Adapted from Levitt MA. New and exciting advances in pediatric colorectal and pelvic reconstructive surgery – 2021 update. Semin Pediatr Surg. 2020 Dec;29(6):150992.
Adapted from Levitt MA. New and exciting advances in pediatric colorectal and pelvic reconstructive surgery – 2021 update. Semin Pediatr Surg. 2020 Dec;29(6):150992.
As demonstrated in the popular children’s book by Taro Gomi, Everyone Poops, the physiology of stooling is a focus of early childhood development and a subject of concern for both parents and children. References to pediatric colorectal problems go back many thousands of years. In fact, the Babylonian Talmud, written in the year 200 CE, recommends that “an infant whose anus is not visible should be rubbed with oil and stood in the sun… and where it shows transparent the area should be torn crosswise with a barley grain.” Surgical techniques to manage such patients have certainly evolved since that time, but the basic principles of care remain the same.
How we got here
The modern story of the care of patients with anorectal malformations (ARMs) began in the 1940s in Melbourne, Australia, when Henry Douglas Stephens worked to define the anatomy of children with ARMs by analyzing the anatomy of twelve deceased patients with these conditions. He continued to focus on this specialty for the rest of his career and published two books on the topic in 1963 and 1971. Prior to his groundbreaking work, the anatomy of such patients was only a concept that existed in surgeons’ minds – without anatomic precision – since no one had actually seen the anatomy. These concepts were depicted in the bible of pediatric surgery in North America, the Gross textbook, which in retrospect was both oversimplified and inaccurate.
During his autopsy dissections, Stephens came to the key anatomic conclusion that the puborectalis muscle (the sphincters) lay behind the urethra. He devised an operation based on this concept: first, the urethra was identified, then a space between it and the puborectalis was dissected, and then the rectosigmoid was pulled through that space. A small incision in the perineum for the pulled-through bowel was made, within which the new anus was created. The perineal dissection was a blind maneuver. During the same time period, William Kiesewetter in Pittsburgh proposed his version of the sacral abdominoperineal pull-through using similar anatomic principles.
Justin Kelly was one of Stephens’ trainees in Australia who learned how to do this operation. At Boston Children’s Hospital in the late 1960’s, he taught what he had learned from Stephens to the surgeons there, including another trainee, Alberto Pena. Pena and his fellow surgical residents benefitted from exposure to Kelly as well as master surgeon Hardy Hendren, who operated on patients across town at Massachusetts General Hospital. Hendren, the pioneer in the care of children with cloacal anomalies, passed away this year at the age of 96.
Pena completed his training in Boston and went to Mexico City in 1972 at the age of 34 to become the head of surgery at the National Pediatric Institute. He tells the story that when he asked his new pediatric surgery faculty to choose an area of specialization, no one chose colorectal, so he decided to take on that group of patients and thus embarked on his revolutionary colorectal career. Pena at first applied the technique he had learned from Kelly to repair anorectal malformations, but he became increasingly frustrated by the procedure. He felt that the maneuvers offered very poor exposure to the anatomy, and over time his incision grew longer and longer. In 1980, Pena’s collaboration with Pieter de Vries – who had come to Mexico City to work on these cases with Pena – culminated in the first posterior sagittal anorectoplasty [Figure 1]. That same year, Pena presented his findings at a meeting of the Pacific Association of Pediatric Surgeons.
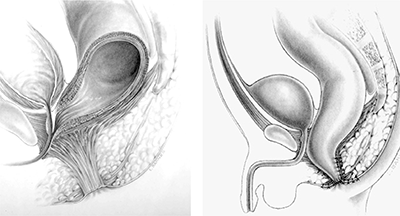
Figure 1: Original diagrams of the posterior sagittal approach to repair imperforate anus
This posterior sagittal approach opened a beneficial Pandora’s Box in surgery. It allowed for a true understanding of the pelvic anatomy and led to the care of many conditions that were previously – to use Pena’s words – “too difficult to reach from above (via laparotomy) and too difficult to reach from below (perineally).” This new approach influenced the repair of cloacal malformations, urogenital sinus, pelvic tumors, urethral problems, reoperations for imperforate anus and Hirschsprung disease (HD), a transpubic approach for complex genitourinary problems, and a comprehensive strategy for the management of cloacal exstrophy. In addition to his surgical contributions, Pena also conceived of the intervention that has arguably improved patients’ quality of life the most: a focused approach to the bowel management of fecal incontinence. Thanks to such bowel management programs, now available at many centers across the world, thousands of children are no longer wearing diapers and have said goodbye to their stomas. The impact of bowel management is perhaps comparable to that of intermittent catheterization for patients with urinary incontinence.
