging in FGR,” says Nickie Andescavage, M.D., a neonatologist at Children’s National and the study’s lead author. “While morphometric and textural features can discriminate placental differences between FGR cases with and without Doppler abnormalities, the pattern of affected features differs between these sub-groups. Of note, placental insufficiency with abnormal Doppler findings have significant differences in the signal-intensity metrics, perhaps related to differences of water content within the placenta.”
The placenta, an organ shared by the pregnant woman and the developing fetus, delivers oxygen and nutrients to the developing fetus and ferries away waste products. Placental insufficiency is characterized by a placenta that develops poorly or is damaged, impairing blood flow, and can result in still birth or death shortly after birth. Surviving infants may be born preterm or suffer early brain injury; later in life, they may experience cardiovascular, metabolic or neuropsychiatric problems.
Because there are no available tools to help clinicians identify small but critical changes in placental architecture during pregnancy, placental insufficiency often is found after some damage is already done. Typically, it is discovered when FGR is diagnosed, when a fetus weighs less than 9 of 10 fetuses of the same gestational age.
“There is a growing appreciation for the prenatal origin of some neuropsychiatric disorders that manifest years to decades later. Those nine months of gestation very much define the breath of who we later become as adults,” says Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain at Children’s National and the study’s senior author. “By identifying better biomarkers of fetal distress at an earlier stage in pregnancy and refining our imaging toolkit to detect them, we set the stage to be able to intervene earlier and improve children’s overall outcomes.”
The research team studied 32 healthy pregnancies and compared them with 34 pregnancies complicated by FGR. These women underwent up to two MRIs between 20 weeks to 40 weeks gestation. They also had abdominal circumference, fetal head circumference and fetal femur length measured as well as fetal weight estimated.
In pregnancies complicated by FGR, placentas were smaller, thinner and shorter than uncomplicated pregnancies and had decreased placental volume. Ten of 13 textural and morphometric features that differed between the two groups were associated with absolute birth weight.
“Interestingly, when FGR is diagnosed in the second trimester, placental volume, elongation and thickness are significantly reduced compared with healthy pregnancies, whereas the late-onset of FGR only affects placental volume,” Limperopoulos adds. “We believe with early-onset FGR there is a more significant reduction in the developing placental units that is detected by gross measures of size and shape. By the third trimester, the overall shape of the placenta seems to have been well defined so that primarily volume is affected in late-onset FGR.”
In addition to Dr. Andescavage and Limperopoulos, study co-authors include Sonia Dahdouh, Sayali Yewale, Dorothy Bulas, M.D., chief of the Division of Diagnostic Imaging and Radiology, and Biostatistician, Marni Jacobs, Ph.D., MPH, all of Children’s National; Sara Iqbal, of MedStar Washington Hospital Center; and Ahmet Baschat, of Johns Hopkins Center for Fetal Therapy.
Financial support for research described in this post was provided by the National Institutes of Health under award number 1U54HD090257, R01-HL116585, UL1TR000075 and KL2TR000076, and the Clinical-Translational Science Institute-Children’s National.
$5M in federal funding to help patients with urea cycle disorders

Andrea L. Gropman, M.D.: We have collected many years of longitudinal clinical data, but with this new funding now we can answer questions about these diseases that are meaningful on a day-to-day basis for patients with urea cycle disorders.
An international research consortium co-led by Andrea L. Gropman, M.D., at Children’s National Hospital has received $5 million in federal funding as part of an overall effort to better understand rare diseases and accelerate potential treatments to patients.
Urea cycle disorder, one such rare disease, is a hiccup in a series of biochemical reactions that transform nitrogen into a non-toxic compound, urea. The six enzymes and two carrier/transport molecules that accomplish this essential task reside primarily in the liver and, to a lesser degree, in other organs.
The majority of patients have the recessive form of the disorder, meaning it has skipped a generation. These kids inherit one copy of an abnormal gene from each parent, while the parents themselves were not affected, says Dr. Gropman, chief of the Division of Neurodevelopmental Pediatrics and Neurogenetics at Children’s National. Another more common version of the disease is carried on the X chromosome and affects boys more seriously that girls, given that boys have only one X chromosome.
Regardless of the type of urea cycle disorder, when the urea cycle breaks down, nitrogen converts into toxic ammonia that builds up in the body (hyperammonemia), particularly in the brain. As a result, the person may feel lethargic; if the ammonia in the bloodstream reaches the brain in high concentrations, the person can experience seizures, behavior changes and lapse into a coma.
Improvements in clinical care and the advent of effective medicines have transformed this once deadly disease into a more manageable chronic ailment.
“It’s gratifying that patients diagnosed with urea cycle disorder now are surviving, growing up, becoming young adults and starting families themselves. Twenty to 30 years ago, this never would have seemed conceivable,” Dr. Gropman says. “We have collected many years of longitudinal clinical data, but with this new funding now we can answer questions about these diseases that are meaningful on a day-to-day basis for patients with urea cycle disorders.”
In early October 2019, the National Institutes of Health (NIH) awarded the Urea Cycle Disorders Consortium for which Dr. Gropman is co-principal investigator a five-year grant. This is the fourth time that the international Consortium of physicians, scientists, neuropsychologists, nurses, genetic counselors and researchers has received NIH funding to study this group of conditions.
Dr. Gropman says the current urea cycle research program builds on a sturdy foundation built by previous principal investigators Mendel Tuchman, M.D., and Mark Batshaw, M.D., also funded by the NIH. While previous rounds of NIH funding powered research about patients’ long-term survival prospects and cognitive dysfunction, this next phase of research will explore patients’ long-term health.
Among the topics they will study:
Long-term organ damage. Magnetic resonance elastrography (MRE) is a state-of-the-art imaging technique that combines the sharp images from MRI with a visual map that shows body tissue stiffness. The research team will use MRE to look for early changes in the liver – before patients show any symptoms – that could be associated with long-term health impacts. Their aim is spot the earliest signs of potential liver dysfunction in order to intervene before the patient develops liver fibrosis.
Academic achievement. The research team will examine gaps in academic achievement for patients who appear to be underperforming to determine what is triggering the discrepancy between their potential and actual scholastics. If they uncover issues such as learning difficulties or mental health concerns like anxiety, there are opportunities to intervene to boost academic achievement.
“And if we find many of the patients meet the criteria for depression or anxiety disorders, there are potential opportunities to intervene. It’s tricky: We need to balance their existing medications with any new ones to ensure that we don’t increase their hyperammonemia risk,” Dr. Gropman explains.
Neurologic complications. The researchers will tap continuous, bedside electroencephalogram, which measures the brain’s electrical activity, to detect silent seizures and otherwise undetectable changes in the brain in an effort to stave off epilepsy, a brain disorder that causes seizures.
