JAMA study shows no benefit to nitric oxide in cardiopulmonary bypass for young children
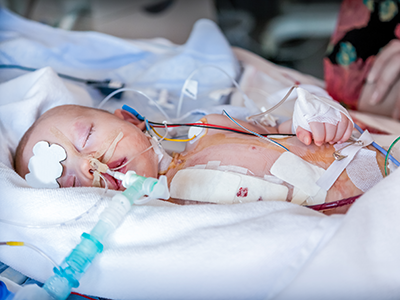
An international clinical trial showed that nitric oxide doesn’t help kids recover faster from cardiac surgery with cardiopulmonary bypass.
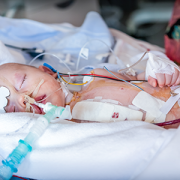
A study published in JAMA finds that the practice of introducing nitric oxide into the gas flow of the cardiopulmonary bypass oxygenator does not improve recovery or reduce the amount of time a child under age 2 needs to be on a ventilator after cardiac surgery.
Children’s National Cardiac Surgery Chief Yves d’Udekem, M.D., Ph.D., co-authored the international study, which is already leading to changes in how hospitals around the world care for children with congenital heart disease (CHD).
The results are from a double-blind, randomized controlled trial with more than 1,200 participants across six centers in Australia, New Zealand and the Netherlands. The research team found that children under age 2 who had cardiac surgery with cardiopulmonary bypass spent about the same number of days on ventilators after surgery, whether nitric oxide was used during surgery or not.
“These findings do not support the use of nitric oxide delivered into the cardiopulmonary bypass oxygenator during heart surgery,” the authors conclude.
What this means
Previous smaller, single center studies had shown early indications that nitric oxide delivered during heart surgery could possibly improve recovery and shorten the need for respiratory support after surgery by reducing the occurrence of low cardiac output syndrome in children under age 2.
This large-scale international trial showed that this is not the case.
Why it matters
Based on these earlier studies, many hospitals in the United States and around the world who perform critical heart surgery on young children with congenital heart disease had already started to incorporate nitric oxide into cardiopulmonary bypass. This new, more robust data is helping hospitals reassess this practice. Many are stopping it altogether based on the findings.
This work is an important reminder of how valuable well-designed, large-scale, double-blind, randomized, controlled trials are to defining, improving and refining best practices in clinical care.
Also, trials of this size and significance in pediatrics generally, and CHD specifically, take a very long time to complete, if they are ever able to be completed at all. That’s because the number of children with these conditions is relatively small and spread out, even though CHD is the most common birth defect in the world. The authors say it is a major accomplishment to have completed a trial of this size and in such a short time. Even better, the data gathered from this sample of patients from across international borders can be used to provide even more insights into how best to care for these children as they continue to grow and develop.