Community-wide program to support teen parents serves as a model for engagement
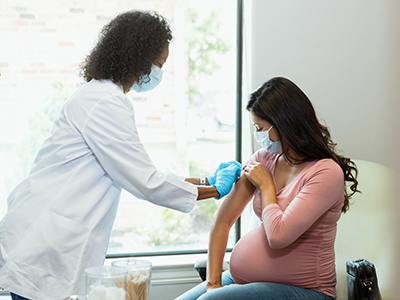
More than 500 adolescent mothers, caregivers and community members benefitted from a coordinated “collective impact” model to provide support aimed at addressing the litany of strains faced by teen parents, according to a case study published Wednesday in the journal Pediatrics.
Known as the District of Columbia Network for Parenting and Expectant Teens (DC NEXT), the model used well-tested pillars of community organization to provide services and care that bolstered the well-being of pregnant and expectant teens in the city. The case study found that the 3-year-old program, which included teen advisors to help guide the mission, could serve as a model in other areas with high rates of teen parents.
“There’s no system of care for teen parents in Washington, D.C., or really anywhere in our country,” said Yael Smiley, M.D., Children’s National Hospital pediatrician and the study’s lead author. “Our coalition came together to connect the resources and the people who care about young parents and their families to improve their health, their outcomes, their well-being, and set them up for success. We fuse representatives from healthcare, education and housing policy — people who care about creating the very best outcomes possible.”
The patient benefit
More than 140,000 teens became parents in 2021 in the United States. In Washington, D.C., more than 300 babies are born to teens each year, and the rate of births to young teens ages 15 to 17 is double the national average. These young parents often face barriers to childcare and education and poor birth outcomes, often stemming from a lack of access to prenatal care.
To address these obstacles, the network assembled a city-wide network in 2020 to follow the five pillars of an evidence-based “collective impact” model:
- Create a common agenda
- Design shared measurement systems
- Host mutually reinforcing activities
- Foster continuous communication
- Rely on trusted backbone support
What’s unique
DC NEXT was housed at the District of Columbia Primary Care Association (DCPCA), which received the 3-year, $4.5 million federal grant and provided the leadership. At Children’s National, Dr. Smiley led a team that partnered with DCPCA and Howard University to direct a network of community clinics, nonprofits and other organizations whose mission was to support adolescent parents. Hundreds of client-facing staff members were trained to provide trauma-informed, human-centered care. DC NEXT also engaged directly with teen mothers through well-being surveys that led to improved access to essential programs, including resources for housing and food security. Over 550 young parents and caregivers have been impacted by the program.
The leadership quickly realized that the teen parents needed a voice and created a “context team” of paid teen advisors who provided insights into their unique experiences and needs as parents. They help set the agenda, choose program offerings and tailor communications to their peers.
“If the network can continue to grow and support young parents to achieve their health and well-being goals, the impact will be felt across generations,” said Dr. Smiley.










 A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.






 Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its
Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its