“This is really the first time we will examine babies’ brains,” she adds. “Our previous imaging studies looked at kids and adults who were 6 years and older. Now, we’re lowering that age range down to infants. By tracking such images over time, the field has described the trajectory of what normal brain development should look like. We can use that as a background and comparison point.”
In the future, newborns may be screened for urea cycle disorder shortly after birth. Because it is not possible to diagnose it in the womb in cases where there is no family history, the team aims to better counsel families contemplating pregnancy about their possible risks.
Research described in this post was underwritten by the NIH through its Rare Diseases Clinical Research Network.
Preserving brain function by purposely inducing strokes
Born to young parents, no prenatal testing had suggested any problems with Bella’s brain. But just a few hours after birth, Bella suffered her first seizure – one of many that would follow in the ensuing days. After brain imaging, her doctors in Iowa diagnosed her with hemimegalencephaly.
Strokes are neurologically devastating events, cutting off life-sustaining oxygen to regions of the brain. If these brain tissues are deprived of oxygen long enough, they die, leading to critical loss of function – and sometimes loss of life.
“As physicians, we’re taught to prevent or treat stroke. We’re never taught to inflict it,” says Taeun Chang, M.D., director of the Neonatal Neurology and Neonatal Neurocritical Care Program at Children’s National Hospital.
That’s why a treatment developed at Children’s National for a rare brain condition called hemimegalencephaly is so surprising, Dr. Chang explains. By inflicting controlled, targeted strokes, Children’s National physician-researchers have treated five newborns born with intractable seizures due to hemimegalencephaly before they’re eligible for epilepsy surgery, the standard of care. In the four surviving infants, the procedures drastically reduced or completely relieved the infants of hemimegalencephaly’s characteristic, uncontrollable seizures.
The most recent patient to receive this life-changing procedure is Bella, a 13-month-old from Iowa whose treatment at Children’s National began within her second week of life. Born to young parents, no prenatal testing had suggested any problems with Bella’s brain. But just a few hours after birth, Bella suffered her first seizure – one of many that would follow in the ensuing days. After brain imaging, her doctors in Iowa diagnosed her with hemimegalencephaly.
A congenital condition occurring in just a handful of children born worldwide each year, hemimegalencephaly is marked by one brain hemisphere growing strikingly larger and dysplastic than the other, Dr. Chang explains. This abnormal half of the brain is highly vascularized, rippled with blood vessels needed to support the seizing brain. The most conspicuous symptoms of hemimegalencephaly are the numerous seizures that it causes, sometimes several in the course of an hour, which also may prevent the normal half of the brain from developing and learning.
Prior studies suggest early surgery achieves better developmental outcomes with one study reporting as much as a drop of 10-20 IQ points with every month delay in epilepsy surgery.
The standard treatment for unilateral megalencephaly is a dramatic procedure called a hemispherectomy, in which surgeons remove and disconnect the affected half of the brain, allowing the remaining half to take over its neurological duties. However, Dr. Chang says, implementing this procedure in infants younger than 3 months of age is highly dangerous. Excessive, potentially fatal blood loss is likely in infants younger than 3 months who have a highly vascularized brain in the setting of an immature coagulation system. That leaves their doctors with no choice but to wait until these infants are at least 3 months old, when they are more likely to survive the surgery.
However, five years ago, Dr. Chang and her colleagues came up with a different idea when a newborn continued to have several seizures per hour despite multiple IV seizure medications: Because strokes cause irreversible tissue death, it might be possible to effectively incapacitate the enlarged hemisphere from within by inflicting a stroke on purpose. At the very least, this “functional embolization” might buy time for a traditional hemispherectomy, and slow or halt ongoing brain damage until the infants are able to withstand surgery. Ideally, this procedure may be all some children need, knocking out the offending hemisphere completely so they’d never need a hemispherectomy, which has late complications, such as hydrocephalus.
A pediatrician friend of Bella’s paternal grandparents read a story on Children’s National website about Darcy, another baby who’d received functional embolization a year earlier and was doing well. She contacted Dr. Chang to see if the procedure would be appropriate for Bella.
Within days, Bella and her family headed to Washington, D.C., to prepare for functional embolization herself. Within the first weeks of life, Bella underwent three separate procedures, each three to four hours long. Under real-time fluoroscopic and angiographic guidance, interventional neuroradiologist Monica Pearl, M.D., threaded a micro-catheter up from the baby’s femoral artery through the complex network of blood vessels all the way to her brain. There, in targeted branches of her cerebral arteries, Dr. Pearl strategically placed liquid embolic agent to obstruct blood flow to the abnormal half of Bella’s brain.
Immediately after the first procedure, the team had to contend with the same consequences that come after any stroke: brain swelling that can cause bleeding and herniation, complicated further by the already enlarged hemisphere of Bella’s brain. Using neuroprotective strategies learned from treating hundreds of brain-injured newborns, the neonatal neurocritical care team and the neonatal intensive care unit (NICU) minimized the brain swelling and protected the normal half of the brain by tightly controlling the brain temperature, her sugar and electrolyte levels, her blood pressure and coagulation system.
As the brain tissue in the oversized hemisphere died, so did the seizures that had plagued Bella since birth. She has not had a seizure since she left Children’s National more than one year ago. Her adoptive parents report that Bella is hitting many of the typical developmental milestones for her age: She’s getting ready to walk, blowing kisses and saying a few words. Physical, speech and occupational therapy will keep her moving in the right direction, Dr. Chang says.
“We believe that Children’s National is the only place in the world that’s treating newborns in this way to preserve their futures,” Dr. Chang says. “We’re privileged to be able to care for Bella and other kids with this rare condition.”
Bella’s transfer and successful procedures required the support and collective efforts of many within the hospital organization including William D. Gaillard, M.D., and his surgical epilepsy team; interventional neuroradiology with Dr. Monica Pearl; Neurosurgery; Neonatology and the NICU; social work; and even approval from Robin Steinhorn, M.D., senior vice president of the Center for Hospital-Based Specialties, and David Wessel, M.D., executive vice president and Chief Medical Officer.
“While obvious credit goes to the medical team who saved Bella’s future and the neonatal intensive care nurses who provided exceptional, intensive, one-on-one care, Bella’s team of supporters extend to all levels within our hospital,” Dr. Chang adds.
Also read:
“Liquid biopsies” could track diffuse midline gliomas
A multi-institutional team led by researchers at Children’s National in Washington, D.C., developed and tested “liquid biopsy,” a measure of circulating tumor DNA in patients’ cerebrospinal fluid and blood plasma. They show that quantifying the amount of circulating tumor DNA possessing key mutations characteristic of diffuse midline gliomas could reliably predict the tumors’ response to radiotherapy.
Diffuse midline gliomas are rare, diagnosed in fewer than 800 Americans every year, the majority of whom are children. These cancers arise in the cellular “glue” that holds the brain and spinal cord’s neurons together, grow swiftly and have no cure. About half of patients with these cancers, including diffuse intrinsic pontine glioma, die within one year of diagnosis.
