Advancing clinical deployment of artificial intelligence in pediatric radiology
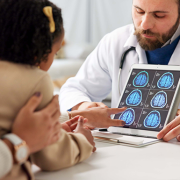
Researchers at Children’s National Hospital are helping advance pediatric AI with a first-of-its-kind open-access brain tumor imaging dataset designed to improve radiology and precision medicine for children.
Artificial intelligence (AI) is moving quickly in medicine, and radiology is one of the easiest places to see that progress. But in pediatrics, the path forward is more complicated. Children are not just smaller versions of adult patients. They have different diseases, different biology and different care processes, which means AI has to be built with that reality in mind from the beginning.
That challenge is clear in a new effort focused on pediatric brain tumors. While rare, these tumors remain the most common solid tumors in children and the leading cause of cancer-related mortality in the pediatric population. Compared to adult brain tumors, they behave differently across biology, anatomy and clinical care, which makes diagnosis, monitoring and treatment response especially complex. AI has the potential to improve all of this. But progress has been slow, largely because the field has been working without the kind of large, standardized and accessible datasets that make AI actually work at scale.
Dive deeper
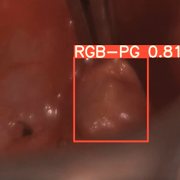
A new international effort, co-led by Marius George Linguraru, DPhil, MA, MSc, of Children’s National Hospital and Anahita Fathi Kazerooni, PhD of Children’s Hospital of Philadelphia, is helping change that. The Brain Tumor Segmentation in Pediatrics dataset, known as BraTS-PEDs, is the first large-scale, open-access benchmark dataset designed specifically for pediatric brain tumor segmentation and analysis. It brings together MRI data from multiple imaging sequences from 457 pediatric patients with high-grade gliomas, collected across several institutions and international research consortia.
Each case includes a full set of clinically relevant imaging sequences, including pre- and postcontrast T1-weighted, T2-weighted and T2-FLAIR MRI. Experts labeled different parts of each tumor using established pediatric neuro-oncology guidelines. They combined automated tools designed for pediatric cases with careful review and refinement by neuroradiologists. The result is a dataset that is not only large but consistent in a way the field has not had before.
Just as important, the dataset is structured to support real-world use. It is divided into training, validation and hidden testing subsets, allowing researchers to benchmark models in a reproducible way and evaluate how well those models generalize across institutions.
Why this matters
This kind of resource addresses one of the biggest barriers in pediatric AI. Without enough high-quality, standardized data, even the most promising algorithms struggle to translate into clinical use.
BraTS-PEDs changes that by giving researchers a shared foundation. It allows for consistent method comparison, supports model development across institutions and opens the door for integrating imaging with molecular and clinical data, which is where things start to move toward precision medicine in a meaningful way.
For pediatric radiology, that shift is critical. AI is not just about improving image analysis. It is about building tools that can help clinicians better understand disease, track how it evolves and make more informed decisions over time. Dr. Linguraru’s team has created public tools to provide open access to the volumetric analysis of pediatric brain tumors to doctors and researchers everywhere.
What comes next
At Children’s National, this work fits into a broader effort to rethink how AI is developed and deployed in pediatric care. Through its Division of AI Research, the focus is on creating a clinically grounded ecosystem where data, technology and clinical expertise are tightly connected. The goal is to move beyond isolated projects and build infrastructure that allows ideas to move from concept to application in a way that is scalable and sustainable. As Dr. Linguraru notes, “AI tools can play a key role in radiology, but radiologists must be able to trust in the systems’ design and receive adequate training.”
Radiology is already leading the way when it comes to AI in medicine, but pediatrics is still catching up in terms of data and infrastructure. Efforts like BraTS-PEDs are a step toward closing that gap. By creating shared, standardized resources, they make it possible to build and evaluate models in a way that is consistent, collaborative and clinically relevant. For children, that matters. Getting AI right in pediatrics is not just about innovation. It is about making sure the tools being developed actually reflect the patients they are meant to serve.
This research, Brain Tumor Segmentation in Pediatrics (BraTS-PEDs): A Multi-Institutional Benchmark Dataset for Pediatric Neuro-Oncology, was published in Radiology: Artificial Intelligence. Authors from Children’s National Hospital include Marius George Linguraru, DPhil, MA, MSc; Zhifan Jiang, PhD; Xinyang Liu, PhD; Brian Rood, MD; and Roger J. Packer, MD; in collaboration with colleagues from multiple institutions and international consortia working to advance artificial intelligence in pediatric neuro-oncology.