New brain “pacemaker” offers new hope for refractory epilepsy
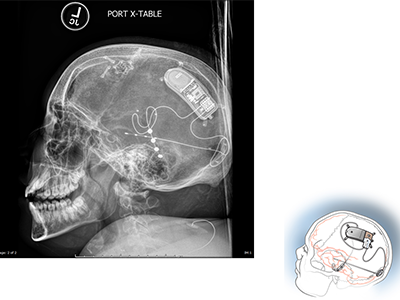
Example of NeuroPace RNS System placement.
If a child’s refractory seizures – seizures that don’t respond to medication – are originating in a part of the brain that is central to function (for example, impacting memory or verbal skills) the standard next step – surgical resection – is not an option for seizure reduction or relief. In most cases, these children are followed, more medications are tried, and other strategies attempted, but few viable options exist to ease their symptoms.
It’s possible that the next generation of implantable neurostimulators, which act as a type of pacemaker for the brain, might make a difference for some children previously left with no answers. Children’s National neurosurgeon Chima Oluigbo, M.D., in collaboration with the Comprehensive Pediatric Epilepsy Program at Children’s National, is looking at how these devices might be used to reduce or eliminate refractory seizures in pediatric patients. One example of this type of device is the RNS System.
“The RNS has been FDA approved for adults since 2013,” says Dr. Oluigbo, who recently implanted a NeuroPace RNS in the first pediatric patient at Children’s National, and one of the first young patients in the country. “The safety and efficacy data in the adult population, now gathered from a cohort of more than 800 adults, is showing positive outcomes so far. That allows pediatric neurosurgeons to consider an off-label use of this device for patients under the approved usage age of 18, when no other treatments exist.”
The RNS operates differently from previous neuro pacemaker-style devices. It is a “closed-loop” system that doesn’t require external activation once a seizure has started. Instead, the precise location of seizure origination is identified via functional magnetic resonance imaging (fMRI). Leads are then placed at the seizure site via surgery, and once activated, the RNS monitors and self-activates when pre-seizure electrical impulses are detected. The device responds by emitting a series of its own electrical impulses to interrupt and reset the brain’s seizure activity. The RNS system’s ability to continuously monitor the patient also allows physicians to get an inside look at the ongoing brain function of these young patients.
“Children’s National is one of the first places to apply the use of this device in children, because we are one of the few locations on the East coast with the multi-disciplinary expertise to implement it safely and effectively,” says Dr. Oluigbo. “Our clinical epilepsy team has been imaging and treating children with epilepsy for almost 30 years. With one of the oldest neurosurgical programs in country and our technological capabilities, Children’s National becomes the perfect location to explore how technology like this can improve the quality of life for our patients, many of whom have previously been told there is nothing more we can do to help.”
De-personalized data from patients who receive the NeuroPace RNS will be shared with the company in the hopes that the data will assist the FDA in assessing the appropriateness of extending the age range of approval from 18 and above to 12 and above.
“Our hope is to contribute to the body of data about this device and determine if it will improve the lives of our younger patients the way it has already been done for adults,” Dr. Oluigbo concludes. “Kids’ brains may respond differently, however, sharing our patients’ experiences and outcomes will help us identify whether or not this is a viable and promising option for more children with refractory epilepsy.”