Clinical trials are increasingly investigating new treatments that could offer hope for patients and their families. Yet, thus far, there have been few ways to track the progression of these conditions, offering little insight on whether a treatment is hitting its intended goal.
To solve this problem, a multi-institutional team led by researchers at Children’s National in Washington, D.C., developed and tested “liquid biopsy,” a measure of circulating tumor DNA in patients’ cerebrospinal fluid and blood plasma. They show that quantifying the amount of circulating tumor DNA possessing key mutations characteristic of these cancers could reliably predict the tumors’ response to radiotherapy. The scientists published their results online Oct. 15, 2018, in Clinical Cancer Research.
“We heard from our clinician colleagues that many kids were coming in and their magnetic resonance imaging (MRI) suggested a particular type of tumor. But it was always problematic to identify the tumor’s molecular subtype,” says Javad Nazarian, Ph.D., MSC, a principal investigator in Children’s Center for Genetic Medicine Research. “Our colleagues wanted a more accurate measure than MRI to find the molecular subtype. That raised the question of whether we could actually look at their blood to determine the tumor subtype.”
Children’s liquid biopsy, which remains at the research phase, starts with a simple blood draw using the same type of needle as is used when people donate blood. When patients with brain tumors provide blood for other laboratory testing, a portion of it is used for the DNA detective work. Just as a criminal leaves behind fingerprints, tumors shed telltale clues in the blood. The team at Children’s National searches for the histone 3K27M (H3K27M), a mutation associated with worse clinical outcomes.
“With liquid biopsy, we were able to detect a few copies of tumor DNA that were hiding behind a million copies of healthy DNA,” Nazarian says. “The blood draw and liquid biopsy complement the MRI. The MRI gives the brain tumor’s ZIP code. Liquid biopsy gives you the demographics within that ZIP code.”
Working with collaborators around the nation, Children’s National continues to refine the technology to improve its accuracy.
Even though this research technique is in its infancy, the rapid, cheap and sensitive technology already is being used by people around the globe.
“People around the world are sending blood to us, looking for this particular mutation, H3K27M,” says Lindsay B. Kilburn, M.D., a neuro–oncologist, principal investigator at Children’s National for the Pacific Pediatric Neuro-Oncology Consortium, and study co-author. “In many countries or centers children to not have access to teams experienced in taking a biopsy of tumors in the brainstem, they can perform a simple blood draw and have that blood processed and analyzed by us. In only a few days, we can provide important molecular information on the tumor subtype previously only available to patients who had undergone a tumor biopsy.”
With that DNA finding, physicians can make more educated therapeutic decisions, including prescribing medications that could not have been given previously, Nazarian adds.
In addition to Nazarian and Dr. Kilburn, study co-authors include Eshini Panditharatna, Madhuri Kambhampati, Heather Gordish-Dressman, Ph.D., Suresh N. Magge, M.D., John S. Myseros, M.D., Eugene I. Hwang, M.D., and Roger J. Packer, M.D., all of Children’s National; Mariam S. Aboian, Nalin Gupta, Soonmee Cha, Michael Prados and Co-Senior Author Sabine Mueller, all of University of California, San Francisco; Cassie Kline, UCSF Benioff Children’s Hospital; John R. Crawford, UC San Diego; Katherine E. Warren, National Cancer Institute; Winnie S. Liang and Michael E. Berens, Translational Genomics Research Institute; and Adam C. Resnick, Children’s Hospital of Philadelphia.
Financial support for the research described in the report was provided by the V Foundation for Cancer Research, Goldwin Foundation, Pediatric Brain Tumor Foundation, Smashing Walnuts Foundation, The Gabriella Miller Kids First Data Resource Center, Zickler Family Foundation, Clinical and Translational Science Institute at Children’s National under award 5UL1TR001876-03, Piedmont Community Foundation, Musella Foundation for Brain Tumor Research, Mathew Larson Foundation, The Lilabean Foundation for Pediatric Brain Cancer Research, The Childhood Brain Tumor Foundation, the National Institutes of Health and American Society of Neuroradiology.
Neuroimaging essential for Zika cases
About three years ago, Zika virus emerged as a newly recognized congenital infection, and a growing body of research indicates the damage it causes differs from other infections that occur in utero.
Seventy-one of 110 Brazilian infants at the highest risk for experiencing problems due to exposure to the Zika virus in the womb experienced a wide spectrum of brain abnormalities, including calcifications and malformations in cortical development, according to a study published July 31, 2019 in JAMA Network Open.
The infants were born at the height of Brazil’s Zika epidemic, a few months after the nation declared a national public health emergency. Already, many of the infants had been classified as having the severe form of congenital Zika syndrome, and many had microcephaly, fetal brain disruption sequence, arthrogryposis and abnormal neurologic exams at birth.
These 110 infants “represented a group of ZIKV-exposed infants who would be expected to have a high burden of neuroimaging abnormalities, which is a difference from other reported cohorts,” Sarah B. Mulkey, M.D., Ph.D., writes in an invited commentary published in JAMA Network Open that accompanies the Rio de Janeiro study. “Fortunately, many ZIKV-exposed infants do not have abnormal brain findings or a clinical phenotype associated with congenital Zika syndrome,” adds Dr. Mulkey, a fetal–neonatal neurologist in the Division of Fetal and Transitional Medicine at Children’s National in Washington, D.C.
Indeed, a retrospective cohort of 82 women exposed to Zika during their pregnancies led by a research team at Children’s National found only three pregnancies were complicated by severe fetal brain abnormalities. Compared with the 65% abnormal computed tomography (CT) or magnetic resonance imaging (MRI) findings in the new Brazilian study, about 1 in 10 (10%) of babies born to women living in the continental U.S. with confirmed Zika infections during pregnancy had Zika-associated birth defects, according to the Centers for Disease Control and Prevention.
“There appears to be a spectrum of brain imaging abnormalities in ZIKV-exposed infants, including mild, nonspecific changes seen at cranial US [ultrasound], such as lenticulostriate vasculopathy and germinolytic cysts, to more significant brain abnormalities, such as subcortical calcifications, ventriculomegaly and, in its most severe form, thin cortical mantle and fetal brain disruption sequence,” Dr. Mulkey writes.
About three years ago, Zika virus emerged as a newly recognized congenital infection, and a growing body of research indicates the damage it causes differs from other infections that occur in utero. Unlike congenital cytomegalovirus infection, cerebral calcifications associated with Zika are typically subcortical, Dr. Mulkey indicates. What’s more, fetal brain disruption sequence seen in Zika-exposed infants is unusual for other infections that can cause microcephaly.