My personal journey in this field began in 1992, when I was an eager medical student and signed up for an elective in pediatric surgery with Alberto Pena. This experience changed my career trajectory in a very dramatic and positive way. Medicine was becoming increasingly complex, and fields such as neonatal care, transplantation, and cardiology were benefitting from a collaborative approach. It became clear that the colorectal field needed the same approach. As a comparative example, consider the project of building a bridge. How does such a project start? The cement layers do not show up one day and lay cement prior to the steel team deciding where to place the beams. The project must begin with all parties meeting together to develop a comprehensive plan. Amazingly, however, that type of collaborative planning does not often happen in the care of medically complex patients. It most assuredly needs to.
The value of multi-disciplinary care
Medical complexity requires integrated and collaborative care because all the anatomic structures that need to be managed are located right next to each other and because each has a unique and complex physiology that can affect the other systems. To achieve success, patients with anorectal malformations, Hirschsprung disease, fecal incontinence (related to a variety of conditions), and colonic motility disorders require care from a variety of specialists throughout their lives. These include providers in the fields of colorectal surgery, urology, gynecology, gastroenterology, motility, orthopedics, neurosurgery, anesthesia, pathology, radiology, psychology, social work, nutrition, and many others. Perhaps most important to the achievement of a good functional result, however, is a patient’s connection to superb nursing care. A complex colorectal operation takes about four hours to perform, but to get a good result, it takes an additional 96 hours of work, the vast majority of which involves nursing care. The value of good nursing partners to ensure successful surgery cannot be overemphasized. They have unique skills in identifying and solving problems, a willingness to get down in the weeds, and are always striving to fill the gaps in care.
What parents want and need
Having met many parents with newborns diagnosed with colorectal problems, I have made several observations. First, it seems that no parent has ever imagined that their child could have a problem with stooling – this is a physiologic ability that is taken for granted. When they are told about the problem with their baby, they are uniformly shocked that something like this could happen. Second, when discussing that their child will need surgery to correct their colorectal anatomy, parents don’t focus on the surgical technique and elegance of the reconstruction, as surgeons tend to do. Instead, parents dwell on whether the surgery will create a working reconstructed anatomy that will allow their child to stool without difficulty or embarrassing accidents. As surgeons we need to remember this. We always need to understand what it is that the family and patient wishes us to deliver, and we need to strive to achieve those goals. As proud of our surgical skills as we are, it is the functional outcome that matters most.
Where we are in 2022
In 2022, the advances in the field of pediatric colorectal and pelvic reconstruction are significant. They include new techniques and ideas that over time have made a dramatic and positive impact on the care and quality of life of children who suffer from colorectal problems. Here are a few such advances:
- Prenatal diagnosis of anorectal and cloacal malformations has been progressively improving. Perinatologists have learned to look for specific findings, such as a pelvic mass in a female with a single kidney, and consider that it could be a cloaca. Assessment of perineal anatomy, pubic bone integrity, sacral development, abnormalities of the radius bone, as well as cardiac, spinal, and renal anomalies may lead the clinician to consider that a fetus may have an anorectal malformation.
- Management of the newborn, particularly in the fields of newborn radiology and neonatal care, has dramatically improved as neonatal techniques have advanced. Specific to the colorectal patient have been advances in radiology such as assessments of hydronephrosis, 3D reconstruction of cloacal anomalies, and ultrasound-guided distal colostography. Further advances include improved techniques in the management of hydrocolpos and stoma care, to name a few.
- The treatment of associated urologic anomalies has diminished chronic renal disease, and proactive bladder management is reducing the need for bladder augmentations and renal transplantation.
- Understanding the gynecologic collaboration has helped clinicians define the Mullerian anatomy and better plan for menstruation and future obstetric potential.
