Preliminary study points to efficacy of PBMC-based immunotherapy for neuroblastoma
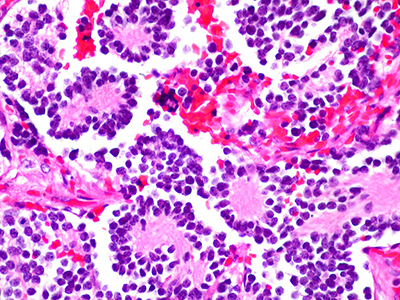
Pediatric solid tumors like neuroblastoma remain a major cause of illness and death, because traditional treatments effective for other tumor types have had only limited success.
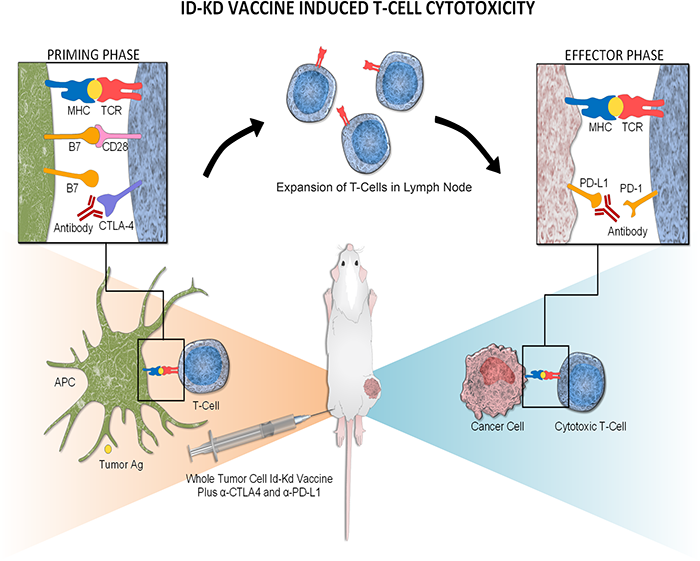
A novel immunotherapy approach that involves training autologous T cells derived from peripheral blood mononuclear cells (PBMCs) has shown early signs of safety and efficacy in a small pre-clinical model. The approach, pioneered by researchers within the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children’s National Hospital, may have future implications for the treatment of intractable pediatric solid tumors such as neuroblastoma.
What it means
Pediatric solid tumors like neuroblastoma remain a major cause of illness and death, because traditional treatments effective for other tumor types have had only limited success. There is an urgent need for innovative strategies to effectively target these tumors.
This study presents another approach to cell therapy that collects peripheral blood mononuclear cells from the patient and teaches them to target and eliminate solid tumor cells through exposure to similar tumor cells in a controlled laboratory setting. The cells are then primed to find and attack the solid tumor cells when reintroduced, an approach known as adoptive immunotherapy.
What’s new
The authors note that more well-established modalities such as CAR-T and TCR-T therapies have also made significant advances and demonstrate clinical promise at battling cancers such as neuroblastoma. However, the approach presented in the paper offers early promise of an additional potential strategy, especially in a context “where simplicity, speed and safety are priorities.”
By using small molecule treatments, the authors induce an immunogenic response in neuroblastoma tumor cells, making them more recognizable and attackable by immune cells. Ex vivo training of PBMCs with treated neuroblastoma cells exhibited strong tumor-killing activity.
The authors highlight a few key differences of this approach versus existing adoptive cellular therapies. The method “is technically straightforward, requiring only small tumor samples and peripheral blood mononuclear cells (PBMCs), and avoids the need for the complex genetic engineering intrinsic to CAR-T and TCR-T manufacturing,” the authors write. This method leverages ex vivo tumor cell modification, which may mitigate systemic toxicities. “Additionally, because our approach is not limited to a few surface antigens, it may expand the repertoire of actionable tumor-associated targets.”
Children’s National leads the way
Children’s National is a leader in the development of cell therapies for a wide range of pediatric conditions, including pediatric cancer, HIV/AIDS, sickle cell disease and others.
This research is the latest development in a decade-plus of focused research in adoptive immunotherapy within the Center for Cancer and Immunology Research and the Sheikh Zayed Institute for Pediatric Surgical Innovation.
What’s next
The authors write that there are several critical areas for future research to advance this approach, including a need to understand the specific T cell component and receptors involved in recognizing tumor antigens. They will also need to explore the longevity of the trained T cell response including studies of the memory and persistence of trained PBMCs to ensure lasting anti-tumor effects. Finally, the team will explore the risk of T cell exhaustion, which could reduce the effectiveness of the therapy over time.
Though work remains, the authors note, “Our findings lay the groundwork for developing this approach into a viable therapy for neuroblastoma and possibly other solid tumors as well.”
Read the full study: Trained autologous cytotoxic T cells derived from PBMCs or splenocytes for immunotherapy of neuroblastoma in Frontiers in Immunology.