“Centered on the findings of Pool, et al, and others, early neuroimaging remains one of the most valuable investigations of the Zika-exposed infant,” Dr. Mulkey writes, including infants who are not diagnosed with congenital Zika syndrome. She recommends:
- Cranial ultrasound as the first-line imaging option for infants, if available, combined with neurologic and ophthalmologic exams, and brainstem auditory evoked potentials
- Zika-exposed infants with normal cranial ultrasounds do not need additional imaging unless they experience a developmental disturbance
- Zika-exposed infants with abnormal cranial ultrasounds should undergo further neuroimaging with low-dose cranial CT or brain MRI.
Focusing on the “little brain” to rescue cognition
Research faculty at Children’s National in Washington, D.C., with colleagues recently published a review article in Nature Reviews Neuroscience that covers the latest research about how abnormal development of the cerebellum leads to a variety of neurodevelopmental disorders.
Cerebellum translates as “little brain” in Latin. This piece of anatomy – that appears almost separate from the rest of the brain, tucked under the two cerebral hemispheres – long has been known to play a pivotal role in voluntary motor functions, such as walking or reaching for objects, as well as involuntary ones, such as maintaining posture.
But more recently, says Aaron Sathyanesan, Ph.D., a postdoctoral research fellow at the Children’s Research Institute, the research arm of Children’s National in Washington, D.C., researchers have discovered that the cerebellum is also critically important for a variety of non-motor functions, including cognition and emotion.
Sathyanesan, who studies this brain region in the laboratory of Vittorio Gallo, Ph.D., Chief Research Officer at Children’s National and scientific director of the Children’s Research Institute, recently published a review article with colleagues in Nature Reviews Neuroscience covering the latest research about how altered development of the cerebellum contributes to a variety of neurodevelopmental disorders.
These disorders, he explains, are marked by problems in the nervous system that arise while it’s maturing, leading to effects on emotion, learning ability, self-control, or memory, or any combination of these. They include diagnoses as diverse as intellectual disability, autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder and Down syndrome.
“One reason why the cerebellum might be critically involved in each of these disorders,” Sathyanesan says, “is because its developmental trajectory takes so long.”
Unlike other brain structures, which have relatively short windows of development spanning weeks or months, the principal cells of the cerebellum – known as Purkinje cells – start to differentiate from stem cell precursors at the beginning of the seventh gestational week, with new cells continuing to appear until babies are nearly one year old. In contrast, cells in the neocortex, a part of the brain involved in higher-order brain functions such as cognition, sensory perception and language is mostly finished forming while fetuses are still gestating in the womb.
This long window for maturation allows the cerebellum to make connections with other regions throughout the brain, such as extensive connections with the cerebral cortex, the outer layer of the cerebrum that plays a key role in perception, attention, awareness, thought, memory, language and consciousness. It also allows ample time for things to go wrong.
“Together,” Sathyanesan says, “these two characteristics are at the root of the cerebellum’s involvement in a host of neurodevelopmental disorders.”
For example, the review article notes, researchers have discovered both structural and functional abnormalities in the cerebellums of patients with ASD. Functional magnetic resonance imaging (MRI), an imaging technique that measures activity in different parts of the brain, suggests that significant differences exist between connectivity between the cerebellum and cortex in people with ASD compared with neurotypical individuals. Differences in cerebellar connectivity are also evident in resting-state functional connectivity MRI, an imaging technique that measures brain activity in subjects when they are not performing a specific task. Some of these differences appear to involve patterns of overconnectivity to different brain regions, explains Sathyanesan; other differences suggest that the cerebellums of patients with ASD don’t have enough connections to other brain regions.
These findings could clarify research from Children’s National and elsewhere that has shown that babies born prematurely often sustain cerebellar injuries due to multiple hits, including a lack of oxygen supplied by infants’ immature lungs, he adds. Besides having a sibling with ASD, premature birth is the most prevalent risk factor for an ASD diagnosis.
The review also notes that researchers have discovered structural changes in the cerebellums of patients with Down syndrome, who tend to have smaller cerebellar volumes than neurotypical individuals. Experimental models of this trisomy recapitulate this difference, along with abnormal connectivity to the cerebral cortex and other brain regions.
Although the cerebellum is a pivotal contributor toward these conditions, Sathyanesan says, learning more about this brain region helps make it an important target for treating these neurodevelopmental disorders. For example, he says, researchers are investigating whether problems with the cerebellum and abnormal connectivity could be lessened through a non-invasive form of brain stimulation called transcranial direct current stimulation or an invasive one known as deep brain stimulation. Similarly, a variety of existing pharmaceuticals or new ones in development could modify the cerebellum’s biochemistry and, consequently, its function.
“If we can rescue the cerebellum’s normal activity in these disorders, we may be able to alleviate the problems with cognition that pervade them all,” he says.
In addition to Sathyanesan and Senior Author Gallo, Children’s National study co-authors include Joseph Scafidi, D.O., neonatal neurologist; Joy Zhou and Roy V. Sillitoe, Baylor College of Medicine; and Detlef H. Heck, of University of Tennessee Health Science Center.
Financial support for research described in this post was provided by the National Institute of Neurological Disorders and Stroke under grant numbers 5R01NS099461, R01NS089664, R01NS100874, R01NS105138 and R37NS109478; the Hamill Foundation; the Baylor College of Medicine Intellectual and Developmental Disabilities Research Center under grant number U54HD083092; the University of Tennessee Health Science Center (UTHSC) Neuroscience Institute; the UTHSC Cornet Award; the National Institute of Mental Health under grant number R01MH112143; and the District of Columbia Intellectual and Developmental Disabilities Research Center under grant number U54 HD090257.
Early lipids in micropreemies’ diets can boost brain growth
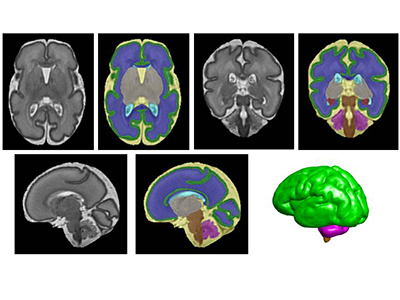
Segmentation of a preterm brain T2-weighted MRI image at 30 gestational weeks [green=cortical grey matter; blue=white matter; grey=deep grey matter; cyan=lateral ventricle; purple=cerebellum; orange=brainstem; red=hippocampus; yellow=cerebrospinal fluid].
“Compared with macronutrients like carbohydrates and proteins, lipid intake during the first month of life is associated with increased overall and regional brain volume for micro-preemies,” says Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain at Children’s National and senior author. “Using non-invasive magnetic resonance imaging, we see increased volume in the cerebellum by 2 weeks of age. And at four weeks of life, lipids increase total brain volume and boost regional brain volume in the cerebellum, amygdala-hippocampus and brainstem.”
