Mitochondria key for repairing cell damage in DMD
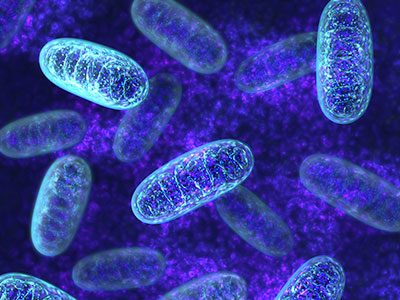 A research team led by Jyoti K. Jaiswal, M.S.C., Ph.D., found that dysfunctional mitochondria prevent repair of muscle cells in Duchenne muscular dystrophy. What’s knownDuchenne muscular dystrophy (DMD), one of the most severe forms of muscular dystrophy, is caused by a defect in the dystrophin gene. The protein that this gene encodes is responsible for anchoring muscle cells’ inner frameworks, or cytoskeletons, to proteins and other molecules outside these cells, the extracellular matrix. Without functional dystrophin protein, the cell membranes of muscle cells become damaged, and the cells eventually die. This cell death leads to the progressive muscle loss that characterizes this disease. Why these cells are unable to repair this progressive damage has been unknown. What’s newA research team led by Jyoti K. Jaiswal, M.S.C., Ph.D., a principal investigator in the Center for Genetic Medicine Research at Children’s National Health System, investigated this question in two experimental models of DMD that carry different mutations of the dystrophin gene. The researchers monitored the effects of the lack of functional dystrophin protein in these preclinical models on the level and function of muscle cell. They found that mitochondria – organelles that act as powerhouses to supply the chemical energy to drive cellular activities – are among the first to be affected. They found that the decline in mitochondrial level and activity over time in these experimental models preceded the onset of symptoms. The research team also looked at the ability of the experimental models’ muscle cells to repair damage. As the muscle cell mitochondria lost function, the cells’ ability to repair damage also declined. Efforts to increase mitochondrial activity after these organelles became dysfunctional did not improve muscle repair. This suggests that poor muscle repair may not be caused by a deficit in energy production by mitochondria. Questions for future researchQ: Does similar mitochondrial dysfunction occur in human patients with DMD? Source: “Mitochondria mediate cell membrane repair and contribute to Duchenne muscular dystrophy.” Vila, M.C., S. Rayavarapu, M.W. Hogarth, J.H. Van der Meulen, A. Horn, A. Defour, S. Takeda, K.J. Brown, Y. Hathout, K. Nagaraju and J.K. Jaiswal. Published by Cell Death and Differentiation February 2017. |