Revolutionizing pediatric nephrology one patient at a time

“With each kidney transplant, we’re not just restoring the health of children, we’re pioneering progress and setting new benchmarks for the field of pediatric nephrology,” says Marva Moxey-Mims, M.D., chief of Nephrology at Children’s National Hospital.
“With each kidney transplant, we’re not just restoring the health of children, we’re pioneering progress and setting new benchmarks for the field of pediatric nephrology,” says Marva Moxey-Mims, M.D., chief of Nephrology at Children’s National Hospital. “We offer not just treatment, but a chance for our patients to flourish and live their best life.”
Big picture
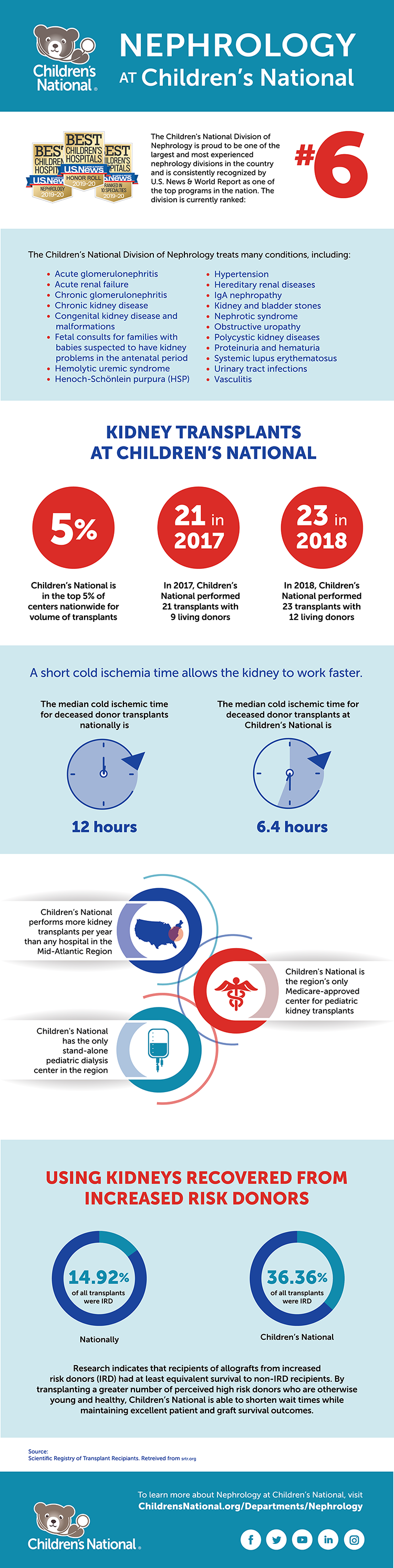
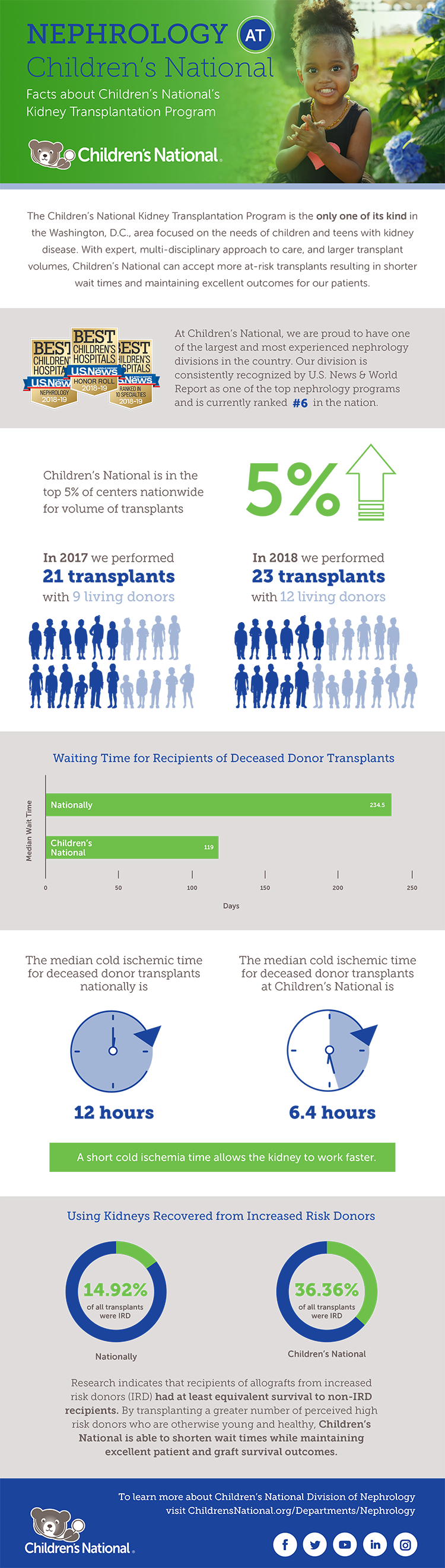
The Kidney Transplantation Program at Children’s National is the only one of its kind in the Washington, D.C., region focused on the needs of children and teens with kidney disease.
Performing an average of 15-20 kidney transplants per year, the program’s latest one- and three-year graft and patient survival data from the Scientific Registry for Transplant Recipients is 100%.
“Our program surpasses national numbers, reflecting a commitment to exceptional care, superior patient outcomes and a focus on setting a new standard for excellence in transplant support,” says Dr. Moxey-Mims.
Children’s National leads the way
At the forefront of groundbreaking treatment, the nephrology team is pioneering progress by providing innovative care and resources. One example is the hospital’s collaboration with the National Kidney Registry and MedStar Georgetown Transplant Institute’s Living Donor Program. This provides a greater chance of finding more suitable donors for difficult-to-match children and has resulted in altruistic donations.
“Through living kidney donations, we redefine the timeline for pediatric transplants. Children experience shorter waits and swifter paths to renewed health compared to the national wait list,” says Asha Moudgil, M.D., medical director of the Kidney Transplantation Program at Children’s National. “It’s a testament to our commitment to making a difference in every child’s life.”
That commitment is also seen in the hospital’s focus on making sure families have the resources they need during their medical journey.
“Caring for a child with renal disease is an immense challenge, not only for the young patients but also for their families. The demanding schedule of hospital visits, often three times a week, adds an extra layer of stress and disruption to daily life,” says Dr. Moudgil.
Through grant funding from donors and national programs, families with children undergoing kidney transplants and dialysis can receive financial assistance. In some cases, families receive up to $2,000 of mortgage or rental payment assistance. Families can also receive funds for medication co-pays and other expenses incurred around the time of the transplant.
“In the face of medical adversity, these funds become a source of support,” says Dr. Moudgil. “We make sure every family, regardless of financial constraints, can access the care and resources their child needs.”
Bottom line
Children’s National is at the forefront of pediatric kidney transplantation. “Our unique approach ensures every facet of a patient’s journey is considered,” says Dr. Moxey-Mims. “We’ve created an environment where success is not just a medical outcome but a comprehensive and sustained improvement in a child’s overall quality of life.”
Learn more about our latest advances in pediatric Nephrology.