Fetal MRI study reveals links between neighborhood disadvantages and brain development
New research from the Developing Brain Institute used fetal MRI to explore how neighborhood environments may influence brain development before birth.
New research from scientists at Children’s National Hospital suggests that the environments families live in may begin shaping brain development even before a baby is born. In a study published in the journal Cerebral Cortex, researchers from the Developing Brain Institute used fetal MRI to examine how neighborhood conditions are associated with brain growth during pregnancy.
The findings suggest that factors linked to neighborhood disadvantage may influence the trajectory of brain development in utero, particularly in regions involved in emotion, memory and stress regulation.
Mechanics of the study
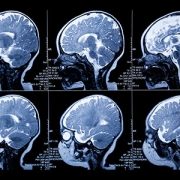
Neighborhood conditions are known to affect health outcomes across the lifespan. Children growing up in disadvantaged communities face higher risks for a range of health and developmental challenges. But researchers have long wondered when these effects first begin. To explore this question, investigators studied 199 healthy pregnancies in the Washington, D.C., region. Using advanced fetal MRI imaging, the team analyzed 298 brain scans collected between 18 and 39 weeks of gestation.
Researchers then compared patterns of fetal brain growth with neighborhood-level measures from the CDC’s Social Vulnerability Index. This index reflects factors such as income, education, housing conditions and access to transportation. The goal was to understand whether broader community conditions are associated with differences in early brain development.
What the study found
The study found that higher levels of neighborhood disadvantage were associated with differences in fetal brain development over the course of pregnancy. Early in gestation, fetuses from more disadvantaged neighborhoods showed slightly larger total brain volumes. As pregnancy progressed, however, the pattern shifted. By the end of gestation, those same fetuses tended to show smaller overall brain volumes and differences in subcortical brain structures.
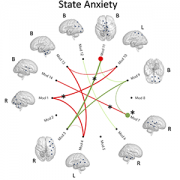
Many of the strongest effects were seen in limbic regions, which help regulate emotions, stress responses and memory. “These findings suggest that the developmental pathways linking neighborhood conditions and brain development may begin earlier than previously understood,” the authors wrote. The results align with previous research in children and adolescents showing that socioeconomic environments can influence brain structure and connectivity.
What this means
Because fetuses do not directly interact with their neighborhoods, the researchers believe the association likely occurs through maternal experiences during pregnancy. One possible pathway is maternal stress, which has been linked to hormonal changes that can affect fetal brain development. Other potential contributors include environmental exposures, nutrition and access to health resources. While the study cannot determine which factors drive the relationship, it highlights how broader community conditions may influence development before birth.
What’s Next
The research provides new insight into how social environments intersect with early brain development. Understanding when these differences emerge may help guide future public health strategies aimed at supporting families during pregnancy. “Our findings emphasize the importance of considering the communities mothers live in during pregnancy,” the authors wrote. By identifying when developmental trajectories begin to diverge, scientists hope to better understand where early interventions may have the greatest impact.
Read the full study “Prenatal experience of greater neighborhood disadvantage is associated with altered fetal volumetric brain growth in utero” in Cerebral Cortex here.
Authors from Children’s National include Kevin M. Cook, PhD; Josepheen De Asis-Cruz, PhD; Kushal Kapse; Kelsey Christoffel, MD; Caitlin McDermott; Nickie Andescavage, MD; and Catherine Limperopoulos, PhD.