Nikki Posnack, Ph.D., assistant professor with the Children’s National Heart Institute, continues to explore how repeat chemical exposure from medical devices influences cardiovascular function.
In a review published in HeartRhythm, Nikki Posnack, Ph.D., an assistant professor at the Children’s National Heart Institute, and Larisa Tereshchenko, M.D., Ph.D., FHRS, a researcher with the Knight Cardiovascular Institute at Oregon Health and Science University, establish a strong foundation for a running hypothesis: Replacing BPA- and DEHP- leaching plastics for alternative materials used to create medical devices may help patients on dialysis, and others with impaired immune function, live longer.
While Drs. Tereshchenko and Posnack note clinical studies and randomized controlled trials are needed to test this theory, they gather a compelling argument by examining the impact exposure to chemicals from plastics used in dialysis have on a patient’s short- and long-term health outcomes, including sudden cardiac death (SCD).
“As our society modifies our exposure to plastics to mitigate health risks, we should think about overexposure to plastics in a medical setting,” says Posnack. “The purpose of the review in HeartRhythm is to gather data about the impact chemical compounds, leached from plastic devices, have on cardiovascular outcomes for patients spending prolonged periods of time in the hospital.”
In this review, the authors explore chemical risk exposures in a medical setting, starting with factors that influence sudden cardiac death (SCD) among dialysis patients.
Why study dialysis patients?
SCD in dialysis patients accounts for one-third of deaths in this population. This prompts a need to develop prevention strategies, especially among patients with end-stage renal disease (ESRD).
The highest mortality rate observed among dialysis patients is during the first year of hemodialysis, a dialysis process that requires a machine to take the place of the kidneys and remove waste from the bloodstream and replenish it with minerals, such as potassium, sodium and calcium. During this year, mortality during hemodialysis is observed more frequently during the first three months of treatment, especially among older patients.
Possible reasons for an increased risk of an earlier death include chemical exposure, which is casually associated with altered cardiac function, as well as genetic risks for irregular heart rhythms and heart failure. In the HeartRhythm review, Drs. Tereshchenko and Posnack analyze factors that influence mortality:
Hemodialysis treatment, dialysis, is associated with plastic chemical exposure
Drs. Tereshchenko and Posnack note that dialysis tubing and catheters are commonly manufactured using polyvinyl chloride (PVC) polymers. The phthalate plastics used to soften PVC can easily leech if exposed to lipid-like substances, like blood. Research shows phthalate chemical concentrations increase during a four-hour dialysis.
Di(2-ethylhexyl) phthalate (DEHP) is a common plastic used to manufacture dialysis tubes, thanks to its structure and economy.
Bisphenol-A (BPA) is another common material used in medical device manufacturing. From the membranes of medical tools to resins, or external coatings and adhesives, BPA leaves behind a chemical residue on PVC medical devices.
In reviewing the research, the authors find dialysis patients are often exposed to high levels of DEHP and BPA. The amount of exposure to these chemicals varies in regards to room temperature, time of contact, other circuit coatings and the flow rate of dialysis. A faster flow rate correlates with reductions in chemical leaching and lower mortality rates.
Plastic chemical exposure is casually associated with altered cardiac function
Drs. Tereshchenko and Posnack note a causal relationship already exists between chemicals absorbed from plastics and cardiovascular outcomes.
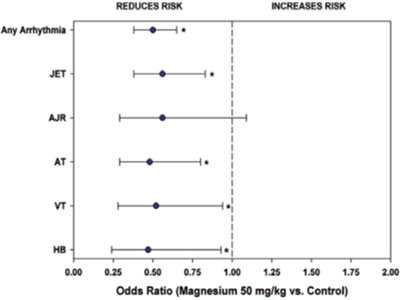
Dr. Posnack’s previous research found BPA concentrations impaired electrical conduction in neonatal cardiomyocytes – young, developing heart cells – potentially altering the heart’s normal rhythm and function.
To the best of their knowledge, no clinical research has been conducted on DEHP exposure and SCD. However, proof-of-concept models find in vivo phthalate exposure alters autonomic regulation, which can slow down natural heart-rate rhythm and create a lag in recovery time to stressful stimuli. For humans, this type of stressful stimulation would be equivalent to recovering from a bike ride, car accident, or in this case, ongoing dialysis treatment with impaired immune function.
In other models, BPA exposure has been shown to cause bradycardia, or a delayed heart rate. In excised whole heart models, BPA has also been shown to alter cardiac electrical activity.
Abnormal electrophysiological substrate in end-stage renal disease
Since the heart and kidneys work in tandem to transport blood throughout the body, and manage vital functions, such as our heart rate, blood flow and breathing, the authors cite additional factors that lead to ongoing heart and kidney problems, with a look at end-stage renal disease (ESRD).
General heart-function kidney risks include abnormal electrophysiological (EP) substrate, the underlying electrical activity of the cardiac tissue, and genetic risk factors, including the TBX3 gene, a gene associated with a unique positioning of the heart and SCD.
“We don’t want to cite alarm about having a medical procedure or about relying on external help, such as dialysis, for proper kidney function,” says Posnack. “Especially since dialysis is a life-saving medical intervention for patients with inadequate kidney function.”
Pre-existing abnormal EP substrate interacts with plastic chemical exposure in incident dialysis, which increases risk of SCD in genetically predisposed ESRD patients
To summarize their findings, Drs. Tereshchenko and Posnack list a handful of support areas, starting with observations about reductions in cardiovascular mortality and SCD following kidney transplants. They note hemodialysis catheters are associated with larger DEHP exposure and a higher risk of SCD, compared to arteriovenous fistulas, highways surgically created to connect blood from the artery to the vein.
Drs. Posnack and Tereshchenko also note a correlative observation about higher SCD rates observed six hours after hemodialysis, when peak levels of DEHP and BPA are circulating in the bloodstream.
To compare and control for these factors among dialysis patients, the researchers cite different mortality patterns with hemodialysis and peritoneal dialysis. Patients on hemodialysis experience higher mortality during the first year of treatment, compared to peritoneal dialysis, who have higher mortality rates after the second year of treatment. Hemodialysis relies on a machine to take the place of kidney function, while peritoneal dialysis relies on a catheter, a small tube surgically inserted into the stomach.
“Our goal is to build on our previous research findings by analyzing variables that have yet to be studied before, and to update the field of medicine in the process,” says Dr. Posnack. “This includes investigating the cardiovascular risks of using BPA- and DEHP-materials to construct medical devices. Ultimately, we hope to determine whether plastic materials contribute to cardiovascular risks, and investigate whether patients might benefit from the use of alternative materials for medical devices.
Drs. Tereshchenko and Posnack note that despite the associations between chemical exposure from medical devices and increased cardiovascular risks, there are no restrictions in the United States on the use of phthalates and BPA chemicals used to manufacture medical devices.
Their future research will explore how replacing BPA- and DEHP-leaching plastics influence mortality and morbidity rates of ESRD patients on dialysis, as well as other patients exposed to repeat chemical exposure, such as patients having cardiac surgery.
“We want to make sure we identify and then work to minimize any potential risks of plastic exposure in a medical setting,” adds Dr. Posnack. “Our goal is to put the health and safety of patients first.”
Dr. Posnack’s research is funded by two grants (R01HL139472, R00ES023477) from the National Institutes of Health.