- Prediction of continence, even in the newborn period, requires an understanding of the associated problems with the sacrum and spine. This knowledge has allowed clinicians to have more robust conversations with families about their child’s future.
- The decision of whether to do a newborn repair versus a colostomy must be guided by the surgeon’s experience and the clinical circumstances in which they find themselves.
- The defining of anatomy allows patients to be compared across medical centers, and for treatment options and outcomes to be uniformly analyzed. Keeping track of one’s outcomes and always striving to improve should be basic tenets of surgical practice.
- Recognizing the value of laparoscopy and knowing for which cases this approach should be applied. Morbidities associated with a laparoscopic approach for a rectum in an ARM patient well below the peritoneal reflection have been noted. In HD cases, laparoscopy can limit the stretching of the sphincters which occurs during the transanal rectal dissection.
- Development of a treatment algorithm for the management of cloacal malformations which considers the importance of their common channel and urethral lengths.
- Recognizing key complications after ARM and Hirschsprung surgery, knowing when and how to do a reoperation, determining the outcomes of such reoperations, and ultimately figuring out how to avoid complications altogether.
- Understanding the causes of fecal incontinence, the amount of incontinence that can be anticipated, and the surgical contributors to achieving continence.
- Development of bowel management programs in multiple centers and committing to following these patients in the long term.
- Learning the pathophysiology of motility disorders and developing treatment protocols, as a result of the vital collaboration between surgery and GI/motility. Medical treatments with laxatives, rectal enemas, botox injection of the anal canal, and surgical adjuncts such as antegrade colonic flush options and sometimes colon resections are key aspects of the treatment armamentarium.
- Developing a collaboration between colorectal surgery and urology allows the clinical team to know when the colon can be used for a bladder augment (which not only offers an augment option but also can improve antegrade flushes of the colon) or if the appendix can be shared between Malone and Mitrofanoff. The collaboration with urology to plan the management of both urinary and fecal continence during the same operation is a very valuable trend. This proactive planning has improved the lives of many patients and has reduced the numbers of operations they need as well as their hospital stays.
- Sacral Nerve Stimulation (SNS) has shown promise in the management of urinary incontinence and seems to have a role in improving fecal continence and promoting motility, as an adjunct to treatments for constipation.
- Development of dedicated colorectal centers that are integrated and collaborative allows a team to tabulate their results and strive for better outcomes. The value of a collaborative model for the care of such patients cannot be overstated, not only for patient convenience, but also for creating an integrated plan for their care. These exist now in many parts of the country and care is available in nearly every region, reducing a family’s need to travel great distances away from their home to access care.
- Establishing a transition to adult programs, as with congenital heart disease and cystic fibrosis. Colorectal surgeons are obligated to develop a transition plan for their patients as they enter adulthood.
- Using Basic Science to advance the field, including tissue engineering and genetics, will be revolutionary. We should be able to imagine the day when cloacal reconstruction could be based on a previously tissue-engineered segment of vagina, produced by the patient’s own stem cells. In addition, the genetics of anorectal malformations as well as Hirschsprung disease are being vigorously pursued which will impact parental counseling and potential therapies.
- Real time data used to follow outcomes is needed to keep track of complications as well. That information can be used to adjust protocols which will improve results.
- Development of international consortiums will help patients in a way that is not achievable by a single institution. Consortiums allow ideas to be spread rapidly which will dramatically affect how many patients can be helped and how quickly. The Pediatric Colorectal and Pelvic Learning Consortium, pcplc.org, is well on its way to achieving these goals.
- Bringing complex care to all corners of the world because there is a great deficiency in advanced colorectal care in the developing world. The care of colorectal patients in a resource-limited setting has unique challenges, but creative solutions by innovative surgeons in those areas have a led to dramatic improvements in care.
- Parent/patient organizations provide education, advocacy, and support for families at all stages of their child’s care. With internet access readily available, colorectal patients and their families can now access the welcoming environment of these organizations, and no longer feel as lost and alone as in previous years.
Why it matters
Given all these wonderful advances, we must continue to reaffirm the key principles stated by Sir Dennis Browne that “the aim of pediatric surgery is to set a standard, not to seek a monopoly.” With an integrated approach to the care of this complex group of patients, great things can be achieved. I am hopeful that the caregivers and parent/patient group organizations who commit to the care of children with colorectal problems and understand the daily struggle of improving a patient’s quality of life will learn the skills and tricks necessary to achieve good results. If they do, they will help many children.
