Cell therapy mitigates neurological impacts of cardiac surgery in pre-clinical model
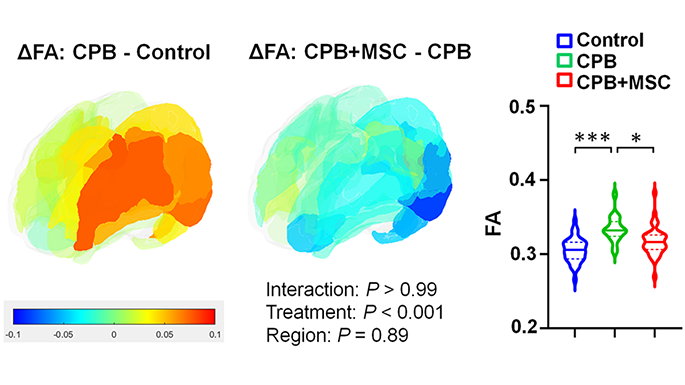
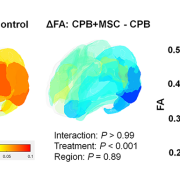
Differences of cortical fractional anisotropy between cardiopulmonary bypass and control (left), cardiopulmonary bypass + mesenchymal stromal cells and cardiopulmonary bypass (center), and 3 groups (right).
A pre-clinical study in the journal JACC: Basic to Translational Science shows that infusing bone marrow-derived mesenchymal stromal cells (BM-MSCs) during cardiac surgery provides both cellular-level neuroprotection for the developing brain and improvements in behavior alterations after (or resulting from) surgery.
What this means
According to lead author Nobuyuki Ishibashi, M.D., Oxidative and inflammatory stresses that are thought to be related to cardiopulmonary bypass cause prolonged microglia activation and cortical dysmaturation in the neonatal and infant brain. These issues are a known contributor to neurodevelopmental impairments in children with congenital heart disease.
This study found that, in a pre-clinical model, the innovative use of cardiopulmonary bypass to deliver these mesenchymal stromal cells minimizes microglial activation and neuronal apoptosis (cell death), with subsequent improvement of cortical dysmaturation and behavioral alteration after neonatal cardiac surgery.
Additionally, the authors note that further transcriptomic analyses provided a possible mechanism for the success: Exosome-derived miRNAs such as miR-21-5p, which may be key drivers of the suppressed apoptosis and STAT3-mediated microglial activation observed following BM-MSC infusion.
Why it matters
Significant neurological delay is emerging as one of the most important current challenges for children with congenital heart disease, yet few treatment options are currently available.
Applications of BM-MSC treatment will provide a new therapeutic paradigm for potential MSC-based therapies as a form of neuroprotection in children with congenital heart disease.
Children’s National Hospital leads the way
The Ishibashi lab is the first research team to demonstrate the safety, efficacy and utility of using cardiopulmonary bypass to deliver BM-MSCs with the goal of improving neurological impairments in children undergoing surgery for congenital heart disease. In addition to this pre-clinical research, a phase 1 clinical trial, MeDCaP, is underway at Children’s National.
Recent additional funding from the NIH will allow the team to identify molecular signatures of BM-MSC treatment and mine specific BM-MSC exosomes for unique cardiopulmonary bypass pathology to further increase understanding of precisely how and why this cell-based treatment shows success.