Cas9-mediated correction of metabolic liver disease
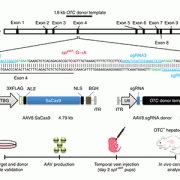
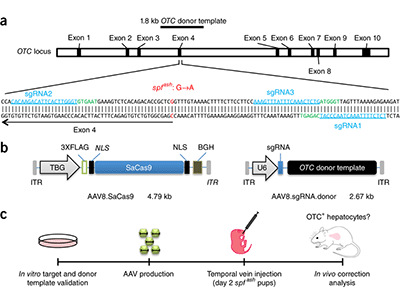 In vivo gene correction of the OTC locus in the mouse liver by AAV.CRISPR-SaCas9. Source: Nature Publishing Group copyright 2016. What’s knownA deficiency of the enzyme ornithine transcarbamylase (OTC) in humans causes life-threatening hyperammonemic crises. The OTC gene enables the body to make an enzyme that is a critical player in the urea cycle, a process that ensures excess nitrogen is excreted by the kidneys. Left unchecked, accumulating nitrogen becomes a toxic form of ammonia. Infants with OTC deficiency can suffer their first metabolic crisis as newborns. Up to 50 percent die or sustain severe brain injury, and survivors typically need a liver transplant by age 1. Gene therapy could cure OTC deficiency, but currently used viruses, such as adeno-associated virus (AAV), are not optimal in the neonatal setting. What’s newA research team led by Children’s National Health System and the University of Pennsylvania reasoned that the newborn liver may be an ideal setting for AAV-mediated gene correction using CRISPR-Cas9 gene editing. They intravenously infused two AAVs into two-day-old mice with partial OTC deficiency. One AAV expressed Cas9 and the other expressed a guide RNA and a donor OTC DNA. This resulted in correction of the mutation in 10 percent of liver cells and increased survival in mice challenged with a high-protein diet, which normally exacerbates disease. After consuming a high-protein diet for one week, the treated newborns had a 40 percent reduction in ammonia compared with the untreated group. The correction appears to last long term. The study “provides evidence for efficacy of gene editing in neonatal onset OTC deficiency,” says Mark L. Batshaw, M.D., Physician-In-Chief and Chief Academic Officer at Children’s National, and a study co-author. “This study provides convincing evidence for efficacy of in vivo genome editing in an authentic animal model of a lethal human metabolic disease,” the research team concludes. Questions for future researchQ: More than 400 mutations can cause OTC deficiency, and each would require a separate gene-editing approach. Is it possible instead to insert the OTC genome using CRISPR-Cas9 to correct the disorder irrespective of the mutation? Source: Yang, Y., L. Wang, P. Bell, D. McMenamin, Z. He, J. White, H. Yu, C. Xu, H. Morizono, K. Musunuru, M.L. Batshaw and J.M. Wilson. “A dual AAV system enables the Cas9-mediated correction of a metabolic liver disease in newborn mice.” Published Feb. 1, 2016 by Nature Biotechnology.
|