FDA approves muscular dystrophy drug built on Children’s National research
Duchenne muscular dystrophy (DMD) is the most common hereditary neuromuscular disease.
Boys with Duchenne muscular dystrophy (DMD) have a clinically proven, new treatment option with the Food and Drug Administration’s approval of vamorolone, a steroidal-type, anti-inflammatory drug developed based on research performed at Children’s National Hospital.
Created by ReveraGen BioPharma Inc., vamorolone has a molecular structure similar to traditional corticosteroids, which are currently used to treat DMD. Yet its structure was found to be chemically different enough to reduce unwanted side effects, including brittle bones and reduced stature. Nearly two decades ago, ReveraGen leaders – President and CEO Eric Hoffman, Ph.D., and Vice President for Research Kanneboyina Nagaraju, D.V.M., Ph.D. – launched research efforts into the drug when they led the Center for Genetic Medicine Research at Children’s National. They worked with then-Chief Academic Officer Mark Batshaw, M.D., on the new clinical option.
“Throughout my career, I have treated children with DMD, and I have seen over time how their shorter heights and brittle bones impact them physically and emotionally – in terms of their self-esteem and ability to participate in activities,” Dr. Batshaw said. “This drug should help these boys function more effectively and prevent certain long-term complications.”
The patient benefit
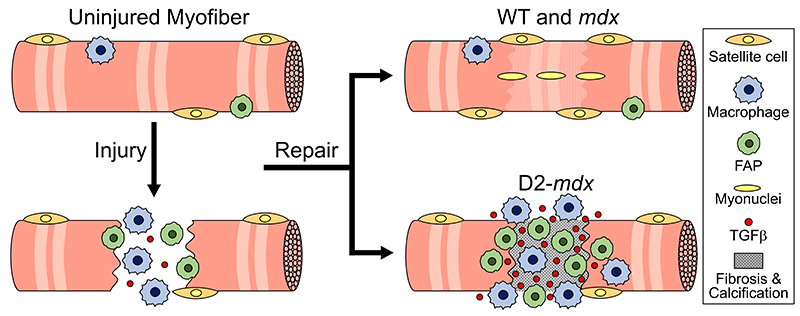
Muscular dystrophy includes a group of degenerative genetically inherited neuro-muscular diseases that strike only boys. DMD is the most common, severe and life-threatening form of muscular dystrophy. ReveraGen studied vamorolone for patients ages two years and up in the hopes of providing a new, FDA-approved treatment option for these children. In clinical trials, daily treatment with vamorolone improved muscle strength and stature with results comparable to prednisolone, but without some of the most impactful side effects of steroids, particularly the stunted growth and weakened bones.
Children’s National Hospital leads the way
Kolaleh Eskandanian, Ph.D., M.B.A., P.M.P., vice president and chief innovation officer for Children’s National, said Drs. Hoffman and Nagaraju’s work on the drug paved the way for entrepreneurship at the hospital, as they were the first faculty members to launch a spin-off company. Since then, more than 130 faculty members have been named as inventors on 132 patents. Children’s National is now home to Innovation Ventures, the hospital’s intellectual property development and commercialization arm, which provides guidance and resources to academic entrepreneurs who introduce a concept for pediatric medical products.
“We cannot wait to see the tremendous effort behind vamorolone in the hands of patients and clinicians treating Duchenne muscular dystrophy,” Eskandanian said. “Today’s FDA approval for ReveraGen shows the importance of supporting clinicians and researchers who are developing solutions to advance healthcare for children.”
Why we’re excited
Hoffman said the drug has been through a series of clinical trials showing advantages over the current treatment options. In 2024, Catalyst Pharma will market vamorolone under the trade name Agamree in the United States.
“Vamorolone was developed using a different business model and drug development approach, including partnerships with the National Institutes of Health, Department of Defense, the European Commission and more than a dozen international nonprofit foundations,” Dr. Hoffman said. “The collaborative, community-engaged approach—including 32 academic clinical sites in 11 countries — and the participation of hundreds of DMD families led to this approval today.”