Honor bestowed on Adré Jacques du Plessis, M.B.Ch.B.

Dr. du Plessis joins a distinguished group of Children’s National physicians and scientists who hold an endowed chair.
Children’s National Hospital named Adré Jacques du Plessis, M.B.Ch.B., as The People of the United Arab Emirates Distinguished Professor of Prenatal Pediatrics.
Dr. du Plessis serves as director of the Zickler Family Prenatal Pediatrics Institute, division chief of Prenatal and Transitional Pediatrics and director of the Prenatal-Neonatal Neuroscience Program at Children’s National. In addition, Dr. du Plessis is a professor of pediatrics and neurology at George Washington University School of Medicine.
The big picture
Dr. du Plessis joins a distinguished group of Children’s National physicians and scientists who hold an endowed chair. Children’s National is grateful to generous donors who altogether have funded 49 professorships.
Professorships support groundbreaking work on behalf of children and their families and foster new discoveries and innovations in pediatric medicine. These prestigious appointments reflect the recipient’s achievements and a donor’s commitment to advancing and sustaining knowledge.
Why it matters
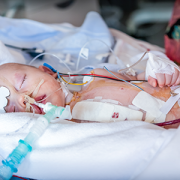
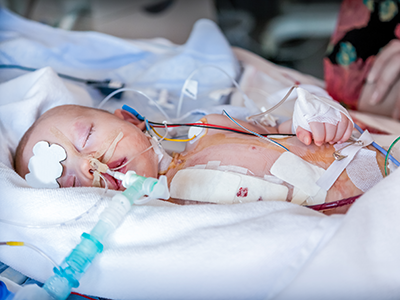
Dr. du Plessis is a leading international expert in the normal and abnormal development of the brain, as well as the mechanisms of injury to the immature brain. Under his leadership, the Zickler Family Prenatal Pediatrics Institute provides personalized specialty care to patients before, during and after their baby’s birth. His career-long research focus has been on the nervous system of the fetus and newborn, and the hazards for, and mechanisms of, brain injury and its prevention.
Moving the field forward
The Government of the United Arab Emirates (UAE) has established this distinguished professorship as part of its recent philanthropic commitment to Children’s National, which is set to further life-changing research breakthroughs and care for children worldwide. The UAE’s visionary generosity enables Dr. du Plessis and future holders of this professorship to launch bold new initiatives. The professorship will offer a platform to advance the field of prenatal, neonatal and maternal research and care while elevating our leadership and improving outcomes for children who need neonatal care.
Children’s National has been honored to treat patients from the UAE for more than 30 years, with more than 100 Emirati families traveling between the Emirates and Children’s National each year for advanced pediatric care and life-saving treatments.
The UAE’s long-standing philanthropic partnership with Children’s National has resulted in the 2009 establishment of the Sheikh Zayed Institute for Pediatric Surgical Innovation (SZI), the 2021 launch of the Children’s National Research and Innovation Campus, 82 U.S. patents and countless medical breakthroughs for kids and their families. Through this transformational partnership, the UAE and Children’s National are unlocking new possibilities for pediatric medicine and accomplishing what once was thought impossible. Children’s National remains deeply grateful to the UAE for its extraordinary support.