Clinical Trial Spotlight: Can Nivolumab make cellular therapy more effective for treating relapsed lymphomas?
Each year, about 9,000 new patients are diagnosed with Hodgkin lymphoma, 10-15% of them children.
Each year, about 9,000 new patients are diagnosed with Hodgkin lymphoma, 10-15% of them children. Despite a relatively high cure rate for children with Hodgkin lymphoma, there are many debilitating long-term side effects of the treatments currently used. Additionally, 15-20% of children have a relapse and only half of them experience a long-term cure. Diffuse large B cell Lymphomas are another type of aggressive lymphoma that are difficult to cure, especially when they do not respond to upfront chemotherapy (refractory). Patients who experience relapse have to undergo more intensive chemotherapy followed by autologous stem cell transplantation and yet often times their lymphoma comes back.
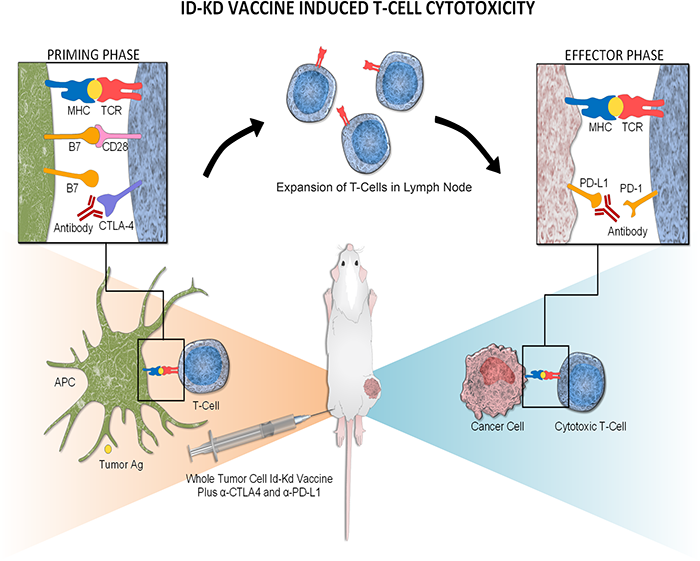
Physicians at Children’s National Hospital, in partnership with the Huntsman Cancer Institute at the University of Utah School of Medicine, are enrolling patients in a clinical trial to test the safety of administering PD-1 inhibitor Nivolumab given prior to and following the infusions of the patients’ own TAA-T cells which have been trained to target tumor cells in the laboratory. Nivolumab is currently approved by the FDA for relapsed Hodgkin lymphoma. Nivolumab acts by unleashing the brakes put on by the lymphoma cells, and by doing so, Nivolumab allows the immune system to overcome the tumor’s escape mechanism.
“We believe that if our T cells are deemed safe when given in combination with already approved drugs, we may be able to impact multiple lives and reduce long-term toxicities from conventional chemotherapies,” said Hema Dave, M.D., an oncologist at Children’s National. “We’re hopeful that combination immunotherapies will produce more durable responses than when immunotherapies are given alone as a single agent and, additionally, that they will reduce the use of cytotoxic chemotherapy.”
The investigators will collect blood from the patients to isolate peripheral blood mononuclear cells. They will then make special cells called dendritic cells to stimulate the T cells. Then they will add special mixtures of tumor proteins WT1, PRAME and Survivin and provide a cytokine milieu favorable to T cell expansion/activation, inducing selective expansion of T cells targeted to kill tumor cells. This process trains the T cells to recognize the tumor proteins and become specialized TAA-T cells. The cells will be grown and frozen until ready for use. While the T cells are growing, the patients will be given Nivolumab.
“We’re really trying to test if priming the patients with Nivolumab will make their T cells more effective when they get infused,” says Dr. Dave. “The Nivolumab will help prepare the immune system. Then, when we infuse the T cells, our hope is that the environment is primed for the T cells to expand, grow and work to attack the cancer. If we can prime the immune system and make it more conducive for the T cells, then maybe they will have a better chance to get to the lymphoma cells and thus have a more sustained response.”
Patients will then receive two infusions of the TAA-T cells and be monitored for side effects. The anticipated enrollment is 18 patients over the next 2-3 years. If there is a positive response in patients enrolled in this safety trial, it could expand to test for efficacy of the novel combination immunotherapy.
- PI: Hema Dave, M.D.
- Status: Recruiting
For more information about this trial, contact:
Hema Dave, M.D.
202-476-6397
hkdave@childrensnational.org
Fahmida Hoq, MBBS, MS
202-476-3634
fhoq@childrensnational.org
Click here to view Open Phase 1 and 2 Cancer Clinical Trials at Children’s National.
The Children’s National Center for Cancer and Blood Disorders is committed to providing the best care for pediatric patients. Our experts play an active role in innovative clinical trials to advance pediatric cancer care. We offer access to novel trials and therapies, some of which are only available here at Children’s National. With research interests covering nearly every aspect of pediatric cancer care, our work is making great advancements in childhood cancer.