The cerebellum is involved in virtually all physical movement and enables coordination and balance. The amygdala processes and stores short-term memories. The hippocampus manages emotion and mood. And the brainstem acts like a router, passing messages from the brain to the rest of the body, as well as enabling essential functions like breathing, a steady heart rate and swallowing.
According to the Centers for Disease Control and Prevention, about 1 in 10 U.S. babies is born preterm, or before 37 weeks gestation. Regions of the brain that play vital roles in complex cognitive and motor activities experience exponential growth late in pregnancy, making the developing brains of preterm infants particularly vulnerable to injury and impaired growth.
Children’s research faculty examined the impact of lipid intake in the first month of life on brain volumes for very low birth weight infants, who weighed 1,500 grams or less at birth. These micro-preemies are especially vulnerable to growth failure and neurocognitive impairment after birth.
The team enrolled 68 micro-preemies who were 32 weeks gestational age and younger when they were admitted to Children’s neonatal intensive care unit during their first week of life. They measured cumulative macronutrients – carbohydrates, proteins, lipids and calories – consumed by these newborns at 2 and 4 weeks of life. Over years, Limperopoulos’ lab has amassed a large database of babies who were born full-term; this data provides unprecedented insights into normal brain development and will help to advance understanding of brain development in high-risk preterm infants.
“Even after controlling for average weight gain and other health conditions, lipid intake was positively associated with cerebellar and brainstem volumes in very low birthweight preterm infants,” adds Katherine M. Ottolini, the study’s lead author.
According to Limperopoulos, Children’s future research will examine the optimal timing and volume of lipids to boost neurodevelopment for micro-preemies.
Pediatric Academic Societies 2019 Annual Meeting presentation
- “Early lipid intake improves brain growth in premature infants.”
- Saturday, April 27, 2019, 1:15-2:30 p.m. (EST)
Katherine M. Ottolini, lead author; Nickie Andescavage, M.D., Attending, Neonatal-Perinatal Medicine and co-author; Kushal Kapse, research and development staff engineer and co-author; and Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain and senior author, all of Children’s National.
Breastfeeding boosts metabolites important for brain growth

“Proton magnetic resonance spectroscopy, a non-invasive imaging technique that describes the chemical composition of specific brain structures, enables us to measure metabolites that may play a critical role for growth and explain what makes breastfeeding beneficial for newborns’ developing brains,” says Catherine Limperopoulos, Ph.D.
Micro-preemies who primarily consume breast milk have significantly higher levels of metabolites important for brain growth and development, according to sophisticated imaging conducted by an interdisciplinary research team at Children’s National.
“Our previous research established that vulnerable preterm infants who are fed breast milk early in life have improved brain growth and neurodevelopmental outcomes. It was unclear what makes breastfeeding so beneficial for newborns’ developing brains,” says Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain at Children’s National. “Proton magnetic resonance spectroscopy, a non-invasive imaging technique that describes the chemical composition of specific brain structures, enables us to measure metabolites essential for growth and answer that lingering question.”
According to the Centers for Disease Control and Prevention, about 1 in 10 U.S. infants is born preterm. The Children’s research team presented their findings during the Pediatric Academic Societies 2019 Annual Meeting.
The research-clinicians enrolled babies who were very low birthweight (less than 1,500 grams) and 32 weeks gestational age or younger at birth when they were admitted to Children’s neonatal intensive care unit in the first week of life. The team gathered data from the right frontal white matter and the cerebellum – a brain region that enables people to maintain balance and proper muscle coordination and that supports high-order cognitive functions.
Each chemical has its own a unique spectral fingerprint. The team generated light signatures for key metabolites and calculated the quantity of each metabolite. Of note:
- Cerebral white matter spectra showed significantly greater levels of inositol (a molecule similar to glucose) for babies fed breast milk, compared with babies fed formula.
- Cerebellar spectra had significantly greater creatine levels for breastfed babies compared with infants fed formula.
- And the percentage of days infants were fed breast milk was associated with significantly greater levels of both creatine and choline, a water soluble nutrient.
“Key metabolite levels ramp up during the times babies’ brains experience exponential growth,” says Katherine M. Ottolini, the study’s lead author. “Creatine facilitates recycling of ATP, the cell’s energy currency. Seeing greater quantities of this metabolite denotes more rapid changes and higher cellular maturation. Choline is a marker of cell membrane turnover; when new cells are generated, we see choline levels rise.”
Already, Children’s National leverages an array of imaging options that describe normal brain growth, which makes it easier to spot when fetal or neonatal brain development goes awry, enabling earlier intervention and more effective treatment. “Proton magnetic resonance spectroscopy may serve as an important additional tool to advance our understanding of how breastfeeding boosts neurodevelopment for preterm infants,” Limperopoulos adds.
Pediatric Academic Societies 2019 Annual Meeting presentation
- “Improved cerebral and cerebellar metabolism in breast milk-fed VLBW infants.”
- Monday, April 29, 2019, 3:30–3:45 p.m. (EST)
Katherine M. Ottolini, lead author; Nickie Andescavage, M.D., Attending, Neonatal-Perinatal Medicine and co-author; Kushal Kapse, research and development staff engineer and co-author; Sudeepta Basu, M.D., neonatologist and co-author; and Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain and senior author, all of Children’s National.
Research and Education Week honors innovative science

Billie Lou Short, M.D., received the Ninth Annual Mentorship Award in Clinical Science.
People joke that Billie Lou Short, M.D., chief of Children’s Division of Neonatology, invented extracorporeal membrane oxygenation, known as ECMO for short. While Dr. Short did not invent ECMO, under her leadership Children’s National was the first pediatric hospital to use it. And over decades Children’s staff have perfected its use to save the lives of tiny, vulnerable newborns by temporarily taking over for their struggling hearts and lungs. For two consecutive years, Children’s neonatal intensive care unit has been named the nation’s No. 1 for newborns by U.S. News & World Report. “Despite all of these accomplishments, Dr. Short’s best legacy is what she has done as a mentor to countless trainees, nurses and faculty she’s touched during their careers. She touches every type of clinical staff member who has come through our neonatal intensive care unit,” says An Massaro, M.D., director of residency research.
For these achievements, Dr. Short received the Ninth Annual Mentorship Award in Clinical Science.
Anna Penn, M.D., Ph.D., has provided new insights into the central role that the placental hormone allopregnanolone plays in orderly fetal brain development, and her research team has created novel experimental models that mimic some of the brain injuries often seen in very preterm babies – an essential step that informs future neuroprotective strategies. Dr. Penn, a clinical neonatologist and developmental neuroscientist, “has been a primary adviser for 40 mentees throughout their careers and embodies Children’s core values of Compassion, Commitment and Connection,” says Claire-Marie Vacher, Ph.D.
For these achievements, Dr. Penn was selected to receive the Ninth Annual Mentorship Award in Basic and Translational Science.
