Mutations in the NUP160 gene, which encodes one protein component of the nuclear pore complex nucleoporin 160 kD, are implicated in steroid-resistant nephrotic syndrome, an international team reports March 25, 2019, in the Journal of the American Society of Nephrology. Mutations in this gene have not been associated with steroid-resistant nephrotic syndrome previously.
“Our findings indicate that NUP160 should be included in the gene panel used to diagnose steroid-resistant nephrotic syndrome to identify additional patients with homozygous or compound-heterozygous NUP160 mutations,” says Zhe Han, Ph.D., an associate professor in the Center for Genetic Medicine Research at Children’s National and the study’s senior author.
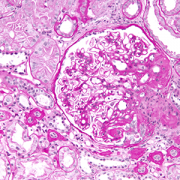
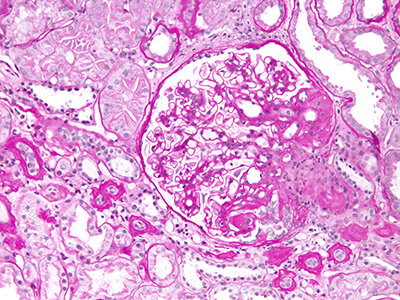
The kidneys filter blood and ferry waste out of the body via urine. Nephrotic syndrome is a kidney disease caused by disruption of the glomerular filtration barrier, permitting a significant amount of protein to leak into the urine. While some types of nephrotic syndrome can be treated with steroids, the form of the disease that is triggered by genetic mutations does not respond to steroids.
The patient covered in the JASN article had experienced persistently high levels of protein in the urine (proteinuria) from the time she was 7. By age 10, she was admitted to a Shanghai hospital and underwent her first renal biopsy, which showed some kidney damage. Three years later, she had a second renal biopsy showing more pronounced kidney disease. Treatment with the steroid prednisone; cyclophosphamide, a chemotherapy drug; and tripterygium wilfordii glycoside, a traditional therapy, all failed. By age 15, the girl’s condition had worsened and she had end stage renal disease, the last of five stages of chronic kidney disease.
An older brother and older sister had steroid-resistant nephrotic syndrome as well and both died from end stage kidney disease before reaching 17. When she was 16, the girl was able to receive a kidney transplant that saved her life.
Han learned about the family while presenting research findings in China. An attendee of his session said that he suspected an unknown mutation might be responsible for steroid-resistant nephrotic syndrome in this family, and he invited Han to work in collaboration to solve the genetic mystery.
By conducting whole exome sequencing of surviving family members, the research team found that the mother and father each carry one mutated copy of NUP160 and one good copy. Their children inherited one mutated copy from either parent, the variant E803K from the father and the variant R1173X, which causes truncated proteins, from the mother. The woman (now 29) did not have any mutations in genes known to be associated with steroid-resistant nephrotic syndrome.
Some 50 different genes that serve vital roles – including encoding components of the slit diaphragm, actin cytoskeleton proteins and nucleoporins, building blocks of the nuclear pore complex – can trigger steroid-resistant nephrotic syndrome when mutated.
With dozens of possible suspects, they narrowed the list to six variant genes by analyzing minor allele frequency, mutation type, clinical characteristics and other factors.
The NUP160 gene is highly conserved from flies to humans. To prove that NUP160 was the true culprit, Dr. Han’s group silenced the Nup160 gene in nephrocytes, the filtration kidney cells in flies. Nephrocytes share molecular, cellular, structural and functional similarities with human podocytes. Without Nup160, nephrocytes had reduced nuclear volume, nuclear pore complex components were dispersed and nuclear lamin localization was irregular. Adult flies with silenced Nup160 lacked nephrocytes entirely and lived dramatically shorter lifespans.
Significantly, the dramatic structural and functional defects caused by silencing of fly Nup160 gene in nephrocytes could be completely rescued by expressing the wild-type human NUP160 gene, but not by expressing the human NUP160 gene carrying the E803K or R1173X mutation identified from the girl’s family.
“This study identified new genetic mutations that could lead to steroid-resistant nephrotic syndrome,” Han notes. “In addition, it demonstrates a highly efficient Drosophila-based disease variant functional study system. We call it the ‘Gene Replacement’ system since it replaces a fly gene with a human gene. By comparing the function of the wild-type human gene versus mutant alleles from patients, we could determine exactly how a specific mutation affects the function of a human gene in the context of relevant tissues or cell types. Because of the low cost and high efficiency of the Drosophila system, we can quickly provide much-needed functional data for novel disease-causing genetic variants using this approach.”
In addition to Han, Children’s co-authors include Co-Lead Author Feng Zhao, Co-Lead Author Jun-yi Zhu, Adam Richman, Yulong Fu and Wen Huang, all of the Center for Genetic Medicine Research; Nan Chen and Xiaoxia Pan, Shanghai Jiaotong University School of Medicine; and Cuili Yi, Xiaohua Ding, Si Wang, Ping Wang, Xiaojing Nie, Jun Huang, Yonghui Yang and Zihua Yu, all of Fuzhou Dongfang Hospital.
Financial support for research described in this post was provided by the Nature Science Foundation of Fujian Province of China, under grant 2015J01407; National Nature Science Foundation of China, under grant 81270766; Key Project of Social Development of Fujian Province of China, under grant 2013Y0072; and the National Institutes of Health, under grants DK098410 and HL134940.