The International Pediatric Transplant Association (IPTA) is hosting their 11th Congress meeting March 26-29, 2022, and many Children’s National Hospital providers will be presenting throughout the conference. We hope you will join us!
The International Pediatric Transplant Association (IPTA) is hosting their 11th Congress meeting March 26-29, 2022, and many Children’s National Hospital providers will be presenting throughout the conference. We hope you will join us!
Diversity, inequity and inclusivity in the practice of pediatric transplantation in the U.S.
Presenter: Marva Moxey-Mims, M.D., division chief, Nephrology
Dr. Moxey-Mims will review the trends in pediatric kidney transplantation over the past decade, focusing on differences by race and ethnicity, whether the gaps have narrowed over that time and steps that can be taken to increase equity.
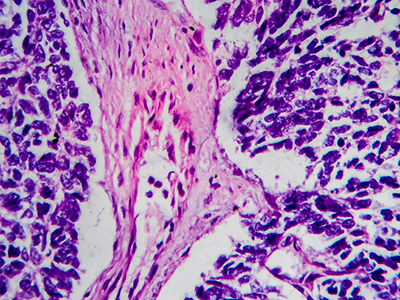
Prevalence of mycophenolate mofetil discontinuation and subsequent outcomes in pediatric kidney transplant recipients: A PNRC study
Presenter: Asha Moudgil, M.D., medical director, Kidney Transplant
Mycophenolate mofetil (MMF) is a common maintenance immunosuppressant in children receiving kidney transplants but is often discontinued for various reasons. In this multi-center Pediatric Nephrology Research Consortium study, researchers sought to determine the prevalence and reasons for MMF discontinuation and its association with patient and allograft outcomes. They will be discussing their methodology and results from this study.
Additional Children’s National staff involved in the study include:
Changes in diastolic function and cardiac geometry after pediatric kidney transplantation: A longitudinal study
Presenter: Kristen Sgambat, Ph.D., clinical assistant professor
Children with end stage kidney disease are at high risk for cardiovascular morbidities. Indicators of systolic function, such as ejection fraction and fractional shortening, are often preserved and may not reveal cardiac dysfunction until it is severe. Longitudinal changes in diastolic function and cardiac geometry have not been well studied.
Additional Children’s National staff involved in the study include:
COVID19 in pediatric kidney transplant recipients: Incidence, outcomes, and response to vaccine
Presenter: Asha Moudgil, M.D., medical director, Kidney Transplant
At the start of the pandemic, no information was available on the outcomes of pediatric kidney transplant recipients with COVID-19. When the COVID-19 vaccine became available, response of immunosuppressed children to the vaccine was not known. While more information has become available in adult transplant recipients, information on pediatric transplant recipients remains limited.
The team will discuss their methodology of collecting information and their results and conclusions.
Additional Children’s National staff involved in the study include:
- Christy Petyak, CPNP-PC, nurse practitioner
- Kristen Sgambat, Ph.D., clinical assistant professor
- Yi Shi, M.D., Nephrology fellow
- Melissa Meyers, M.D.,nephrologist
- Laurie Midgley, RN, kidney transplant coordinator
Poster presentation: Psychological functioning and adaptive behavior in pediatric patients awaiting renal transplantation.
Presenter: Kaushal Amatya, Ph.D., psychologist
Psychosocial functioning of children with chronic kidney disease (CKD) at pretransplant evaluation is associated with transplant readiness and post-transplant outcomes. Higher prevalence of emotional/behavioral issues is noted in children with CKD compared to healthy counterparts. Although issues with functional impairment is often reported, research on adaptive functioning using a validated measure is lacking. The study aimed to explore psychological and adaptive functioning in pediatric pre-transplant patients to identify areas in need of intervention.
Additional Children’s National staff involved in the study include:
- Asha Moudgil, M.D., medical director, Kidney Transplant
- Paige Johnson, Psychology resident


 A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.
A clinical trial testing a new drug to increase growth in children with short stature. The first ever high-intensity focused ultrasound procedure on a pediatric patient with neurofibromatosis. A low dose gene therapy vector that restores the ability of injured muscle fibers to repair. These were among the most popular articles we published on Innovation District in 2022. Read on for our full top 10 list.


 Children’s National Hospital is joining the International Pediatric Nephrology Association (IPNA) to bring care to children with kidney disease in Jamaica. With early screenings, diagnosis and optimal treatments, this collaboration will help decrease the morbidity and mortality associated with renal disease.
Children’s National Hospital is joining the International Pediatric Nephrology Association (IPNA) to bring care to children with kidney disease in Jamaica. With early screenings, diagnosis and optimal treatments, this collaboration will help decrease the morbidity and mortality associated with renal disease.
 Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its
Children’s National Hospital in Washington, D.C., was ranked No. 5 nationally in the U.S. News & World Report 2022-23 Best Children’s Hospitals annual rankings. This marks the sixth straight year Children’s National has made the list, which ranks the top 10 children’s hospitals nationwide. In addition, its 
 The International Pediatric Transplant Association (IPTA) is hosting their 11th Congress meeting March 26-29, 2022, and many Children’s National Hospital providers will be presenting throughout the conference. We hope you will join us!
The International Pediatric Transplant Association (IPTA) is hosting their 11th Congress meeting March 26-29, 2022, and many Children’s National Hospital providers will be presenting throughout the conference. We hope you will join us!
 In the most recent edition of the American Society of Pediatric Nephrology’s Kidney Notes, Children’s National Hospital Chief of Nephrology,
In the most recent edition of the American Society of Pediatric Nephrology’s Kidney Notes, Children’s National Hospital Chief of Nephrology,