The mentorship awards for Drs. Short and Penn were among dozens of honors given in conjunction with “Frontiers in Innovation,” the Ninth Annual Research and Education Week (REW) at Children’s National. In addition to seven keynote lectures, more than 350 posters were submitted from researchers – from high-school students to full-time faculty – about basic and translational science, clinical research, community-based research, education, training and quality improvement; five poster presenters were showcased via Facebook Live events hosted by Children’s Hospital Foundation.
Two faculty members won twice: Vicki Freedenberg, Ph.D., APRN, for research about mindfulness-based stress reduction and Adeline (Wei Li) Koay, MBBS, MSc, for research related to HIV. So many women at every stage of their research careers took to the stage to accept honors that Naomi L.C. Luban, M.D., Vice Chair of Academic Affairs, quipped that “this day is power to women.”
Here are the 2019 REW award winners:
2019 Elda Y. Arce Teaching Scholars Award
Barbara Jantausch, M.D.
Lowell Frank, M.D.
Suzanne Feetham, Ph.D., FAA, Nursing Research Support Award
Vicki Freedenberg, Ph.D., APRN, for “Psychosocial and biological effects of mindfulness-based stress reduction intervention in adolescents with CHD/CIEDs: a randomized control trial”
Renee’ Roberts Turner for “Peak and nadir experiences of mid-level nurse leaders”
2019-2020 Global Health Initiative Exploration in Global Health Awards
Nathalie Quion, M.D., for “Latino youth and families need assessment,” conducted in Washington
Sonia Voleti for “Handheld ultrasound machine task shifting,” conducted in Micronesia
Tania Ahluwalia, M.D., for “Simulation curriculum for emergency medicine,” conducted in India
Yvonne Yui for “Designated resuscitation teams in NICUs,” conducted in Ghana
Xiaoyan Song, Ph.D., MBBS, MSc, “Prevention of hospital-onset infections in PICUs,” conducted in China
Ninth Annual Research and Education Week Poster Session Awards
Basic and Translational Science
Faculty: Adeline (Wei Li) Koay, MBBS, MSc, for “Differences in the gut microbiome of HIV-infected versus HIV-exposed, uninfected infants”
Faculty: Hayk Barseghyan, Ph.D., for “Composite de novo Armenian human genome assembly and haplotyping via optical mapping and ultra-long read sequencing”
Staff: Damon K. McCullough, BS, for “Brain slicer: 3D-printed tissue processing tool for pediatric neuroscience research”
Staff: Antonio R. Porras, Ph.D., for “Integrated deep-learning method for genetic syndrome screening using facial photographs”
Post docs/fellows/residents: Lung Lau, M.D., for “A novel, sprayable and bio-absorbable sealant for wound dressings”
Post docs/fellows/residents: Kelsey F. Sugrue, Ph.D., for “HECTD1 is required for growth of the myocardium secondary to placental insufficiency”
Graduate students: Erin R. Bonner, BA, for “Comprehensive mutation profiling of pediatric diffuse midline gliomas using liquid biopsy”
High school/undergraduate students: Ali Sarhan for “Parental somato-gonadal mosaic genetic variants are a source of recurrent risk for de novo disorders and parental health concerns: a systematic review of the literature and meta-analysis”
Clinical Research
Faculty: Amy Hont, M.D., for “Ex vivo expanded multi-tumor antigen specific T-cells for the treatment of solid tumors”
Faculty: Lauren McLaughlin, M.D., for “EBV/LMP-specific T-cells maintain remissions of T- and B-cell EBV lymphomas after allogeneic bone marrow transplantation”
Staff: Iman A. Abdikarim, BA, for “Timing of allergenic food introduction among African American and Caucasian children with food allergy in the FORWARD study”
Staff: Gelina M. Sani, BS, for “Quantifying hematopoietic stem cells towards in utero gene therapy for treatment of sickle cell disease in fetal cord blood”
Post docs/fellows/residents: Amy H. Jones, M.D., for “To trach or not trach: exploration of parental conflict, regret and impacts on quality of life in tracheostomy decision-making”
Graduate students: Alyssa Dewyer, BS, for “Telemedicine support of cardiac care in Northern Uganda: leveraging hand-held echocardiography and task-shifting”
Graduate students: Natalie Pudalov, BA, “Cortical thickness asymmetries in MRI-abnormal pediatric epilepsy patients: a potential metric for surgery outcome”
High school/undergraduate students: Kia Yoshinaga for “Time to rhythm detection during pediatric cardiac arrest in a pediatric emergency department”
Community-Based Research
Faculty: Adeline (Wei Li) Koay, MBBS, MSc, for “Recent trends in the prevention of mother-to-child transmission (PMTCT) of HIV in the Washington, D.C., metropolitan area”
Staff: Gia M. Badolato, MPH, for “STI screening in an urban ED based on chief complaint”
Post docs/fellows/residents: Christina P. Ho, M.D., for “Pediatric urinary tract infection resistance patterns in the Washington, D.C., metropolitan area”
Graduate students: Noushine Sadeghi, BS, “Racial/ethnic disparities in receipt of sexual health services among adolescent females”
Education, Training and Program Development
Faculty: Cara Lichtenstein, M.D., MPH, for “Using a community bus trip to increase knowledge of health disparities”
Staff: Iana Y. Clarence, MPH, for “TEACHing residents to address child poverty: an innovative multimodal curriculum”
Post docs/fellows/residents: Johanna Kaufman, M.D., for “Inpatient consultation in pediatrics: a learning tool to improve communication”
High school/undergraduate students: Brett E. Pearson for “Analysis of unanticipated problems in CNMC human subjects research studies and implications for process improvement”
Quality and Performance Improvement
Faculty: Vicki Freedenberg, Ph.D., APRN, for “Implementing a mindfulness-based stress reduction curriculum in a congenital heart disease program”
Staff: Caleb Griffith, MPH, for “Assessing the sustainability of point-of-care HIV screening of adolescents in pediatric emergency departments”
Post docs/fellows/residents: Rebecca S. Zee, M.D., Ph.D., for “Implementation of the Accelerated Care of Torsion (ACT) pathway: a quality improvement initiative for testicular torsion”
Graduate students: Alysia Wiener, BS, for “Latency period in image-guided needle bone biopsy in children: a single center experience”
To understand the preterm brain, start with the fetal brain

“My best advice to future clinician-scientists is to stay curious and open-minded; I doubt I could have predicted my current research interest or described the path between the study of early oligodendrocyte maturation to in vivo placental development, but each experience along the way – both academic and clinical – has led me to where I am today,” Nickie Andescavage, M.D., writes.
Too often, medical institutions erect an artificial boundary between caring for the developing fetus inside the womb and caring for the newborn whose critical brain development continues outside the womb.
“To improve neonatal outcomes, we must transform our current clinical paradigms to begin treatment in the intrauterine period and continue care through the perinatal transition through strong collaborations with obstetricians and fetal-medicine specialists,” writes Nickie Andescavage, M.D., an attending in Neonatal-Perinatal Medicine at Children’s National.
