Neighborhood disadvantage alters brain networks in unborn babies
 Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the latest edition of The Journal of Pediatrics, underscore the need to support young families from a baby’s earliest days.
Growing up in a disadvantaged neighborhood changes a child’s brain even before birth. These new findings, in the latest edition of The Journal of Pediatrics, underscore the need to support young families from a baby’s earliest days.
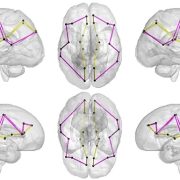
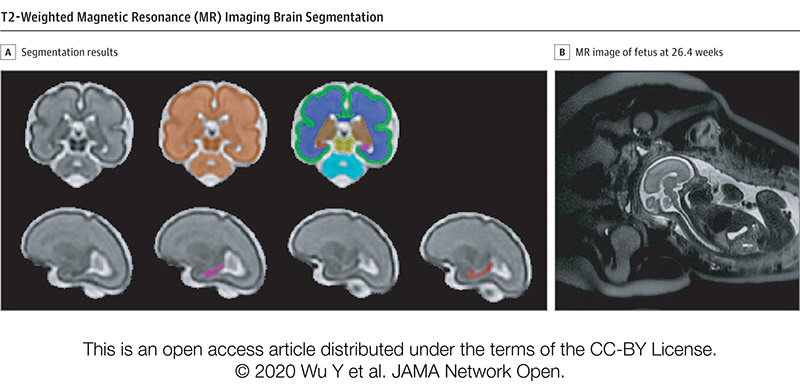
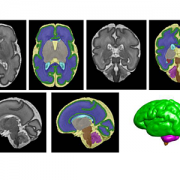
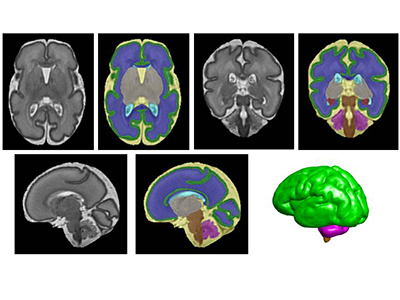
According to this new research from the Center for Prenatal, Neonatal & Maternal Health Research at Children’s National Hospital, exposure to neighborhood disadvantage lessens the functional integration of neural networks in the fetal brain, as seen on functional MRIs of healthy babies. The center compared the brains of 68 healthy babies on 79 scans. Researchers then mapped details about neural activity to a “social vulnerability index” from the Centers for Disease Control and Prevention, which indicates proximity to environmental stressors.
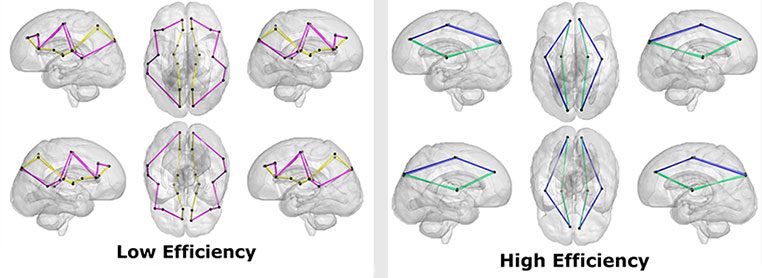
“We specifically looked at brain architecture to see how easily information flows between different regions,” said Kevin Cook, Ph.D., research faculty at the center and the manuscript’s first author. “To do this, we used graph theory, which borrows concepts from social network theory. It’s widely applied in computer science to understand how information flows within groups, and neuroscience has adapted it to study how information travels within the brain.”
What we found
Dr. Cook said researchers focused on the three metrics:
- Path length, which measures how many stops information needs to make along its way through the brain.
- Global efficiency, which measures the overall efficiency of the entire brain’s network.
- Small-world propensity, which describes how the brain’s network is organized and indicates how well the brain is organized into smaller, efficient networks.
As social vulnerability increased, the research team found global efficiency decreased, meaning the brain’s neural network was less efficient. The path lengths were also longer in children with greater neighborhood disadvantage, reinforcing the global efficiency findings.
The fine print
The under-development and over-development of fetal brains may contribute to neurological disorders, such as autism, epilepsy and other conditions of interest to researchers. Yet science’s understanding of how the brain matures in utero is still limited.
In this study, researchers found a notable difference related to age. At the youngest gestational ages, path lengths are longer, and both global efficiency and small world propensity are lower. As the fetus gets closer to term, path length and global efficiency show rapid maturation and less advantaged fetuses catch up to their peers who have greater advantages.
Researchers saw the same findings for small-world propensity, but the maturation didn’t stop. These unborn babies overshot their peers and had greater small-world propensity, suggesting their brains are divided into a greater number of smaller networks than their advantaged peers.
“We believe that length and global efficiency are on a trend to overshoot,” Dr. Cook said. “These findings are notable because they agree with what we know about older children and adolescents. Greater disadvantage is associated with hyper- or over-maturation of the brain. Our findings suggest that this may be starting even before birth.”
What’s ahead
While still early, this research improves the understanding of how environmental complexities can impact an unborn baby. Catherine Limperopoulos, Ph.D., director of the research center, which opened in 2023, said this work will be foundational as they continue to study the impact of a child’s environment on development.
“These findings have important implications for understanding how status and disadvantage may have a cumulative effect on fetal brain development,” Dr. Limperopoulos said. “We must study and consider how to conceptualize the impact of socioenvironmental disadvantage in communities to better care for children and work to improve outcomes.”
Read the full study – “Greater Neighborhood Disadvantage Is Associated with Alterations in Fetal Functional Brain Network Structure” – in The Journal of Pediatrics.