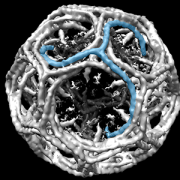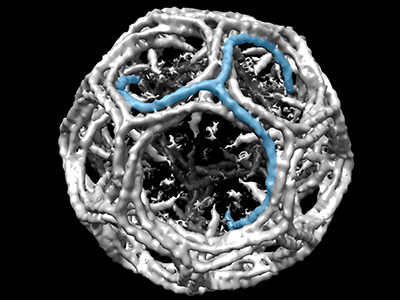
Children’s National Hospital in collaboration with the University of North Carolina Wilmington and Johns Hopkins University developed an enhanced autonomous strategy for laparoscopic soft tissue surgery.
A new approach to soft tissue surgery could simplify autonomous surgical planning and enable collaborative surgery between an autonomous robot and human, a new study published in Science Robotics finds. This is the first time a robot can complete an autonomous soft tissue surgical task under laparoscopic conditions, forming the foundation for future soft tissue surgeries.
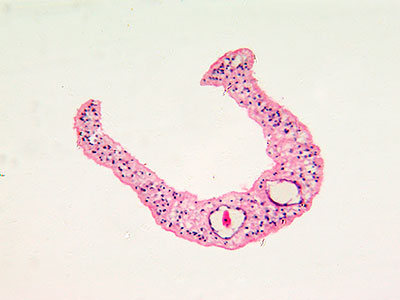
Children’s National Hospital in collaboration with the University of North Carolina Wilmington and Johns Hopkins University developed an enhanced autonomous strategy for laparoscopic soft tissue surgery. The multi-institutional effort made it possible to perform a robotic laparoscopic small bowel anastomosis in phantom and in vivo intestinal tissues. The findings further suggest that autonomous robot-assisted surgery has the potential to provide more efficacy, safety and consistency independent of an individual surgeon’s skill and experience.
The hold-up in the field
Autonomous anastomosis is known to be a challenging soft tissue surgery task. And in the laparoscopic setting, surgeries like these prove to be more challenging because of the need for high maneuverability and repeatability under motion and vision constraints – especially in pediatric patients.
“This work represents the first time autonomous soft tissue surgery has been performed using a laparoscopic technique and is the first step in bridging the gap between human and machine towards completing autonomous surgical tasks in soft tissue surgeries,” says Hamed Saeidi, Ph.D., assistant professor at University of North Carolina Wilmington and lead author of the study.
To overcome the unpredictable motions of the tissue, the experts used machine learning based techniques to track the dynamic motions of the soft tissue during the surgery. These methods also pave the way for markerless methods for tracking the tissue motion in future surgeries.
“Until now, laparoscopic autonomous surgeries were not possible in soft tissue due to the unpredictable motions of the tissue and limitations on the size and capabilities of surgical tools,” says Justin Opfermann, M.S., Ph.D., student and Johns Hopkins University and co-author.
What’s unique
Performing autonomous surgery would require the development of novel suturing tools, imaging systems and robotic controls to visualize a surgical scene, generate an optimized surgical plan and then execute that surgical plan with the highest precision.
The autonomous robot takes its skill one step further when performing surgical tasks on soft tissues by enabling a robot-human collaboration to complete more complicated surgical tasks where preoperative planning is not possible.
Additionally, the robot used in this work uses a novel shared control scheme called “conditional autonomy,” whereby the robot performs the majority of the surgical task, which the surgeon oversees.
Bottom line
“Combining all of these features into a single system is non-trivial,” Opfermann adds. “In 2016, we were the first group to demonstrate feasibility of semi-autonomous small bowel anastomosis with a robot in soft tissue, and now we can perform autonomous laparoscopic anastomosis.”
The resulting anastomosis had more consistency and achieved higher burst strength than surgeons suturing with manual technique, resulting in less anastomotic leak.
In laparoscopic surgeries – and pediatric patients especially – these challenges are even more difficult due to the small size of the patient. Robotic anastomosis is one way to ensure that surgical tasks that require high precision and repeatability can be performed with more accuracy and precision in every patient independent of surgeon skill.
“As a surgeon, I can attest to the potential benefits of improving how we perform surgery on our patients,” says Michael Hsieh, M.D., Ph.D., director of Transitional Urology at Children’s National Hospital. “Working with my engineering colleagues at Johns Hopkins, we’ve been able to develop prototypes of supervised, autonomous suturing robots that may be a step towards such improvements.”