For an elementary school art project, Michael H. Hsieh, M.D., Ph.D., was instructed to fashion a coat of arms out of clay. In addition to panels for truth, justice and Taiwan, in the shield’s M.D. panel, a snake twists around a rod, like the staff for Asclepius, a Greek god associated with healing.
Children’s urologist Michael H. Hsieh, M.D., Ph.D., knew from age 10 that he would become a doctor. Proof is at his parents’ home. For an elementary school art project, students were instructed to fashion a coat of arms out of clay. In addition to panels for truth, justice and Taiwan, in the shield’s M.D. panel, a snake twists around a rod, like the staff for Asclepius, a Greek god associated with healing.
“I liked science. When I can use it to help patients, that is very rewarding,” says Dr. Hsieh, the first doctor in his family.
These days, Dr. Hsieh’s Twitter profile serves as a digital coat of arms, describing him as “tinker, tailor,” #UTI #biologist, epithelial #immunologist, helminthologist and #urologist.
Tinker/tailor is shorthand for the mystery drama, “Tinker Tailor Solider Spy,” he explains, adding that the “tinker” part also refers “to the fact that I am always questioning things, and science is about experimentation, trying to seek answers to questions.”
While still in medical school during a rotation Dr. Hsieh saw a bladder operation on a young child and thought it was “amazing.” That experience in part inspired Dr. Hsieh to become a urologist and bladder scientist. His training in immunology and study of the bladder naturally led him to study urinary tract infections and parasitic worms that affect the urinary tract. In addition, thanks to R01 funding from the National Institutes of Health (NIH), Dr. Hsieh is co-principal investigator with Axel Krieger, University of Maryland, and Jin U. Kang, Johns Hopkins, on a project to develop imaging robots for supervised autonomous surgery on soft tissue.
The $1 million in NIH funding pushes the boundaries on amazing by using multi-spectral imaging technology and improved techniques to reduce surgical complications.
Anastomosis is a technique used by surgeons to join one thing to another, whether it’s a vascular surgeon suturing blood vessels, an orthopedic surgeon joining muscles or a urologist stitching healthy parts of the urinary tract back together. Complications can set in if their stitching is too tight, prompting scar tissue to form, or too loose, letting fluid seep out.
“The human eye can see a narrow spectrum of electromagnetic radiation. These multi-spectral imaging cameras would see across greater set of wavelengths,” he says.
The project has three aims: figuring out the best way to place sutures using multi-spectral imaging, accurately tracking soft tissue as they model suturing and comparing the handicraft of a robot against anastomosis hand-sewn by surgeons.
“I like challenges, and I like new things. I am definitely not interested in doing permutations of other people’s work,” Dr. Hsieh explains. “I would much rather go on a path that hasn’t been tread. It is more difficult in some ways, but on a day-to-day basis, I know I am making a contribution.”
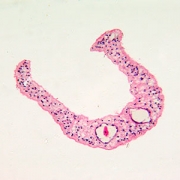
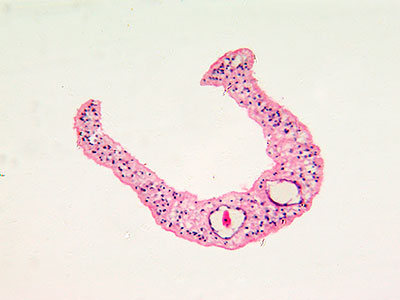
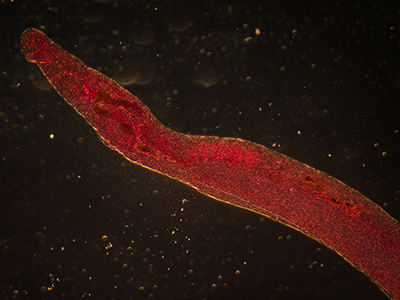
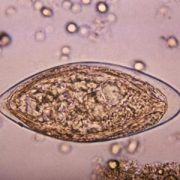
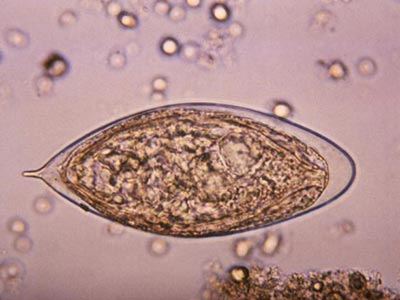
In another innovative research project, Dr. Hsieh leveraged a protein secreted by a parasitic worm, Schistosoma haematobium, that suppresses inflammation in hosts as a new therapeutic approach for chemotherapy-induced hemorrhagic cystitis, a form of inflammation of the bladder.
Watching his first surgery nearly 30 years ago, he had no idea robots might one day vie to take over some part of that complicated procedure, or that parasite proteins could be harnessed as drugs. However, he has a clear idea which innovations could be on the horizon for urology in the next three decades.
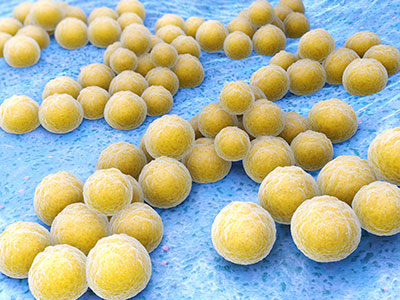
“My hope is 30 years from now, we will have a solid UTI vaccine and more non-antibiotic therapies. UTIs are the second-most common bacterial infection in childhood and, in severe cases, can contribute to kidney failure,” he says.
Globally, parasitic worms pose an ongoing challenge, affecting more than 1 billion worldwide – second only to malaria. People persistently infected by schistosome worms fail to reach their growth potential, struggle academically and lack sufficient energy for exercise or work.
“There is a feeling that the infection prevalence might be decreasing globally, but not as quickly as everyone hopes. In 30 years perhaps with more mass drug administration and additional drugs – including a vaccine – we’ll have it close to eliminated globally. It would become more like polio, casting a slim shadow with small pockets of infection here or there, rather than consigning millions to perpetual poverty.”