Prioritizing equity in pediatric care strategies following the pandemic

Societal inequities can affect differential health outcomes among various populations.
The emergence of the COVID-19 pandemic prompted an examination and acknowledgment of the societal inequities affecting differential health outcomes among various populations. There’s been a heightened focus on identifying and confronting the longstanding factors contributing to health disparities, particularly among historically marginalized and minoritized groups, which expose them to greater vulnerabilities during emergencies like a pandemic.
The hold-up in the field
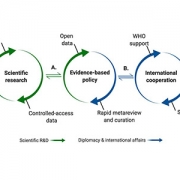
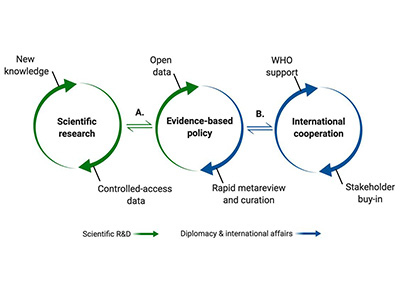
In a recent study published in Pediatric Clinics of North America, the authors introduce a focus on health equity and the social determinants of health as applied to an overarching framework of pandemic preparedness.
“There is little shared about the role of pediatricians in planning for disasters — like in the aftermath of the pandemic — with a focus on health equity,” says Joelle Simpson, M.D., M.P.H., chief of Emergency Medicine at Children’s National Hospital and co-author of the paper.
Moving the field forward
The authors discuss recommendations for pediatricians in planning for disasters with a focus on health equity and social determinants of health. In their observations, they consider:
- The COVID-19 pandemic amplified health inequities and placed tremendous strain on society, economy, education systems and healthcare infrastructure.
- Building on pediatric readiness frameworks is a recommended approach to creating a pediatric pandemic plan.
- Health equity considerations need to be built into all policies, processes and systems at the outset.
- The evolving national discourse on racism and inequities across communities including social injustices faced by minoritized groups is important framing for pandemic planning across all phases of disaster including preparedness, response and recovery.
The patient benefit
“Community engagement and serving as a trusted messenger and committed partner in health is critical in the process of recovery,” the authors write.
They emphasize the importance of integrating health equity viewpoints into communication planning and formulating strategies for community involvement. Fortunately, there has been a rise in opportunities to collaborate and gain insights from established networks dedicated to children’s pandemic or disaster readiness. These networks include the Emergency Medical Services for Children state programs, Pediatric Disaster Centers of Excellence and the Regional Pediatric Pandemic Network.
Children’s National Hospital leads the way
Children’s National, a lead hub for the Pediatric Pandemic Network (PPN), elevates the importance of planning for disasters as it pertains to children. Through the PPN, children’s hospitals in the country coordinate, prepare and enable high-quality, equitable, research-based pediatric care in emergencies, disasters and pandemics. The network prioritizes addressing the distinctive requirements and obstacles faced by children amid pandemics and disasters, emphasizing the importance of integrating health equity into emergency planning efforts.




 A group of children’s hospitals from across the country will coordinate on the response to future pandemics and other disasters through a new $29 million grant from the Health Resources and Services Administration (HRSA). The group, known as the Pediatric Pandemic Network (PPN), will focus on the unique needs and challenges to children during pandemics and disasters, ensuring that health equity is at the forefront of emergency planning.
A group of children’s hospitals from across the country will coordinate on the response to future pandemics and other disasters through a new $29 million grant from the Health Resources and Services Administration (HRSA). The group, known as the Pediatric Pandemic Network (PPN), will focus on the unique needs and challenges to children during pandemics and disasters, ensuring that health equity is at the forefront of emergency planning.


 The impact virtual school had on kids goes well beyond academics, say Children’s National Hospital experts in a new
The impact virtual school had on kids goes well beyond academics, say Children’s National Hospital experts in a new