Finally, I will share a humorous piece written by my daughter, Jess Levitt, regarding the value of bringing order to chaos. Its message is particularly relevant to the care of children with colorectal problems in 2022, as we build on our efforts to improve, streamline, and transform the formerly chaotic process through collaboration and education. As we endeavor to advance this field, we need to remember what Dr. Pena often said: “It is not the unanswered questions, but rather the unquestioned answers that one must pursue.”
“A” must come before “B,” which must come before “C,” everybody knows that. But what if the Millercamps of this world did not have to sit next to the Millerchips when it comes to seating arrangements? Can Pat Zawatsky be called before Jack Aaronson when the teacher is taking attendance? Do those 26 letters that make up all the dialogue, signs, thoughts, books, and titles in the English-speaking department of the world need their specific spots in line? Everyone can sing the well-known jingle from A to Z, but not many people can tell you why the alphabet is the way it is.
For almost as long as humans have had the English language, they have had the alphabet. The good ol’ ABCs. However, the alphabet represents the human need for order and stability. I believe that the same thinking that went into the construct of time and even government went into the alphabet. Justifiably, lack of order leads to chaos. Knife-throwing, gun-shooting chaos, in the case of lack of governmental order. Listen to me when I tell you that there is absolutely no reason that the alphabet is arranged the way that it is. Moreover, the alphabet is simply a product of human nature and how it leads people to establish order for things that do not require it.
Now I know this sounds crazy, but bear with me. Only if you really peel away the layers of the alphabet will you find the true weight it carries. People organized the letters of our speech into a specific order simply because there wasn’t already one. Questioning this order will enlighten you on the true meaning of it. Really dig deep into the meaning behind the social construct that is the alphabet. Short and sweet as it may be, the order of the ABCs is much less than meets the eye. There is no reason that “J” should fall before “K”! Understand this. Very important as order is, it is only a result of human nature.
What’s next? X-rays become independent of Xylophones in children’s books of ABCs?
You know what the best part is? Zero chance you even noticed that each sentence in this essay is in alphabetical order.
– Jess Levitt
 “Within the field of pediatric colorectal and pelvic reconstruction, the most complex anatomic problem a pediatric surgeon can face is that of a cloacal malformation,” writes Marc A. Levitt, M.D., chief of Colorectal and Pelvic Reconstruction at Children’s National Hospital. A new, first-of-its kind textbook, Cloacal Malformations: Case Studies, seeks to shed light on the multi-disciplinary care required to care for people with this rare condition — when the urinary, gynecologic and colorectal system all exit the body via a common channel which requires reconstruction. The book will be available for pre-order on April 9, 2024.
“Within the field of pediatric colorectal and pelvic reconstruction, the most complex anatomic problem a pediatric surgeon can face is that of a cloacal malformation,” writes Marc A. Levitt, M.D., chief of Colorectal and Pelvic Reconstruction at Children’s National Hospital. A new, first-of-its kind textbook, Cloacal Malformations: Case Studies, seeks to shed light on the multi-disciplinary care required to care for people with this rare condition — when the urinary, gynecologic and colorectal system all exit the body via a common channel which requires reconstruction. The book will be available for pre-order on April 9, 2024.






 Advanced MRI visualization techniques to follow blood flow in the hearts of cardiac patients. Gene therapy for pediatric patients with Duchenne muscular dystrophy. 3D-printed casts for treating clubfoot. These were among the most popular articles we published on Innovation District in 2023. Read on for our full list.
Advanced MRI visualization techniques to follow blood flow in the hearts of cardiac patients. Gene therapy for pediatric patients with Duchenne muscular dystrophy. 3D-printed casts for treating clubfoot. These were among the most popular articles we published on Innovation District in 2023. Read on for our full list.

 Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.


 A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.










 Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its
Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its 
 Adapted from
Adapted from 



 Pediatric colorectal specialists are in short supply, and this is particularly true in many areas of the developing world. When
Pediatric colorectal specialists are in short supply, and this is particularly true in many areas of the developing world. When