Katrina Adams, Ph.D., awarded fellowship to help restore functions in MS patients
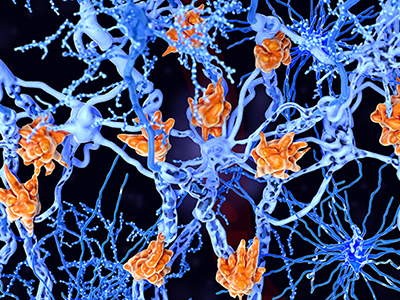
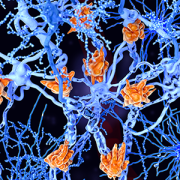
Multiple sclerosis is a demyelinating disease in which the insulating covers of nerve cells are damaged. Microglia cells (orange) attack the oligodendrocytes that form the insulating myelin sheath around neuron axons, leading to the destruction of the myelin sheath and to the loss of nerve function.
For her contributions to Multiple Sclerosis (MS) research, Katrina Adams, Ph.D., postdoctoral researcher at Children’s National Hospital, received the career transition fellowship from The National Multiple Sclerosis Society. The $600,000 fellowship will support a two-year period of advanced postdoctoral training in MS research and the first three years of research support in a new faculty appointment.
MS symptoms, including vision loss, pain, fatigue and reduced motor coordination, result from the demyelination of neuronal axons that transport critical information across the brain and spinal cord. Demyelination is the loss of myelin protein, which is normally produced by oligodendrocyte cells.
In the healthy brain, oligodendrocytes repair demyelinated areas by replacing damaged or lost myelin in a process called remyelination. Recent evidence has shown that oligodendrocytes display differences in their molecular and functional properties. One source of new oligodendrocytes in the adult brain is neural stem cells, which have been shown to generate oligodendrocytes that contribute to remyelination.
“The goal of this project is to determine whether neural stem cell-derived oligodendrocytes are distinct from other oligodendrocytes, both in the healthy brain and in MS,” said Adams. “I aim to understand the molecular mechanisms that regulate generation of oligodendrocytes from neural stem cells, with the goal of identifying signals that could be targeted in MS patients to promote remyelination.”
Remyelination is very limited in MS patients and current therapies for MS have very little impact on promoting remyelination.
This study will take advantage of the state-of-the-art facilities for single-cell analysis, transcriptomics, microscopy, and animal research in Children’s Research Institute at Children’s National. Adams also added that her postdoctoral mentor, Vittorio Gallo, Ph.D., interim chief academic officer and interim director of the Children’s National Research Institute, and principal investigator for the DC-IDDRC, has renowned expertise in glial biology, animal models of MS and white matter injury.
“This research will be the first to directly compare neural stem cell-derived oligodendrocytes with other resident oligodendrocytes in MS brain samples,” said Adams. “The results of this study will provide critical insight into the role that neural stem cells play in repair of MS demyelinated lesions.”
Adams received her doctorate in molecular biology from the University of California, Los Angeles where she used pluripotent stem cells to study motor neuron development. She currently investigates signaling pathways that regulate neural stem and progenitor cell maintenance and differentiation in the developing postnatal and adult brain, with a focus on the Endothelin-1 pathway. She is interested in understanding how stem and progenitor cells respond to disease or injury, such as in Multiple Sclerosis, with the hope of identifying new therapeutic targets.