COVID-19’s impact on pregnant women and their babies

While pregnant women are at higher risk of severe illness, coronavirus can also trigger inflammatory and vascular responses in the placenta during critical periods of fetal development in symptomatic and asymptomatic cases.
Pregnant women should get vaccinated to minimize the detrimental health effects COVID-19 has on the placenta, the fetus and the newborn, states Roberta L. DeBiasi, M.D., M.S., division chief of Pediatric Infectious Diseases at Children’s National Hospital in an editorial published in The Journal of Infectious Diseases.
The editorial provides a comprehensive review of what is known about the harmful effects of SARS-CoV-2 infection in pregnant women themselves, the effects on their newborns, the negative impact on the placenta and what still is unknown amid the rapidly evolving field. The safety and efficacy of vaccination of pregnant women are also addressed.
While pregnant women are at higher risk of severe illness, the virus can also trigger inflammatory and vascular responses in the placenta during critical periods of fetal development in symptomatic and asymptomatic cases.
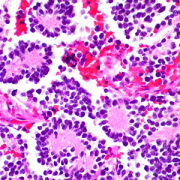
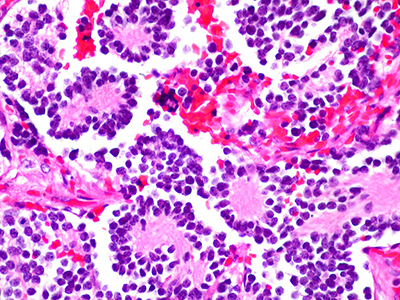
In this piece, Dr. DeBiasi comments on two related studies published in the same issue, Guan et al. and Shook et al., demonstrating pathologic findings in women’s placentas who had COVID-19 during pregnancy. Guan et al. published a detailed analysis of a stillbirth resulting from the delta variant infection during the third trimester.
“The authors present a highly plausible mechanism of stillbirth, namely that the virus-induced proinflammatory state ultimately led to placental abruption,” said Dr. DeBiasi.
Shook et al. presented a case series of pregnant women infected with the delta variant associated with stillbirth in two cases and one with severe neonatal illness.
“Taking the studies together, it’s evident that if a pregnant woman gets COVID-19 they’re at an increased risk of severe infection,” says DeBiasi. “They’re also at increased risk of adverse pregnancy outcomes, due to effects on the placenta, which may vary with specific circulating variants.”
Previous studies have documented that the placenta may be detrimentally affected by SARS-CoV-2 infection of the mother. However, maternal comorbidities such as hypertension, preeclampsia and gestational diabetes could also contribute to these findings.
“Despite these previous studies, the precise mechanisms of placental injury are still not clear and require further evaluation,” says Dr. DeBiasi. “Future research should include appropriate controls to better discern nonspecific versus SARS-CoV-2 specific effects and mechanisms of injury.”
Even though these potential risks exist, the vaccination rate among pregnant women is low. Dr. DeBiasi writes that recent publications have demonstrated vaccine efficacy and safety during pregnancy through programs that tracked the use in pregnant women. This data supports that COVID-19 vaccine offers another layer of protection to pregnant women since infants are not yet eligible for vaccination despite the fact that the youngest infants and children are among the most at risk among children for hospitalization.