Gut microbiome may impact susceptibility to konzo

From left to right: Dr. Matthew Bramble, Vincent Kambale, and Neerja Vashist. Here, the team is processing samples in the field collected from the study cohort prior to storage in liquid nitrogen. Bramble et al. Nature Communications (2021).
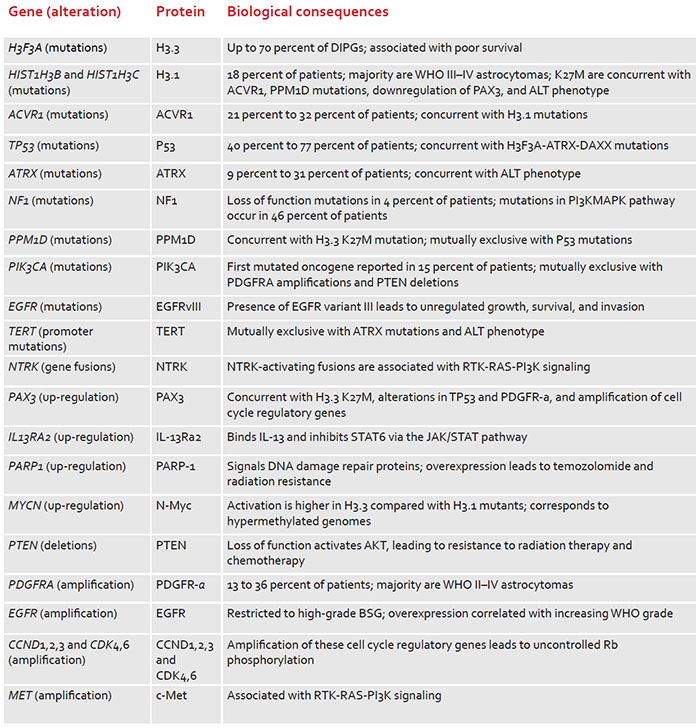
Differences between gut flora and genes from konzo-prone regions of the Democratic Republic of Congo (DRC) may affect the release of cyanide after poorly processed cassava is consumed, according to a study with 180 children. Cassava is a food security crop for over half a billion people in the developing world. Children living in high-risk konzo areas have high glucosidase (linamarase) microbes and low rhodanese microbes in their gut, which could mean more susceptibility and less protection against the disease, suggest Children’s National Hospital researchers who led the study published in Nature Communications.
Konzo is a severe, irreversible neurologic disease that results in paralysis. It occurs after consuming poorly processed cassava — a manioc root and essential crop for DRC and other low-income nations. Poorly processed cassava contains linamarin, a cyanogenic compound. While enzymes with glucosidase activity convert starch to simple sugars, they also break down linamarin, which then releases cyanide into the body.
Neerja Vashist is learning how to make fufu. Fufu is a traditional food made from cassava flour, and the cassava flour used in the making of the fufu here has gone through the wetting method to further remove toxins from the cassava flour prior to consumption. Bramble et al. Nature Communications (2021).
“Knowing who is more at risk could result in targeted interventions to process cassava better or try to diversify the diet,” said Eric Vilain, M.D., Ph.D., director of the Center for Genetic Medicine Research at Children’s National. “An alternative intervention is to modify the microbiome to increase the level of protection. This is, however, a difficult task which may have unintended consequences and other side effects.”
The exact biological mechanisms underlying konzo disease susceptibility and severity remained poorly understood until now. This is the first study to shed light on the gut microbiome of populations that rely on toxic cassava as their primary food source.
“While the gut microbiome is not the sole cause of disease given that environment and malnourishment play a role, it is a required modulator,” said Matthew S. Bramble, Ph.D., staff scientist at Children’s National. “Simply stated, without gut microbes, linamarin and other cyanogenic glucosides would pose little to no risk to humans.”
To understand the influence of a detrimental subsistence on the gut flora and its relationship to this debilitating multifactorial neurological disease, the researchers compared the gut microbiome profiles in 180 children from the DRC using shotgun metagenomic sequencing. This approach evaluates bacterial diversity and detects the abundance of microbes and microbial genes in various environments.
The samples were collected in Kinshasa, an urban area with diversified diet and without konzo; Masi-Manimba, a rural area with predominant cassava diet and low prevalence of konzo; and Kahemba, a region with predominant cassava diet and high prevalence of konzo.

From left to right: Dr. Nicole Mashukano and Dr. Matthew Bramble. Dr. Mashukano leads the efforts in Kahemba to teach the wetting method to individuals in different health zones. The wetting method is used as an additional step to further detoxify toxins from cassava flour prior to consumption. Here, Dr. Mashukano and Dr. Bramble are spreading out the wet mixture of cassava flour and water into a thin layer on a tarp for drying in the sun, which allows cyanogen breakdown and release in the form of hydrogen cyanide gas. Bramble et al. Nature Communications (2021).
“This study overcame many challenges of doing research in low-resource settings,” said Desire Tshala-Katumbay, M.D., M.P.H., Ph.D., FANA, co-senior author and expert scientist at Institut National de Recherche Biomédicale in Kinshasa, DRC, and professor of neurology at Oregon Health & Science University. “It will open novel avenues to prevent konzo, a devastating disease for many children in Sub-Saharan Africa.”
For next steps, the researchers will study sibling pairs from konzo-prone regions of Kahemba where only one sibling is affected with the disease.
“Studying siblings will help us control for factors that cannot be controlled otherwise, such as the cassava preparation in the household,” said Neerja Vashist, Ph.D. candidate and research trainee at Children’s National. “In this work, each sample had approximately 5 million DNA reads each, so for our follow-up, we plan to increase that to greater than 40 million reads per sample and the overall study cohort size. This study design will allow us to confirm that the trends we observed hold on a larger scale, while enhancing our ability to comprehensively characterize the gut microbiome.”