Dr. Andescavage’s commentary was published online March 25, 2019, in Pediatrics Research and accompanies recently published Children’s research about differences in placental development in the setting of placental insufficiency. Her commentary is part of a new effort by Nature Publishing Group to spotlight research contributions from early career investigators.
The placenta, an organ shared by a pregnant woman and the developing fetus, plays a critical but underappreciated role in the infant’s overall health. Under the mentorship of Catherine Limperopoulos, Ph.D., director of MRI Research of the Developing Brain, and Adré J. du Plessis, M.B.Ch.B., MPH, chief of the Division of Fetal and Transitional Medicine, Dr. Andescavage works with interdisciplinary research teams at Children’s National to help expand that evidence base.
While attending Cornell University as an undergraduate, Dr. Andescavage had an early interest in neuroscience and neurobehavior. As she continued her education by attending medical school at Columbia University, she corroborated an early instinct to work in pediatrics.
It wasn’t until the New Jersey native began pediatric residency at Children’s National that those complementary interests coalesced into a focus on brain autoregulation and autonomic function in full-term and preterm infants and imaging the brains of both groups. In normal, healthy babies the autonomic nervous system regulates heart rate, blood pressure, digestion, breathing and other involuntary activities. When these essential controls go awry, babies can struggle to survive and thrive.
“My best advice to future clinician-scientists is to stay curious and open-minded; I doubt I could have predicted my current research interest or described the path between the study of early oligodendrocyte maturation to in vivo placental development, but each experience along the way – both academic and clinical – has led me to where I am today,” Dr. Andescavage writes in the commentary.
Vote for Children’s National in STAT Madness

Children’s National Health System has been selected to compete in STAT Madness for the second consecutive year. Our entry for the bracket-style competition is “Sensitive liquid biopsy platform to detect tumor-released mutated DNA using patient blood and CSF,” a new technique that will allow kids to get better treatment for an aggressive type of pediatric brain tumor.
In 2018, Children’s first-ever STAT Madness entry advanced through five brackets in the national competition and, in the championship round, finished second. That innovation, which enables more timely diagnoses of rare diseases and common genetic disorders, helping to improve kids’ health outcomes around the world, also was among four “Editor’s Pick” finalists, entries that spanned a diverse range of scientific disciplines.
“Children’s National researchers collaboratively work across divisions and departments to ensure that innovations discovered in our laboratories reach clinicians in order to improve patient care,” says Mark Batshaw, M.D., Children’s Executive Vice President, Chief Academic Officer and Physician-in-Chief. “It’s gratifying that Children’s multidisciplinary approach to improving the lives of children with brain tumors has been included in this year’s STAT Madness competition.”
Pediatric brain cancers are the leading cause of cancer-related death in children younger than 14. Children with tumors in their midline brain structures have the worst outcomes, and kids diagnosed with diffuse midline gliomas, including diffuse intrinsic pontine glioma, have a median survival of just 12 months.
“We heard from our clinician colleagues that many kids were coming in and their magnetic resonance imaging (MRI) suggested a particular type of tumor. But it was always problematic to identify the tumor’s molecular subtype,” says Javad Nazarian, Ph.D., MSC, a principal investigator in Children’s Center for Genetic Medicine Research. “Our colleagues wanted a more accurate measure than MRI to find the molecular subtype. That raised the question of whether we could actually look at their blood to determine the tumor subtype.”
Children’s liquid biopsy, which remains at the research phase, starts with a simple blood draw using the same type of needle as is used when people donate blood. When patients with brain tumors provide blood for other laboratory testing, a portion of it is used for the DNA detective work. Just as a criminal leaves behind fingerprints, tumors shed telltale clues in the blood. The Children’s team searches for the histone 3.3K27M (H3K27M), a mutation associated with worse clinical outcomes.
“With liquid biopsy, we were able to detect a few copies of tumor DNA that were hiding behind a million copies of healthy DNA,” Nazarian says. “The blood draw and liquid biopsy complement the MRI. The MRI gives the brain tumor’s ZIP code. Liquid biopsy gives you the demographics within that ZIP code.”
Working with collaborators around the nation, Children’s National continues to refine the technology to improve its accuracy. The multi-institutional team published findings online Oct. 15, 2018, in Clinical Cancer Research.
Even though this research technique is in its infancy, the rapid, cheap and sensitive technology already is being used by people around the globe.
“People around the world are sending blood to us, looking for this particular mutation, H3K27M, ” says Lindsay B. Kilburn, M.D., a Children’s neuro–oncologist, principal investigator at Children’s National for the Pacific Pediatric Neuro-Oncology Consortium, and study co-author. “In many countries or centers, children do not have access to teams experienced in taking a biopsy of tumors in the brainstem, they can perform a simple blood draw and have that blood processed and analyzed by us. In only a few days, we can provide important molecular information on the tumor subtype previously only available to patients that had undergone a tumor biopsy.”
“With that DNA finding, physicians can make more educated therapeutic decisions, including prescribing medications that could not have been given previously,” Nazarian adds.
The STAT Madness round of 64 brackets opened March 4, 2019, and the championship round voting concludes April 5 at 5 p.m. (EST).
In addition to Nazarian and Dr. Kilburn, study co-authors include Eshini Panditharatna, Madhuri Kambhampati, Heather Gordish-Dressman, Ph.D., Suresh N. Magge, M.D., John S. Myseros, M.D., Eugene I. Hwang, M.D. and Roger J. Packer, M.D., all of Children’s National; Mariam S. Aboian, Nalin Gupta, Soonmee Cha, Michael Prados and Co-Senior Author Sabine Mueller, all of University of California, San Francisco; Cassie Kline, UCSF Benioff Children’s Hospital; John R. Crawford, UC San Diego; Katherine E. Warren, National Cancer Institute; Winnie S. Liang and Michael E. Berens, Translational Genomics Research Institute; and Adam C. Resnick, Children’s Hospital of Philadelphia.
Financial support for the research described in the report was provided by the V Foundation for Cancer Research, Goldwin Foundation, Pediatric Brain Tumor Foundation, Smashing Walnuts Foundation, The Gabriella Miller Kids First Data Resource Center, Zickler Family Foundation, Clinical and Translational Science Institute at Children’s National under award 5UL1TR001876-03, Piedmont Community Foundation, Musella Foundation for Brain Tumor Research, Matthew Larson Foundation, The Lilabean Foundation for Pediatric Brain Cancer Research, The Childhood Brain Tumor Foundation, the National Institutes of Health and American Society of Neuroradiology.
Understanding Legg-Calvé-Perthes disease
Legg-Calvé-Perthes disease, which affects between five and 10 of every 100,000 children each year, is so rare that it can sometimes be challenging for clinicians to know how best to care for affected patients.
