Expanding naloxone access could reduce adolescent overdose deaths
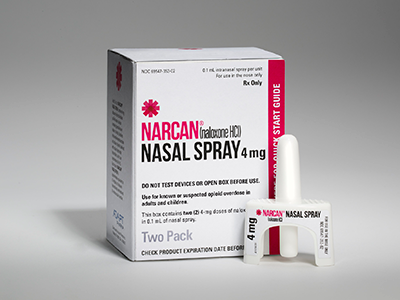
Naloxone, an opioid antagonist available as a nasal spray, rapidly reverses overdose by blocking opioid receptors.
Opioid-related deaths continue to remain high among adolescents. Naloxone, an opioid antagonist available as a nasal spray, rapidly reverses overdose by blocking opioid receptors. The medication is easy to administer, has minimal side effects and produces no effect if opioids are not present. Despite these advantages, naloxone is often not used even when another person is present during an adolescent’s overdose event.
In a recent clinical perspective in JAACAP Connect, researchers recommend that clinicians prescribe naloxone more broadly to adolescents and advocate for fewer restrictions on carrying the medication in schools and communities.
“Naloxone is a lifesaving intervention with minimal risk, yet many adolescents who could benefit from it do not have timely access,” said Sivabalaji Kaliamurthy, MD, director of the Addictions Program at Children’s National Hospital and lead author of the article. “Expanding prescribing practices and reducing stigma around naloxone are critical steps in preventing avoidable overdose deaths.”
The hold-up in the field
Several barriers continue to limit naloxone access for adolescents. Some school policies restrict students from carrying the medication, instead storing it in nurses’ offices, which may delay response during emergencies that occur outside the classroom or off campus. Stigma also remains a concern, with some administrators and community members believing that providing naloxone encourages substance use.
Confidentiality and consent laws can create additional challenges. Because regulations governing adolescent consent for substance use treatment vary by state, prescribing naloxone may sometimes require parental involvement, which can discourage youth from seeking care.
Moving toward solutions
The authors emphasize that evidence does not support the idea that increased naloxone availability leads to increased drug use. Studies examining expanded naloxone access laws have not found meaningful increases in heroin or injection drug use, suggesting that broader distribution can improve safety without worsening substance use.
To improve access, clinicians can screen adolescents routinely for overdose risk factors, prescribe naloxone more broadly and educate families and school personnel about the medication’s safety. Writing notes that allow students to self-carry naloxone, connecting patients with community distribution programs and advocating for clearer school and state policies may also help reduce barriers.
With overdose deaths continuing to be high among adolescents, the authors conclude that improving naloxone access is a practical and evidence-based way to reduce preventable fatalities.
Additional Children’s National authors include Laura Willing, MD.
Read the full clinical perspective, Prescribing Nasal Naloxone in Pediatrics: A Clinical Perspective on Reducing Overdose Deaths, in JAACAP Connect.







 Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.
Children’s National is ranked one of the top 10 pediatric hospitals in the nation by U.S. News & World Report. Our faculty and staff are proud of the impact made on the lives of children and families in our community. Your participation in the U.S. News & World Report annual reputational survey validates the quality of care we provide and reflects the mutual respect and trust we share as healthcare professionals.








 In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.
In 2025, Children’s National Hospital was featured in major national news outlets for pioneering advances in pediatric care, groundbreaking clinical research and powerful human stories of healing and hope. From gene therapy for sickle cell disease and innovative pacemakers for newborns to breakthrough transplants, cancer trials and emerging mental health concerns like AI psychosis, these stories highlight the hospital’s leadership across the full spectrum of pediatric medicine. The following ten highlights showcase the patients, families and experts behind this impact, as reported by outlets including NBC News, The Washington Post, Good Morning America, USA Today, Healio, ABC News and ESPN.












 Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.
Children’s National Hospital in Washington, D.C., was ranked as a top hospital in the nation by the U.S. News & World Report 2025-26 Best Children’s Hospitals annual rankings. This marks the ninth straight year Children’s National has made the Honor Roll list. The Honor Roll is a distinction awarded to only 10 children’s hospitals nationwide.

 Children’s National Hospital affiliates will participate in this year’s
Children’s National Hospital affiliates will participate in this year’s 