The future of Wilms tumor therapies: Q&A with Jeffrey Dome, M.D., Ph.D.
Dr. Dome’s mission is to come up with other therapies to treat Wilms tumor.
Conducting Wilms tumor research has placed Children’s National Hospital as the leader in this space. Jeffrey Dome, M.D., Ph.D., talks about the future of Wilms tumor therapies and what excites him about being able to offer different treatment options to this group of patients.
Q: What’s next for Wilms tumor therapy treatments?
A: Something we’ve learned is that we’re reaching the maximum benefit that we can achieve with conventional chemotherapy and have hit a plateau with some of the high-risk Wilms tumors.
If you look at the history of Wilms tumor, we’ve ratcheted up the cure rate by adding more and more chemotherapy agents. We’re now at a point in which patients can’t tolerate having more toxic chemotherapy added to their regimen. Our mission is to come up with other therapies that work differently.
Q: What have you learned that can lead to new therapy options?
A: I was a co-investigator on a National Cancer Institute-funded project called the TARGET study, which entailed sequencing several hundred Wilms tumors to identify genetic mutations that could be druggable. While a wealth of knowledge was gained on the nature of the biology of Wilms tumor, only a minority of Wilms tumors have targetable mutations. We understand what the mutations are, but most are not mutations that lend themselves to drug therapy.
Therefore, we must think about other forms of therapy for Wilms tumor, such as immunotherapy. That’s where the work with the T cells targeting the WT1 protein Wilms tumor cells come into play.
Q: What do the new therapies look like?
A: I think the future of Wilms tumor therapy will be combining the classic chemotherapy and radiation therapy that we’ve used for many years but also adding another component for the patients with the highest risk of relapse, such as immunotherapy.
Up until now, the studies that we’ve conducted using immunotherapy have introduced this treatment very late as a last resort after everything else has failed. We have seen early signals of efficacy and if we can corroborate this in current studies, I believe the future will be to introduce immunotherapy earlier in the treatment course.
There has been success using immunotherapy in other pediatric solid tumors such as neuroblastoma. I believe the prospects for Wilms tumor are also exciting.


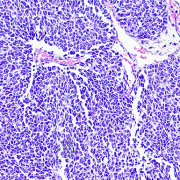
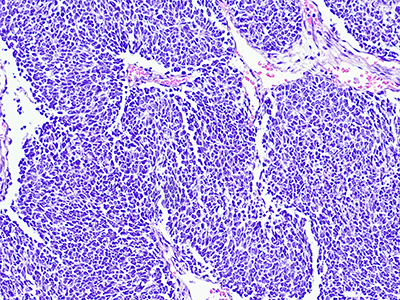



 Wilms tumor, also known as nephroblastoma, is the most common pediatric kidney cancer, typically seen in children ages three to four. Compared to patients with unilateral Wilms tumors, children with bilateral Wilms tumors (BWT) have poorer event-free survival (EFS) and are at higher risk for later effects such as renal failure. The treatment of BWT is challenging because it involves surgical removal of the cancer, while preserving as much healthy kidney tissue as possible to avoid the need for an organ transplant.
Wilms tumor, also known as nephroblastoma, is the most common pediatric kidney cancer, typically seen in children ages three to four. Compared to patients with unilateral Wilms tumors, children with bilateral Wilms tumors (BWT) have poorer event-free survival (EFS) and are at higher risk for later effects such as renal failure. The treatment of BWT is challenging because it involves surgical removal of the cancer, while preserving as much healthy kidney tissue as possible to avoid the need for an organ transplant.