That’s why in 2011 a group of pediatric orthopaedic specialists led by Texas Scottish Rite Children’s Hospital created an international study group dedicated to using research to improve the care of kids with Perthes, a hip disorder characterized by a loss of blood flow to the immature femoral head. Children’s National orthopaedic surgeon Benjamin Martin, M.D., has participated in the group since its launch.
Recently, Dr. Martin and two study group colleagues published a review study that outlines common imaging modalities used in the diagnosis and treatment of Perthes disease.
“There are many imaging options out there, including recent advances in MRI, that can add to our knowledge of the disease and how to treat it so kids have optimal outcomes,” Dr. Martin says. “Our goal was to review what’s out there, how it’s used, and identify any shortcomings of these approaches for this particular patient population.”
The authors note that imaging, in various forms, has been a crucial contributor to understanding and treatment of this disease since it was first discovered. Today, radiography remains the most common imaging technique used to diagnose and follow Perthes over time. However, some MRI applications may offer additional insight into the disorder.
Perfusion MRI allows for early understanding of extent of disease and perfusion patterns may correlate with outcomes. Diffusion weighted imaging (DWI) MRI is another promising avenue for tracking disease progression. Additionally, dynamic MRI might provide range of motion assessments that could be used in the surgical planning process.
This study was one of a handful that the international Perthes group has published so far, with several more currently under development. Exploring treatments and technology applications will enhance early diagnosis and treatment for Perthes, which is a crucial component of treatment success and improved quality of life for affected children.
MRI and ultrasound imaging detect the spectrum of Zika’s impact
“A combination of prenatal MRI and US was able to detect Zika-related brain abnormalities during pregnancy, giving families timely information to prepare for the potential complex care needs of these infants,” says Sarah B. Mulkey, M.D., Ph.D.
Worldwide, thousands of babies have been born to mothers who were infected during pregnancy with Zika, a virus associated with neurological deficits, impaired vision and neurodevelopmental disabilities, among other birth defects. These birth defects are sometimes severe, causing lifelong disability. But they’re also relatively rare compared with the overall rates of infection.
Predicting how many Zika-exposed babies would experience neurological birth defects has been challenging.
However, an international study led by Children’s faculty suggests that ultrasound (US) imaging performed during pregnancy and after childbirth revealed most Zika-related brain abnormalities experienced by infants exposed to the Zika virus during pregnancy, according to a prospective cohort study published online Nov. 26, 2018, in JAMA Pediatrics. Some Zika-exposed infants whose imaging had been normal during pregnancy had mild brain abnormalities detected by US and magnetic resonance imaging (MRI) after they were born.
“A combination of prenatal MRI and US was able to detect Zika-related brain abnormalities during pregnancy, giving families timely information to prepare for the potential complex care needs of these infants,” says Sarah B. Mulkey, M.D., Ph.D., a fetal-neonatal neurologist at Children’s National Health System and the study’s lead author. “In our study, we detected mild brain abnormalities on postnatal neuroimaging for babies whose imaging was normal during pregnancy. Therefore, it is important for clinicians to continue to monitor brain development for Zika-exposed infants after birth.”
As of Nov. 20 2018, nearly 2,500 pregnant women in the U.S. had laboratory confirmed Zika infection, and about 2,400 of them had given birth, according to the Centers for Disease Control and Prevention (CDC). While more than 100 U.S. infants were born with Zika-associated birth defects, the vast majority of Zika-exposed U.S. infants were apparently normal at birth. The sequential neuroimaging study Dr. Mulkey leads seeks to determine the spectrum of brain findings in infants exposed to Zika in the womb using both US and MRI before and after birth.
The international research team enrolled 82 women in the study from June 15, 2016, through June 27, 2017. All of the women had been exposed to Zika during pregnancy; all but one experienced clinical symptoms by a mean gestational age of 8.2 weeks. Eighty of those women lived in or near Barranquilla, Colombia, and were exposed to Zika there. Two U.S. study participants were exposed to the primarily mosquito-borne illness during travel to Zika hot zones.
All women received fetal MRIs and US during the second and/or third trimester of pregnancy. After their infants were born, the children received brain MRI and cranial US. Blood samples from both mothers and babies were tested for Zika using polymerase chain reaction and serology.
Fetal MRI was able to discern Zika-related brain damage as early as 18 weeks gestation and picked up significant fetal brain abnormalities not fully appreciated in US imaging. In one case, the US remained normal while fetal MRI alone detected brain abnormalities. Three fetuses (4 percent) had severe fetal brain abnormalities consistent with Zika infection, including:
- Two cases of heterotopias and malformations in cortical development, and
- One case of parietal encephalocele, Chiari II malformation and microcephaly.
Seventy-five infants were born at term. One pregnancy was terminated at 23 weeks gestation due to the gravity of the fetal brain abnormalities. One fetus with normal imaging died during pregnancy. One newborn who was born with significant fetal brain abnormalities died at age 3 days.
Cranial US and brain MRI was performed on the majority of infants whose prenatal imaging had been normal. Seven of 53 (13 percent) Zika-exposed infants had mild brain abnormalities detected by MRI after birth. In contrast, postnatal cranial US was better at detecting changes of lenticulostriate vasculopathy, cysts within the brain’s choroid plexus (cells that produce cerebrospinal fluid), germinolytic/subependymal cysts and/or calcifications, which were seen in 21 of 57 (37 percent) infants.
“Sequential neuroimaging revealed that the majority of Zika-exposed fetuses had normal brain development. Tragically, in a small number of pregnancies, Zika-related brain abnormalities were quite severe,” Dr. Mulkey adds. “Our data support the CDC’s recommendation that cranial US be performed after Zika-exposed babies are born. In addition, there is clearly a need to follow these babies over time to gauge whether the brain anomalies we see in imaging affects language, motor and social skills.”
Companion editorial: Revealing the effects of Zika
In addition to Dr. Mulkey, study co-authors include Dorothy I. Bulas, M.D., Gilbert Vezina, M.D., Margarita Arroyave-Wessel, MPH, Stephanie Russo, B.S, Youssef A. Kousa, D.O, Ph.D., Roberta L. DeBiasi, M.D., MS, Senior Author Adré J. du Plessis, M.B.Ch.B., MPH, all of Children’s National; Christopher Swisher, BS, Georgetown University and Caitlin Cristante, BS, Loyola University, both of whose contributions included research performed at Children’s National; Yamil Fourzali, M.D., Armando Morales, M.D., both of Sabbag Radiologos; Liliana Encinales, M.D., Allied Research Society; Nelly Pacheco, Bacteriologa, Bio-Nep; Robert S. Lanciotti, Ph.D., Arbovirus Diseases Branch, Centers for Disease Control and Prevention; and Carlos Cure, M.D., BIOMELAB.
Research reported in this news release was supported by the IKARIA fund.