Artificial – and accelerated – intelligence: endless applications to expand health equity
In the complex world of pediatric diseases, researchers need access to data to develop clinical trials and the participation of vulnerable patients to develop new devices and therapies. Both are in short supply, given that most children are born healthy, and most severe pediatric diseases are rare.
That creates a dilemma: how do researchers build a foundation to advance new treatments? Enter artificial intelligence (AI).
“AI is the equalizer: accelerated intelligence for sick kids. No other advance on the horizon holds more promise for improving equity and access to pediatric healthcare when diseases are rare and resources are limited,” says Marius George Linguraru, D.Phil., M.A., M.Sc., the Connor Family Professor in Research and Innovation and principal investigator in the Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI). “AI will shrink the distance between patient and provider, allowing our physicians and scientists to provide targeted healthcare for children more efficiently. The possibilities are endless.”
Why we’re excited
By pioneering AI innovation programs at Children’s National Hospital, Dr. Linguraru and the AI experts he leads are ensuring patients and families benefit from a coming wave of technological advances. The team is teaching AI to interpret complex data that could otherwise overwhelm clinicians. Their work will create systems to identify at-risk patients, forecast disease and treatment patterns, and support complex clinical decisions to optimize patient care and hospital resources. Already, the AI team at SZI has developed data-driven tools touching nearly every corner of the hospital:
- AI for rheumatic heart disease (RHD): In partnership with Children’s National cardiology leaders, including Craig Sable, M.D., the Uganda Heart Institute and Cincinnati Children’s Hospital, the AI team has developed an algorithm that can use low-cost, portable ultrasound imaging to detect RHD in children and young adults, a disease that takes nearly 400,000 lives annually in limited-resource countries. Early testing shows the AI platform has the same accuracy as a cardiologist in detecting RHD, paving the way for earlier treatment with life-saving antibiotics. This year, Children’s National physicians will be in Uganda, screening 200,000 children with local cardiology experts and AI technology.
- Newborn screening for genetic conditions with mGene: Working with Rare Disease Institute clinicians and Chief of Genetics and Metabolism Debra Regier, M.D., the AI team has built technology to detect rare genetic disorders, using an algorithm and a smartphone camera to identify subtle changes in facial features. Tested on patients from over 30 countries and published in The Lancet Digital Health, the application helps screen children for advanced care when a geneticist may not be within reach. With funding from the National Institutes of Health, Children’s National and its research partners are piloting a newborn screening program in the Democratic Republic of the Congo.
- Pediatric brain tumors: To improve and personalize the treatment decisions for children with brain tumors, Dr. Linguraru’s team is working with Brain Tumor Institute Director Roger Packer, M.D., the Gilbert Family Distinguished Professor of Neurofibromatosis, on algorithms that can characterize and measure brain tumors with unprecedented precision. The team recently won the International Pediatric Brain Tumor Segmentation Challenge, distinguishing the Children’s National algorithm as among the best in the world.
- Ultra-low field magnetic resonance imaging (MRI): With a grant from the Bill & Melinda Gates Foundation, the AI team is working alongside Children’s Hospital Los Angeles, King’s College London and the UNITY Consortium to expand global brain imaging capacity. The consortium is helping clinicians in limited-resource countries improve the treatment of neonatal neurological conditions, using AI to boost the quality of ultra-low field MRI and expand access to this portable and more affordable imaging option.
- Federated learning: Children’s National has collaborated with NVIDIA and other industry leaders to accelerate AI advances through federated learning. Under this approach, institutions share AI models rather than data, allowing them to collaborate without exposing patient information or being constrained by essential data-sharing restrictions. The SZI team was the only pediatric partner invited to join the largest federated learning project of its kind, studying the lungs of COVID-19 patients. Details were published in Nature Medicine.
Children’s National leads the way
Looking ahead, the Children’s National AI team is pursuing a wide range of advances in clinical care. To support patients treated at multiple clinics, they are developing systems to harmonize images from different scanners and protocols, such as MRI machines made by different manufacturers. Similar work is underway to analyze pathology samples from different institutions consistently.
Automation is also making care more efficient. For example, using data from 1 million chest X-rays, the team is collaborating with NVIDIA to develop a conversational digital assistant that will allow physicians to think through 14 possible diagnoses.
Dr. Linguraru says he and his colleagues are galvanized by the jarring statistic that one in three children with a rare disease dies before age 5. While well-implemented AI initiatives can change outcomes, he says the work must be done thoughtfully.
“In the future, patients will be evaluated by human clinicians and machines with extraordinary powers to diagnose illness and determine treatments,” Dr. Linguraru said. “Our team at Children’s National is leading conversations about the future of pediatric healthcare with a focus on safety, resource allocation and basic equity.”






 Advanced MRI visualization techniques to follow blood flow in the hearts of cardiac patients. Gene therapy for pediatric patients with Duchenne muscular dystrophy. 3D-printed casts for treating clubfoot. These were among the most popular articles we published on Innovation District in 2023. Read on for our full list.
Advanced MRI visualization techniques to follow blood flow in the hearts of cardiac patients. Gene therapy for pediatric patients with Duchenne muscular dystrophy. 3D-printed casts for treating clubfoot. These were among the most popular articles we published on Innovation District in 2023. Read on for our full list.





 Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked #5 in the nation on the U.S. News & World Report 2023-24 Best Children’s Hospitals annual rankings. This marks the seventh straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.










 A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
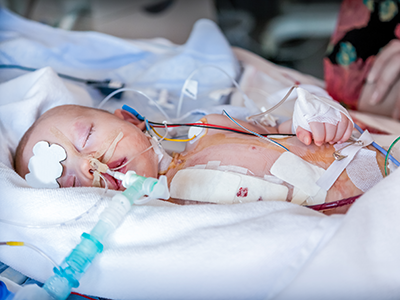

 New research found that mepolizumab, a monoclonal antibody, decreased asthma flare-ups by 27% in Black and Hispanic children and adolescents who have a severe form of asthma, are prone to asthma attacks, and live in low-income urban neighborhoods.
New research found that mepolizumab, a monoclonal antibody, decreased asthma flare-ups by 27% in Black and Hispanic children and adolescents who have a severe form of asthma, are prone to asthma attacks, and live in low-income urban neighborhoods.


 Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its
Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its 